Initial_gene_level
Briana Mittleman
5/26/2018
Last updated: 2018-05-26
workflowr checks: (Click a bullet for more information)-
✔ R Markdown file: up-to-date
Great! Since the R Markdown file has been committed to the Git repository, you know the exact version of the code that produced these results.
-
✔ Environment: empty
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
-
✔ Seed:
set.seed(12345)The command
set.seed(12345)was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible. -
✔ Session information: recorded
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
-
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility. The version displayed above was the version of the Git repository at the time these results were generated.✔ Repository version: 2076ce9
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can usewflow_publishorwflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.Ignored files: Ignored: .Rhistory Ignored: .Rproj.user/ Untracked files: Untracked: data/gene_cov/ Untracked: data/reads_mapped_three_prime_seq.csv
Expand here to see past versions:
| File | Version | Author | Date | Message |
|---|---|---|---|---|
| Rmd | 2076ce9 | Briana Mittleman | 2018-05-26 | initial commit, gene level analysis |
I will use this analysis to take a look at the initial protein conding gene counts.
library(workflowr)Loading required package: rmarkdownThis is workflowr version 1.0.1
Run ?workflowr for help getting startedlibrary(ggplot2)
library(dplyr)
Attaching package: 'dplyr'The following objects are masked from 'package:stats':
filter, lagThe following objects are masked from 'package:base':
intersect, setdiff, setequal, unionlibrary(edgeR)Warning: package 'edgeR' was built under R version 3.4.3Loading required package: limmaWarning: package 'limma' was built under R version 3.4.3Imput the data that was created from my coding gene rule in the snakefile.
N_18486_cov= read.table("../data/gene_cov/YL-SP-18486-N_S10_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_18486_cov= read.table("../data/gene_cov/YL-SP-18486-T_S9_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_18497_cov= read.table("../data/gene_cov/YL-SP-18497-N_S12_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_18497_cov= read.table("../data/gene_cov/YL-SP-18497-T_S11_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_18500_cov= read.table("../data/gene_cov/YL-SP-18500-N_S20_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_18500_cov= read.table("../data/gene_cov/YL-SP-18500-T_S19_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_18505_cov= read.table("../data/gene_cov/YL-SP-18505-N_S2_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_18505_cov= read.table("../data/gene_cov/YL-SP-18505-T_S1_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_18508_cov= read.table("../data/gene_cov/YL-SP-18508-N_S6_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_18508_cov= read.table("../data/gene_cov/YL-SP-18508-T_S5_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_18853_cov= read.table("../data/gene_cov/YL-SP-18853-N_S32_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_18853_cov= read.table("../data/gene_cov/YL-SP-18853-T_S31_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_18870_cov= read.table("../data/gene_cov/YL-SP-18870-N_S24_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_18870_cov= read.table("../data/gene_cov/YL-SP-18870-T_S23_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19128_cov= read.table("../data/gene_cov/YL-SP-19128-N_S30_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19128_cov= read.table("../data/gene_cov/YL-SP-19128-T_S29_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19141_cov= read.table("../data/gene_cov/YL-SP-19141-N_S18_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19141_cov= read.table("../data/gene_cov/YL-SP-19141-T_S17_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19193_cov= read.table("../data/gene_cov/YL-SP-19193-N_S22_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19193_cov= read.table("../data/gene_cov/YL-SP-19193-T_S21_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19209_cov= read.table("../data/gene_cov/YL-SP-19209-N_S16_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19209_cov= read.table("../data/gene_cov/YL-SP-19209-T_S15_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19223_cov= read.table("../data/gene_cov/YL-SP-19223-N_S8_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19223_cov= read.table("../data/gene_cov/YL-SP-19233-T_S7_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19225_cov= read.table("../data/gene_cov/YL-SP-19225-N_S28_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19225_cov= read.table("../data/gene_cov/YL-SP-19225-T_S27_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19238_cov= read.table("../data/gene_cov/YL-SP-19238-N_S4_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19238_cov= read.table("../data/gene_cov/YL-SP-19238-T_S3_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19239_cov= read.table("../data/gene_cov/YL-SP-19239-N_S14_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19239_cov= read.table("../data/gene_cov/YL-SP-19239-T_S13_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
N_19257_cov= read.table("../data/gene_cov/YL-SP-19257-N_S26_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))
T_19257_cov= read.table("../data/gene_cov/YL-SP-19257-T_S25_R1_001-genecov.txt", stringsAsFactors = FALSE, header = F, col.names = c("chr", "start", "end", "gene", "score", "strand", "count" ))Look at the total libraries first:
total_count_matrix=cbind(T_18486_cov$count, T_18497_cov$count, T_18500_cov$count, T_18505_cov$count, T_18508_cov$count, T_18853_cov$count, T_18870_cov$count, T_19128_cov$count, T_19141_cov$count, T_19193_cov$count, T_19209_cov$count, T_19223_cov$count, T_19225_cov$count, T_19238_cov$count,T_19239_cov$count, T_19257_cov$count)
#gene length vector
gene_length=T_18497_cov %>% mutate(genelength=end-start) %>% select(genelength)
gene_length_vec=as.vector(gene_length$genelength)
total_count_matrix_cpm=cpm(total_count_matrix, log=T, gene.length=gene_length_vec )Plot distribution of log2 cpm for total libraries.
plotDensities(total_count_matrix_cpm, legend = "bottomright", main="Pre-filtering total fraction")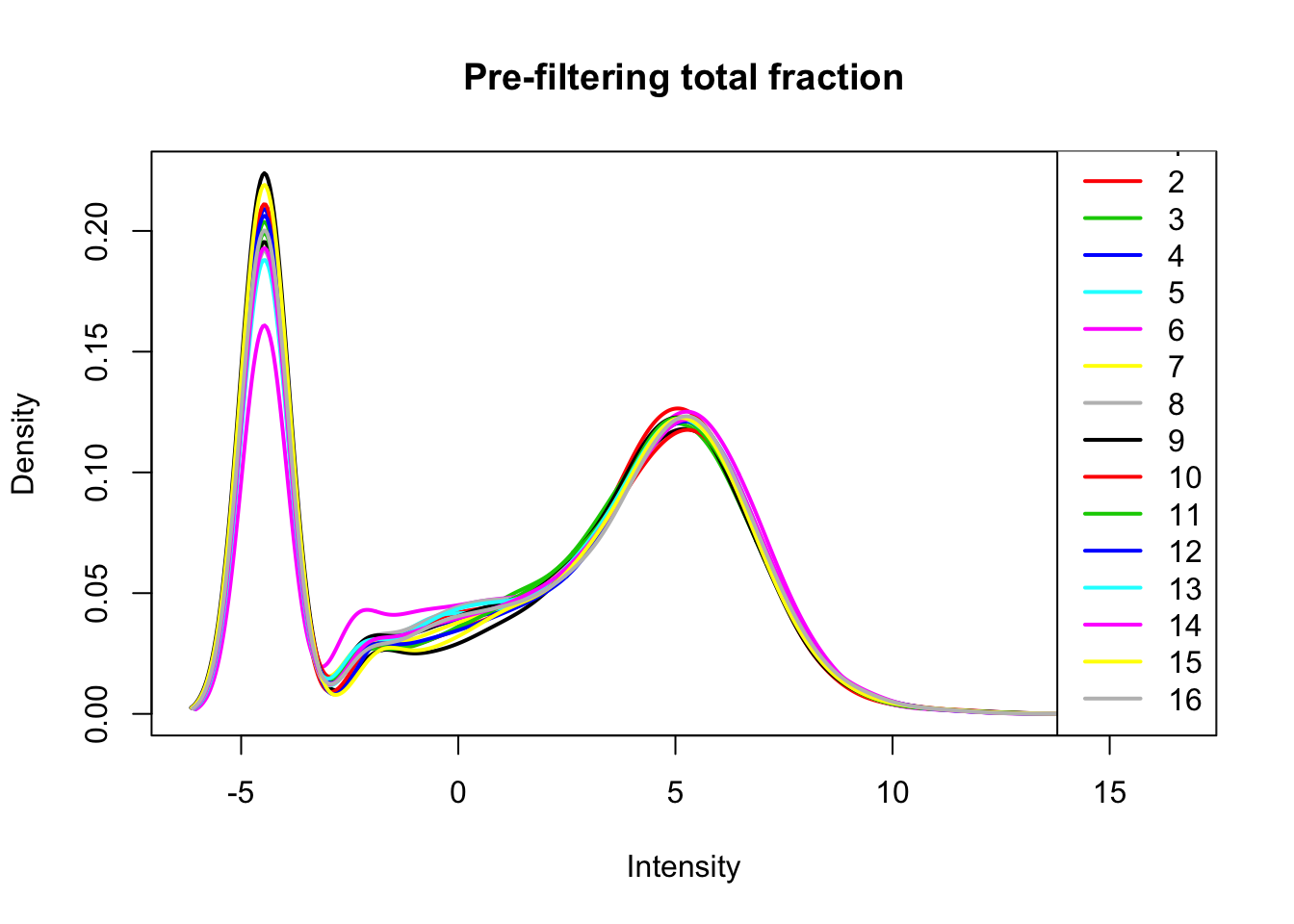
Look at gene distributions for the nuclear fractions.
nuclear_count_matrix=cbind(N_18486_cov$count, N_18497_cov$count, N_18500_cov$count, N_18505_cov$count, N_18508_cov$count, N_18853_cov$count, N_18870_cov$count, N_19128_cov$count, N_19141_cov$count, N_19193_cov$count, N_19209_cov$count, N_19223_cov$count, N_19225_cov$count, N_19238_cov$count,N_19239_cov$count, N_19257_cov$count)
#cpm
nuclear_count_matrix_cpm=cpm(nuclear_count_matrix, log=T, gene.length=gene_length_vec )Plot distribution of log2 cpm for nuclear libraries.
plotDensities(nuclear_count_matrix_cpm, legend = "bottomright", main="Pre-filtering nuclear fraction")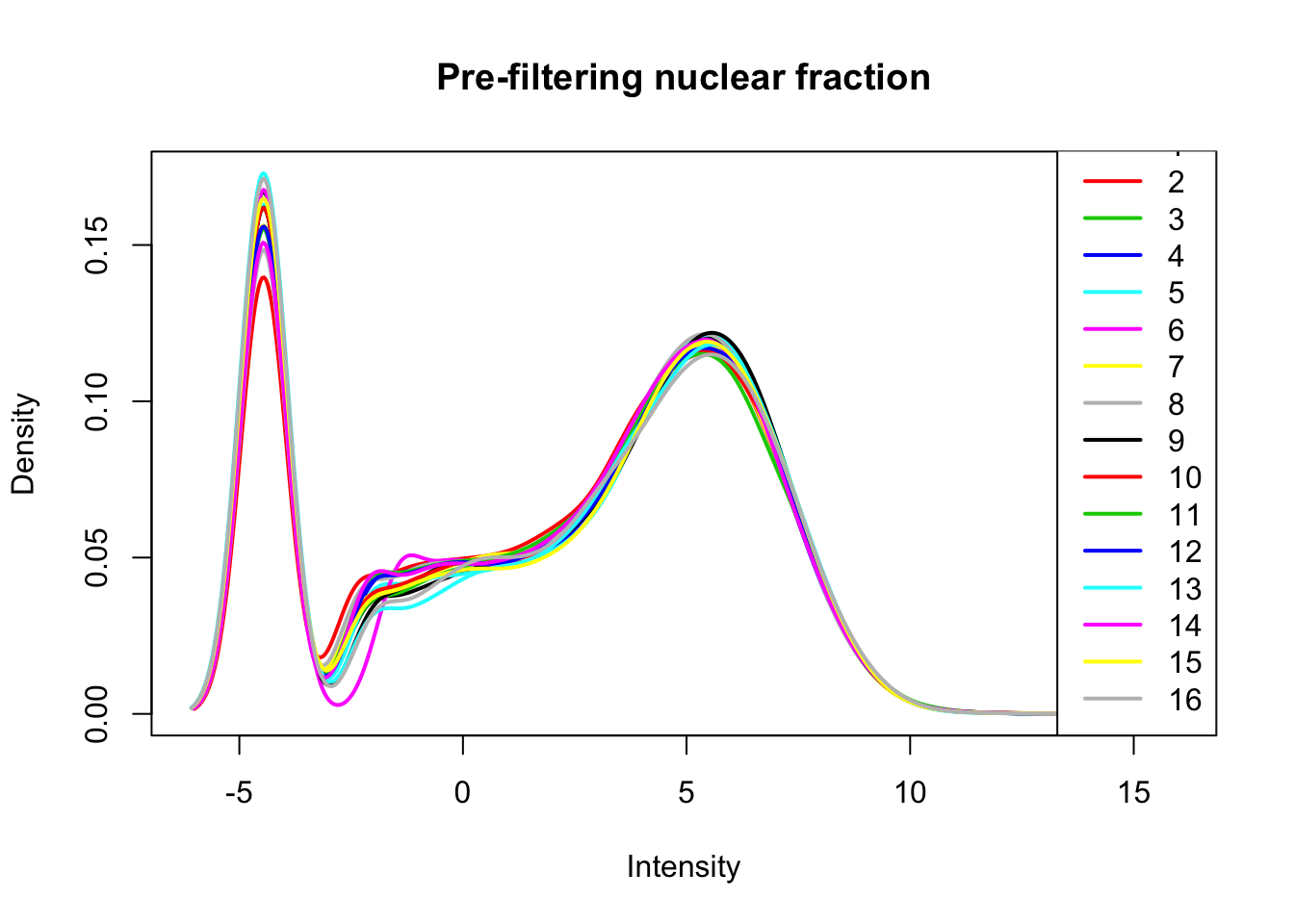
The distributions look similar. I can filter based on alll of the libraries. I will filter for 1cpm in more than half of the libraries. After this I can ask how many genes are detected in each library.
all_count_matrix=cbind(T_18486_cov$count, T_18497_cov$count, T_18500_cov$count, T_18505_cov$count, T_18508_cov$count, T_18853_cov$count, T_18870_cov$count, T_19128_cov$count, T_19141_cov$count, T_19193_cov$count, T_19209_cov$count, T_19223_cov$count, T_19225_cov$count, T_19238_cov$count,T_19239_cov$count, T_19257_cov$count,N_18486_cov$count, N_18497_cov$count, N_18500_cov$count, N_18505_cov$count, N_18508_cov$count, N_18853_cov$count, N_18870_cov$count, N_19128_cov$count, N_19141_cov$count, N_19193_cov$count, N_19209_cov$count, N_19223_cov$count, N_19225_cov$count, N_19238_cov$count,N_19239_cov$count, N_19257_cov$count )
#cpm
all_count_matrix_cpm=cpm(all_count_matrix, log=T, gene.length=gene_length_vec )
plotDensities(all_count_matrix_cpm, legend = "bottomright", main="Pre-filtering all libraries")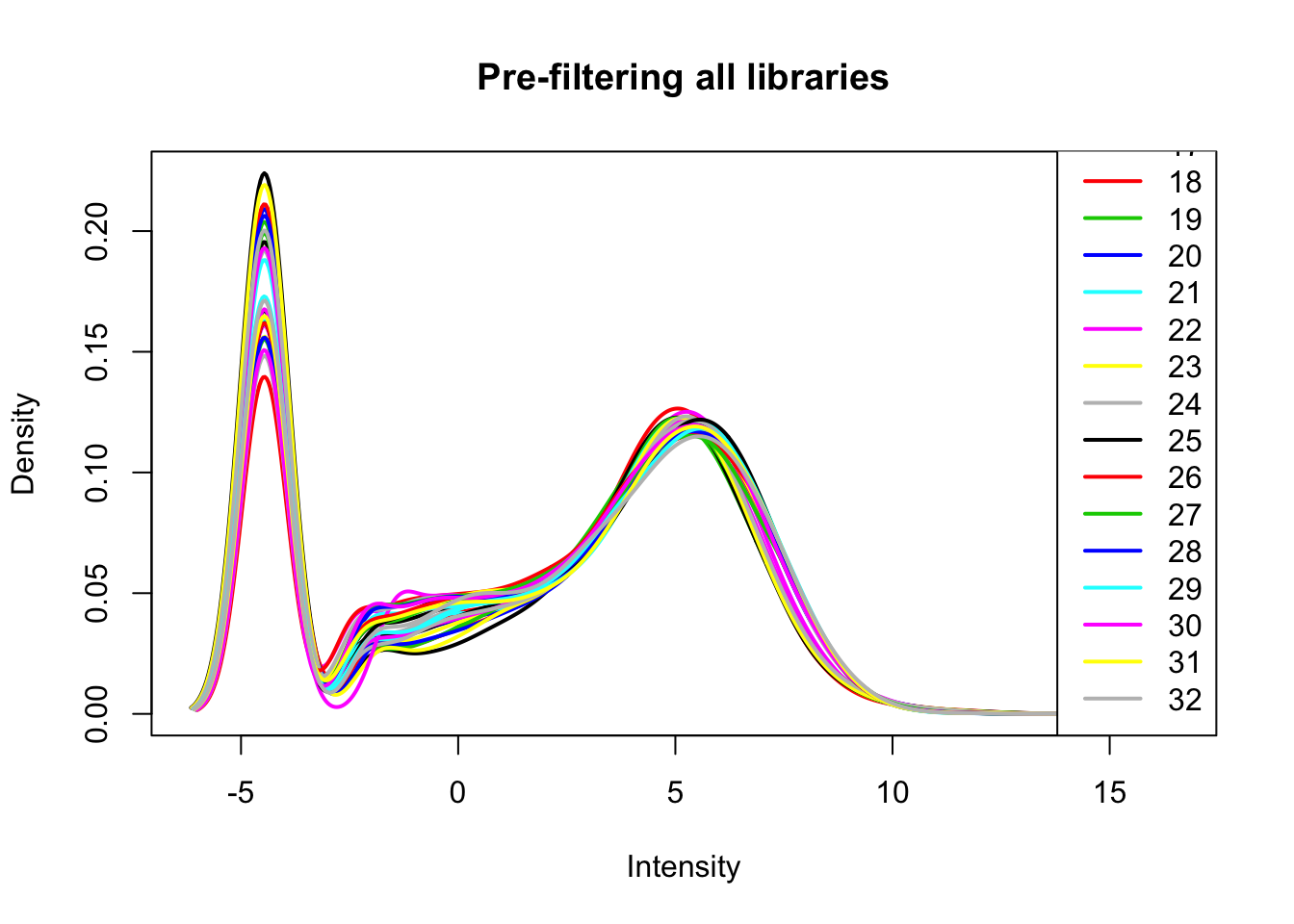
Filter:
keep.exprs=rowSums(all_count_matrix_cpm>1) >= 16
all_count_matrix_cpm_filt= all_count_matrix_cpm[keep.exprs,]
plotDensities(all_count_matrix_cpm_filt, legend = "bottomright", main="Post-filtering all libraries")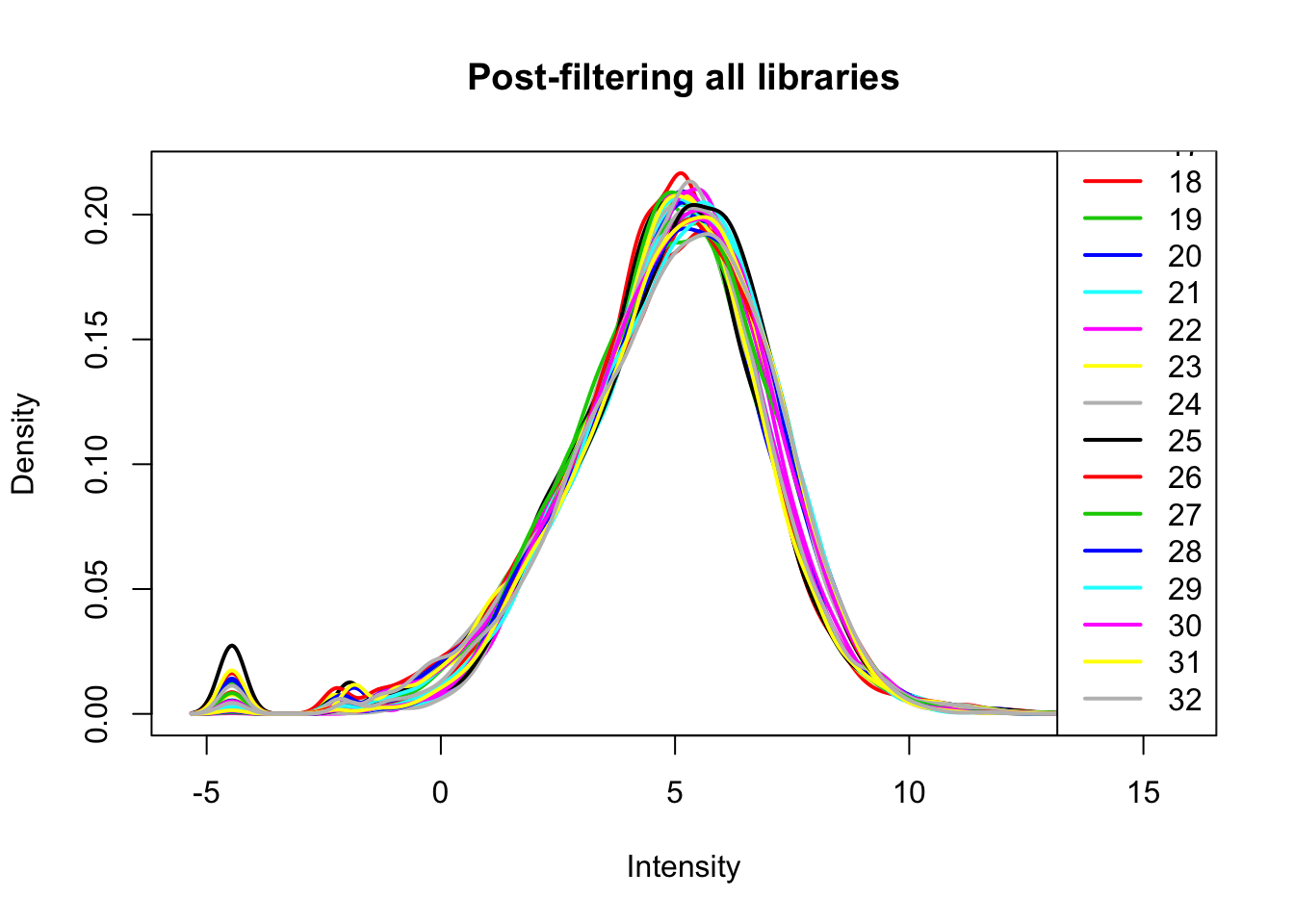
Post filtering we are left with 12461 protein coding genes.
Session information
sessionInfo()R version 3.4.2 (2017-09-28)
Platform: x86_64-apple-darwin15.6.0 (64-bit)
Running under: macOS Sierra 10.12.6
Matrix products: default
BLAS: /Library/Frameworks/R.framework/Versions/3.4/Resources/lib/libRblas.0.dylib
LAPACK: /Library/Frameworks/R.framework/Versions/3.4/Resources/lib/libRlapack.dylib
locale:
[1] en_US.UTF-8/en_US.UTF-8/en_US.UTF-8/C/en_US.UTF-8/en_US.UTF-8
attached base packages:
[1] stats graphics grDevices utils datasets methods base
other attached packages:
[1] bindrcpp_0.2 edgeR_3.20.9 limma_3.34.9 dplyr_0.7.4
[5] ggplot2_2.2.1 workflowr_1.0.1 rmarkdown_1.8.5
loaded via a namespace (and not attached):
[1] Rcpp_0.12.15 compiler_3.4.2 pillar_1.1.0
[4] git2r_0.21.0 plyr_1.8.4 bindr_0.1
[7] R.methodsS3_1.7.1 R.utils_2.6.0 tools_3.4.2
[10] digest_0.6.14 evaluate_0.10.1 tibble_1.4.2
[13] gtable_0.2.0 lattice_0.20-35 pkgconfig_2.0.1
[16] rlang_0.1.6 yaml_2.1.16 stringr_1.2.0
[19] knitr_1.18 locfit_1.5-9.1 rprojroot_1.3-2
[22] grid_3.4.2 glue_1.2.0 R6_2.2.2
[25] magrittr_1.5 whisker_0.3-2 backports_1.1.2
[28] scales_0.5.0 htmltools_0.3.6 assertthat_0.2.0
[31] colorspace_1.3-2 stringi_1.1.6 lazyeval_0.2.1
[34] munsell_0.4.3 R.oo_1.22.0
This reproducible R Markdown analysis was created with workflowr 1.0.1