statistics
lukeholman
2019-06-21
Last updated: 2019-06-28
Checks: 5 1
Knit directory: cost_of_SR_Dpseudo/
This reproducible R Markdown analysis was created with workflowr (version 1.3.0). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
The R Markdown is untracked by Git. To know which version of the R Markdown file created these results, you’ll want to first commit it to the Git repo. If you’re still working on the analysis, you can ignore this warning. When you’re finished, you can run wflow_publish to commit the R Markdown file and build the HTML.
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
The command set.seed(20190319) was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
Nice! There were no cached chunks for this analysis, so you can be confident that you successfully produced the results during this run.
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility. The version displayed above was the version of the Git repository at the time these results were generated.
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can use wflow_publish or wflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Ignored files:
Ignored: .DS_Store
Ignored: .Rhistory
Ignored: .Rproj.user/
Untracked files:
Untracked: analysis/population_genetic_model.Rmd
Untracked: analysis/statistics.Rmd
Untracked: data/SR_fitness_data.csv
Untracked: data/simulation_output.rds
Untracked: data/simulation_output_1.rds
Untracked: data/simulation_output_10.rds
Untracked: data/simulation_output_100.rds
Untracked: data/simulation_output_11.rds
Untracked: data/simulation_output_12.rds
Untracked: data/simulation_output_13.rds
Untracked: data/simulation_output_14.rds
Untracked: data/simulation_output_15.rds
Untracked: data/simulation_output_16.rds
Untracked: data/simulation_output_17.rds
Untracked: data/simulation_output_18.rds
Untracked: data/simulation_output_19.rds
Untracked: data/simulation_output_2.rds
Untracked: data/simulation_output_20.rds
Untracked: data/simulation_output_21.rds
Untracked: data/simulation_output_22.rds
Untracked: data/simulation_output_23.rds
Untracked: data/simulation_output_24.rds
Untracked: data/simulation_output_25.rds
Untracked: data/simulation_output_26.rds
Untracked: data/simulation_output_27.rds
Untracked: data/simulation_output_28.rds
Untracked: data/simulation_output_29.rds
Untracked: data/simulation_output_3.rds
Untracked: data/simulation_output_30.rds
Untracked: data/simulation_output_31.rds
Untracked: data/simulation_output_32.rds
Untracked: data/simulation_output_33.rds
Untracked: data/simulation_output_34.rds
Untracked: data/simulation_output_35.rds
Untracked: data/simulation_output_36.rds
Untracked: data/simulation_output_37.rds
Untracked: data/simulation_output_38.rds
Untracked: data/simulation_output_39.rds
Untracked: data/simulation_output_4.rds
Untracked: data/simulation_output_40.rds
Untracked: data/simulation_output_41.rds
Untracked: data/simulation_output_42.rds
Untracked: data/simulation_output_43.rds
Untracked: data/simulation_output_44.rds
Untracked: data/simulation_output_45.rds
Untracked: data/simulation_output_46.rds
Untracked: data/simulation_output_47.rds
Untracked: data/simulation_output_48.rds
Untracked: data/simulation_output_49.rds
Untracked: data/simulation_output_5.rds
Untracked: data/simulation_output_50.rds
Untracked: data/simulation_output_51.rds
Untracked: data/simulation_output_52.rds
Untracked: data/simulation_output_53.rds
Untracked: data/simulation_output_54.rds
Untracked: data/simulation_output_55.rds
Untracked: data/simulation_output_56.rds
Untracked: data/simulation_output_57.rds
Untracked: data/simulation_output_58.rds
Untracked: data/simulation_output_59.rds
Untracked: data/simulation_output_6.rds
Untracked: data/simulation_output_60.rds
Untracked: data/simulation_output_61.rds
Untracked: data/simulation_output_62.rds
Untracked: data/simulation_output_63.rds
Untracked: data/simulation_output_64.rds
Untracked: data/simulation_output_65.rds
Untracked: data/simulation_output_66.rds
Untracked: data/simulation_output_67.rds
Untracked: data/simulation_output_68.rds
Untracked: data/simulation_output_69.rds
Untracked: data/simulation_output_7.rds
Untracked: data/simulation_output_70.rds
Untracked: data/simulation_output_71.rds
Untracked: data/simulation_output_72.rds
Untracked: data/simulation_output_73.rds
Untracked: data/simulation_output_74.rds
Untracked: data/simulation_output_75.rds
Untracked: data/simulation_output_76.rds
Untracked: data/simulation_output_77.rds
Untracked: data/simulation_output_78.rds
Untracked: data/simulation_output_79.rds
Untracked: data/simulation_output_8.rds
Untracked: data/simulation_output_80.rds
Untracked: data/simulation_output_81.rds
Untracked: data/simulation_output_82.rds
Untracked: data/simulation_output_83.rds
Untracked: data/simulation_output_84.rds
Untracked: data/simulation_output_85.rds
Untracked: data/simulation_output_86.rds
Untracked: data/simulation_output_87.rds
Untracked: data/simulation_output_88.rds
Untracked: data/simulation_output_89.rds
Untracked: data/simulation_output_9.rds
Untracked: data/simulation_output_90.rds
Untracked: data/simulation_output_91.rds
Untracked: data/simulation_output_92.rds
Untracked: data/simulation_output_93.rds
Untracked: data/simulation_output_94.rds
Untracked: data/simulation_output_95.rds
Untracked: data/simulation_output_96.rds
Untracked: data/simulation_output_97.rds
Untracked: data/simulation_output_98.rds
Untracked: data/simulation_output_99.rds
Untracked: docs/figure/
Untracked: figures/
Untracked: output/brms_model.rds
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
There are no past versions. Publish this analysis with wflow_publish() to start tracking its development.
Load R libraries
library(tidyverse)
library(brms)
library(bayestestR)
library(kableExtra)
library(ggbeeswarm)
library(RColorBrewer)
library(showtext)
font_add_google(name = "Lato", family = "Lato", regular.wt = 400, bold.wt = 700)
showtext_auto()
options(stringsAsFactors = FALSE)
SE <- function(x) sd(x) / sqrt(length(x))Load the data
fitness_data <- read_csv("data/SR_fitness_data.csv") %>%
filter(!is.na(genotype)) %>%
rename(body_size = `Body size`,
female_age = `F age`) %>%
mutate(genotype = factor(genotype, levels = c("STST", "SRST", "SRSR")))Warning: Missing column names filled in: 'X12' [12]Parsed with column specification:
cols(
ID = col_integer(),
Isoline = col_character(),
genotype = col_character(),
`genotype pooled 2` = col_integer(),
`genotype pooled 1` = col_integer(),
`genotype pooled` = col_integer(),
Gen = col_integer(),
Block = col_integer(),
`F age` = col_integer(),
`Body size` = col_double(),
offspring = col_integer(),
X12 = col_character()
)Make a table of summary statistics and sample sizes
Here, we calculate the mean offspring produced by females from each of the three genotypes (STST, SRST, and SRSR), either within each isoline or across all the isolines. We also calculate the % females that failed to produce any offspring, and provide sample size information.
means_by_isoline <- fitness_data %>%
group_by(genotype, Isoline) %>%
summarise(
Number_of_females_measured = n(),
Mean_offspring_per_female = mean(offspring),
SE = SE(offspring),
n_females_producing_offspring = sum(offspring != 0),
Percent_females_producing_offspring = 100 * n_females_producing_offspring / n())
means <- fitness_data %>%
mutate(Isoline = "Across all isolines") %>%
group_by(genotype, Isoline) %>%
summarise(
Number_of_females_measured = n(),
Mean_offspring_per_female = mean(offspring),
SE = SE(offspring),
n_females_producing_offspring = sum(offspring != 0),
Percent_females_producing_offspring = 100 * n_females_producing_offspring / n())
bind_rows(means_by_isoline, means) %>%
rename_all(function(x) gsub("_", " ", x)) %>%
rename_all(function(x) gsub("Percent", "%", x)) %>%
rename(Genotype = genotype) %>%
kable(digits = 2) %>% kable_styling()| Genotype | Isoline | Number of females measured | Mean offspring per female | SE | n females producing offspring | % females producing offspring |
|---|---|---|---|---|---|---|
| STST | Lew 13 | 37 | 57.81 | 6.46 | 35 | 94.59 |
| STST | Lew 17 | 40 | 56.85 | 5.04 | 39 | 97.50 |
| STST | Slo B3 | 40 | 76.67 | 5.59 | 39 | 97.50 |
| STST | Slo B7 | 35 | 71.14 | 4.71 | 34 | 97.14 |
| SRST | Lew 13 | 39 | 72.82 | 8.70 | 32 | 82.05 |
| SRST | Lew 17 | 37 | 56.24 | 8.11 | 32 | 86.49 |
| SRST | Slo B3 | 31 | 49.10 | 5.20 | 26 | 83.87 |
| SRST | Slo B7 | 39 | 55.26 | 7.07 | 36 | 92.31 |
| SRSR | Lew 13 | 36 | 28.58 | 5.92 | 25 | 69.44 |
| SRSR | Lew 17 | 37 | 32.19 | 3.91 | 34 | 91.89 |
| SRSR | Slo B3 | 31 | 17.19 | 4.56 | 22 | 70.97 |
| SRSR | Slo B7 | 38 | 25.50 | 4.76 | 28 | 73.68 |
| STST | Across all isolines | 152 | 65.59 | 2.81 | 147 | 96.71 |
| SRST | Across all isolines | 146 | 58.89 | 3.83 | 126 | 86.30 |
| SRSR | Across all isolines | 142 | 26.21 | 2.45 | 109 | 76.76 |
Run the Bayesian hurdle model
The model assumes that the response variable, offspring number, is the result of a ‘hurdle’ process. Essentially this means that the model consists of two sub-models: one controlling the probability that offspring number is non-zero, and one controlling the number of offspring produced provided that more than zero are produced (we assume that offspring number follows a negative binomial distribution, because this improved model fit relative to the simpler hurdle-Poisson model).
We assume that the parameters controlling both the hurdle and the distribution of non-zero values are affected by four fixed effects (the female’s genotype: STST, SRST, or SRSR), her isoline, the female’s age, and the interaction between genotype and isoline. We also fit two random effects: isoline, and experimental block. All fixed effects were assumed to have a prior distribution following a normal distribution with mean 0 and SD = 5.
if(!file.exists("output/brms_model.rds")){
model_formula <- bf(
offspring ~ genotype * Isoline + female_age + (1 | Block), # formula for the mean
hu ~ genotype * Isoline + female_age + (1 | Block) # formula for the hurdle
)
model_prior <- c(set_prior("normal(0, 5)", class = "b"),
set_prior("normal(0, 5)", class = "b", dpar = "hu"))
# imputed_data <- fitness_data %>%
# select(offspring, genotype, body_size, female_age, Isoline, Block) %>%
# mice(m = 5, print = FALSE)
model <- brm(model_formula,
family = "hurdle_negbinomial",
chains = 4, cores = 1, iter = 4000, inits = 0, seed = 12345,
control = list(adapt_delta = 0.999, max_treedepth = 15),
prior = model_prior,
data = fitness_data)
saveRDS(model, file = "output/brms_model.rds")
} else model <- readRDS("output/brms_model.rds")Perform a posterior predictive check
The idea behind posterior predictive checking is that if our model is a good fit, then we should be able to use it to generate data that looks a lot like the data we observed. Here, we see 10 draws from the ‘posterior predictive distribution’ (thin lines), which indeed look quite similar to the distribution of the real data (thick line), suggested that our model approximates the processes that generated the real data well enough for reliable inference.
pp_check(model, type = "dens_overlay")Using 10 posterior samples for ppc type 'dens_overlay' by default.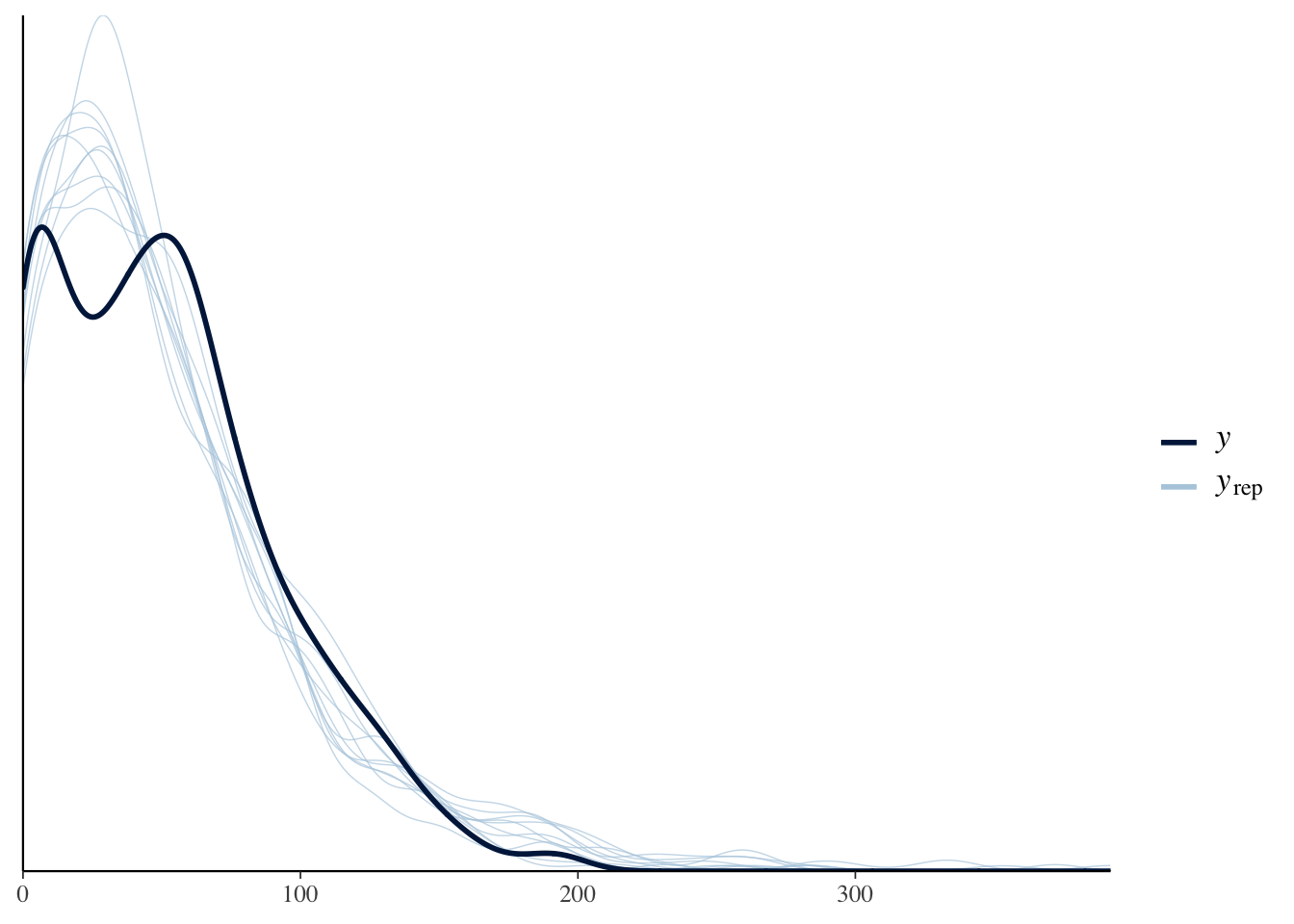
Inspect the model’s parameter estimates
bayesian_p_values <- as.data.frame(p_direction(model)) %>%
mutate(pd = (100 - pd) / 100,
Parameter = gsub("[.]", ":", gsub("b_", "", Parameter)))
random <- summary(model)$random # Finish
summary(model)$fixed %>% as.data.frame() %>%
rownames_to_column("Parameter") %>%
left_join(bayesian_p_values, by = "Parameter") %>%
rename(p = pd) %>% arrange(grepl("hu_", Parameter)) %>%
mutate(Parameter = gsub("hu_", "Hurdle - ", Parameter),
Estimate = format(round(Estimate, 3), nsmall = 3),
Est.Error = format(round(Est.Error, 3), nsmall = 3),
` ` = ifelse(p < 0.05, "*", ""),
p = format(round(p, 4), nsmall = 4),
Rhat = format(round(Rhat, 3), nsmall = 3),
`l-95% CI` = format(round(`l-95% CI`, 3), nsmall = 3),
`u-95% CI` = format(round(`u-95% CI`, 3), nsmall = 3),
Eff.Sample = round(Eff.Sample, 0)
) Parameter Estimate Est.Error l-95% CI u-95% CI
1 Intercept 4.049 0.269 3.520 4.585
2 genotypeSRST 0.366 0.169 0.029 0.698
3 genotypeSRSR -0.423 0.186 -0.790 -0.065
4 IsolineLew17 -0.061 0.164 -0.378 0.261
5 IsolineSloB3 0.287 0.167 -0.048 0.612
6 IsolineSloB7 0.163 0.171 -0.170 0.496
7 female_age 0.023 0.058 -0.091 0.138
8 genotypeSRST:IsolineLew17 -0.225 0.242 -0.690 0.261
9 genotypeSRSR:IsolineLew17 -0.017 0.255 -0.513 0.488
10 genotypeSRST:IsolineSloB3 -0.687 0.253 -1.187 -0.191
11 genotypeSRSR:IsolineSloB3 -0.849 0.268 -1.361 -0.314
12 genotypeSRST:IsolineSloB7 -0.547 0.240 -1.015 -0.069
13 genotypeSRSR:IsolineSloB7 -0.340 0.263 -0.854 0.176
14 Hurdle - Intercept -1.498 1.190 -3.834 0.816
15 Hurdle - genotypeSRST 1.473 0.799 0.003 3.142
16 Hurdle - genotypeSRSR 2.186 0.789 0.750 3.854
17 Hurdle - IsolineLew17 -1.045 1.206 -3.607 1.140
18 Hurdle - IsolineSloB3 -0.925 1.195 -3.408 1.287
19 Hurdle - IsolineSloB7 -0.666 1.197 -3.150 1.504
20 Hurdle - female_age -0.327 0.237 -0.796 0.131
21 Hurdle - genotypeSRST:IsolineLew17 0.564 1.344 -1.982 3.384
22 Hurdle - genotypeSRSR:IsolineLew17 -0.833 1.399 -3.483 2.078
23 Hurdle - genotypeSRST:IsolineSloB3 0.851 1.343 -1.656 3.658
24 Hurdle - genotypeSRSR:IsolineSloB3 1.078 1.307 -1.358 3.753
25 Hurdle - genotypeSRST:IsolineSloB7 -0.590 1.402 -3.270 2.293
26 Hurdle - genotypeSRSR:IsolineSloB7 0.186 1.291 -2.219 2.827
Eff.Sample Rhat p
1 6153 1.000 0.0000 *
2 3221 1.001 0.0166 *
3 3580 1.001 0.0111 *
4 4053 1.000 0.3622
5 3999 1.000 0.0424 *
6 3623 1.000 0.1717
7 9705 1.000 0.3435
8 3965 1.000 0.1704
9 4135 1.001 0.4712
10 4235 1.000 0.0045 *
11 4732 1.001 0.0009 *
12 3817 1.000 0.0115 *
13 4039 1.001 0.0988
14 5225 1.000 0.1005
15 3454 1.000 0.0250 *
16 3472 1.000 0.0009 *
17 3525 1.000 0.1933
18 2814 1.000 0.2150
19 3975 1.000 0.3049
20 10966 1.000 0.0831
21 3611 1.000 0.3439
22 3812 1.000 0.2704
23 3169 1.000 0.2689
24 2985 1.000 0.2051
25 4095 1.000 0.3333
26 3847 1.000 0.4508 Plot the estimated means for each genotype
new <- fitness_data %>% select(genotype, Isoline, body_size, female_age) %>%
mutate(body_size = mean(body_size, na.rm = TRUE),
female_age = mean(female_age)) %>%
distinct()
predicted_mean <- data.frame(new, fitted(model, newdata = new, re_formula = NA)) %>%
mutate(facet = "A. Mean offspring production")
predicted_mean_when_fertile <- data.frame(new, fitted(model, newdata = new, dpar = "mu", re_formula = NA)) %>%
mutate(facet = "B. Mean offspring production\n(excluding infertile females)")
predicted_prop_infertile <- data.frame(new, fitted(model, newdata = new, dpar = "hu", re_formula = NA)) %>%
mutate(facet = "C. % fertile females",
Estimate = 100 * (1 - Estimate), Q2.5 = (1 - Q2.5) * 100, Q97.5 = (1 - Q97.5) * 100)
posterior_means <-
data.frame(new, as.data.frame(t(fitted(model, newdata = new, re_formula = NA, summary = FALSE)))) %>%
select(-body_size, -female_age, -Isoline) %>%
group_by(genotype) %>%
summarise_all(mean) %>% select(-genotype) %>% t() %>% as.data.frame() %>%
rename(STST = V1, SRST = V2, SRSR = V3)
predicted_mean_all_iso <- data.frame(
genotype = c("STST", "SRST", "SRSR"), Isoline = "All",
posterior_means %>% lapply(posterior_summary) %>% do.call("rbind", .),
facet = "A. Mean offspring production")
posterior_means_when_fertile <-
data.frame(new, as.data.frame(t(fitted(model, newdata = new, dpar = "mu", re_formula = NA, summary = FALSE)))) %>%
select(-body_size, -female_age, -Isoline) %>%
group_by(genotype) %>%
summarise_all(mean) %>% select(-genotype) %>% t() %>% as.data.frame() %>%
rename(STST = V1, SRST = V2, SRSR = V3)
predicted_mean_when_fertile_all_iso <- data.frame(
genotype = c("STST", "SRST", "SRSR"), Isoline = "All",
posterior_means_when_fertile %>% lapply(posterior_summary) %>% do.call("rbind", .),
facet = "B. Mean offspring production\n(excluding infertile females)")
posterior_means_prop_infertile <-
data.frame(new, as.data.frame(t(fitted(model, newdata = new, dpar = "hu", re_formula = NA, summary = FALSE)))) %>%
select(-body_size, -female_age, -Isoline) %>%
group_by(genotype) %>%
summarise_all(~ 100 * mean(.x)) %>% select(-genotype) %>% t() %>% as.data.frame() %>%
rename(STST = V1, SRST = V2, SRSR = V3)
predicted_prop_infertile_all_iso <- data.frame(
genotype = c("STST", "SRST", "SRSR"), Isoline = "All",
posterior_means_prop_infertile %>% lapply(posterior_summary) %>% do.call("rbind", .),
facet = "C. % fertile females")
preds_figure1 <- bind_rows(predicted_mean_all_iso,
predicted_mean_when_fertile_all_iso,
predicted_prop_infertile_all_iso) %>%
mutate(genotype = factor(genotype, levels = c("STST", "SRST", "SRSR")))
preds_figure2 <- bind_rows(predicted_mean,
predicted_mean_when_fertile,
predicted_prop_infertile) %>%
mutate(facet = factor(facet, levels = unique(facet)),
genotype = factor(genotype, levels = c("STST", "SRST", "SRSR"))) %>%
select(-body_size, -female_age)
beeswarm_points <- bind_rows(
fitness_data %>% mutate(facet = "A. Mean offspring production"),
fitness_data %>% filter(offspring != 0) %>% mutate(facet = "B. Mean offspring production\n(excluding infertile females)")) %>%
mutate(Fertility = ifelse(offspring == 0, "Sterile", "Fertile"),
genotype = factor(genotype, levels = c("STST", "SRST", "SRSR"))) %>%
rename(Estimate = offspring)
pos1 <- position_nudge(x = -0.17)
pal <- c(brewer.pal(4, "RdPu")[2], brewer.pal(7, "Purples")[5])
figure_1 <- preds_figure1 %>%
ggplot(aes(genotype, Estimate)) +
geom_errorbar(aes(ymin = Q2.5, ymax = Q97.5), colour = "grey20", position = pos1, size = .8, width = 0.1) +
geom_beeswarm(data = beeswarm_points, aes(colour = Fertility),
size = .7, alpha = 0.6) +
geom_point(size = 3.1, pch = 21, colour = "black", position = pos1, fill = "grey20") +
scale_colour_manual(values = pal) +
facet_wrap(~facet, scale = "free_y") +
labs(y = "Posterior estimate \u00B1 95% CIs", x = "Genotype") +
theme_bw() +
theme(strip.background = element_blank(),
text = element_text(family = "Lato", size = 12),
panel.grid.major.x = element_blank(),
strip.text = element_text(hjust = 0))
dodge <- position_dodge(0.66)
figure_2 <- preds_figure2 %>%
ggplot(aes(genotype, Estimate, fill = Isoline)) +
geom_errorbar(aes(ymin = Q2.5, ymax = Q97.5), size = .7, width = 0.3, colour = "grey40", position = dodge) +
geom_point(size = 3.1, pch = 21, colour = "black", position = dodge) +
facet_wrap(~facet, scale = "free_y") +
scale_fill_brewer(palette = "Pastel1") +
labs(y = "Posterior estimate \u00B1 95% CIs", x = "Genotype") +
theme_bw() +
theme(strip.background = element_blank(),
text = element_text(family = "Lato", size = 12),
panel.grid.major.x = element_blank(),
strip.text = element_text(hjust = 0))
figure_1 %>% ggsave(filename = "figures/figure_1.pdf", width = 9, height = 4)
figure_2 %>% ggsave(filename = "figures/figure_2.pdf", width = 9, height = 4)figure_1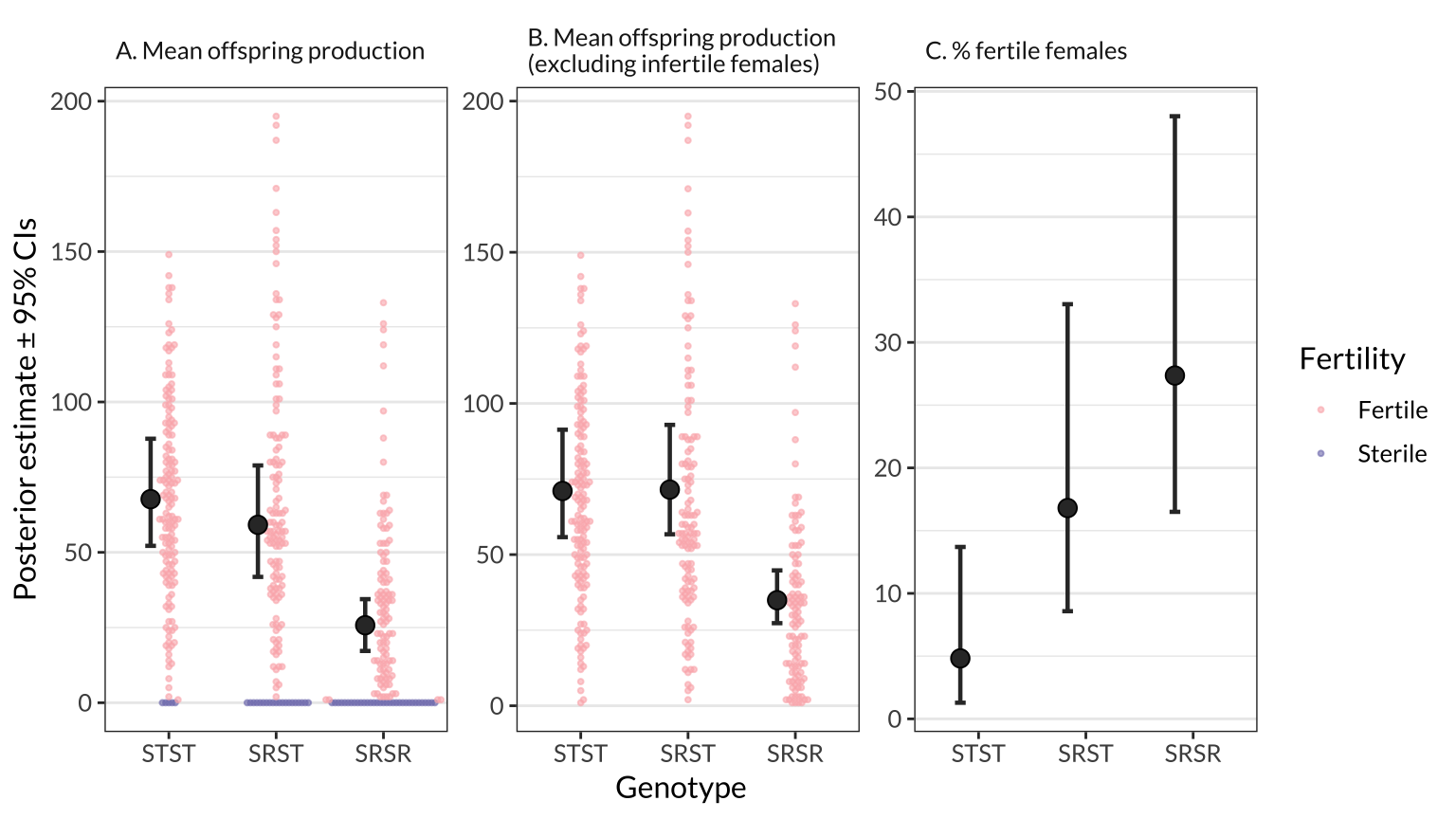
figure_2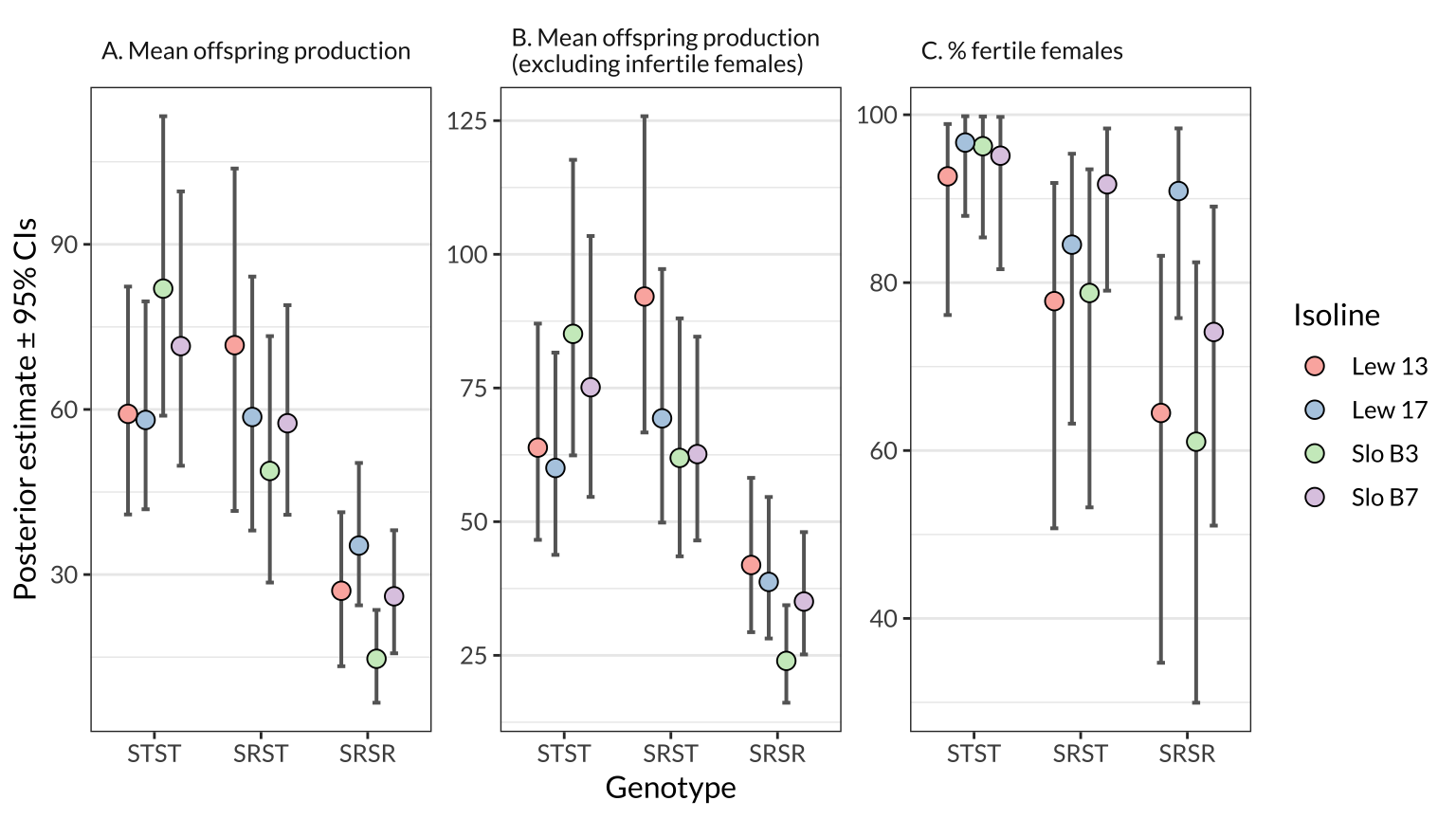
Calculated pairwise differences between genotypes
compare_means <- function(mean1, mean2, posterior){
difference <- posterior[, mean2] - posterior[, mean1]
relative <- median(posterior[, mean2] / posterior[, mean1])
p_value <- as.numeric(p_direction(difference)) / 100
as_tibble(posterior_summary(as.mcmc(difference))) %>%
mutate(Comparison = paste(mean1, mean2, sep = " \u2192 "),
`% difference` = format(round(100 * relative, 1), nsmall = 1),
`95% CIs` = paste(" (", format(round(Q2.5, 1), nsmall = 1), " to ", format(round(Q97.5, 1), nsmall = 1), "%)", sep = ""),
`Difference in means` = paste(format(round(Estimate, 2), nsmall = 2), `95% CIs`, sep = ""),
`Fitness trait` = NA,
p = p_value) %>%
select( -Q2.5, -Q97.5) %>%
select(Comparison, `Fitness trait`, `Difference in means`, Est.Error, `% difference`, p)
}
table_of_contrasts <- bind_rows(
compare_means("STST", "SRST", posterior_means),
compare_means("STST", "SRSR", posterior_means),
compare_means("SRST", "SRSR", posterior_means),
compare_means("STST", "SRST", posterior_means_when_fertile),
compare_means("STST", "SRSR", posterior_means_when_fertile),
compare_means("SRST", "SRSR", posterior_means_when_fertile),
compare_means("STST", "SRST", posterior_means_prop_infertile),
compare_means("STST", "SRSR", posterior_means_prop_infertile),
compare_means("SRST", "SRSR", posterior_means_prop_infertile)
) %>% mutate(`Fitness trait` = rep(c("Mean offspring production",
"Mean offspring production (excluding infertile females)",
"% fertile females"), each = 3)) %>%
mutate(Est.Error = format(round(Est.Error, 2), nsmall = 2),
` ` = ifelse(p < 0.05, "*", " "),
p = format(round(p, 4), nsmall = 4))
table_of_contrasts %>%
kable() %>% kable_styling()| Comparison | Fitness trait | Difference in means | Est.Error | % difference | p | |
|---|---|---|---|---|---|---|
| STST → SRST | Mean offspring production | -8.53 (-23.1 to 4.7%) | 6.98 | 87.7 | 0.8986 | |
| STST → SRSR | Mean offspring production | -41.92 (-57.2 to -29.5%) | 7.27 | 38.3 | 1.0000 | |
| SRST → SRSR | Mean offspring production | -33.39 (-47.6 to -21.5%) | 6.78 | 43.6 | 1.0000 | |
| STST → SRST | Mean offspring production (excluding infertile females) | 0.46 (-12.5 to 13.5%) | 6.55 | 100.6 | 0.5270 | |
| STST → SRSR | Mean offspring production (excluding infertile females) | -36.12 (-50.5 to -24.5%) | 6.85 | 49.1 | 1.0000 | |
| SRST → SRSR | Mean offspring production (excluding infertile females) | -36.58 (-51.4 to -24.9%) | 6.90 | 48.8 | 1.0000 | |
| STST → SRST | % fertile females | 11.98 (4.0 to 23.6%) | 5.04 | 383.7 | 0.9985 | |
| STST → SRSR | % fertile females | 22.54 (12.3 to 37.8%) | 6.29 | 637.5 | 1.0000 | |
| SRST → SRSR | % fertile females | 10.56 (0.4 to 21.8%) | 5.40 | 165.6 | 0.9796 |
sessionInfo()R version 3.5.1 (2018-07-02)
Platform: x86_64-apple-darwin15.6.0 (64-bit)
Running under: macOS High Sierra 10.13.6
Matrix products: default
BLAS: /Library/Frameworks/R.framework/Versions/3.5/Resources/lib/libRblas.0.dylib
LAPACK: /Library/Frameworks/R.framework/Versions/3.5/Resources/lib/libRlapack.dylib
locale:
[1] en_AU.UTF-8/en_AU.UTF-8/en_AU.UTF-8/C/en_AU.UTF-8/en_AU.UTF-8
attached base packages:
[1] stats graphics grDevices utils datasets methods base
other attached packages:
[1] showtext_0.5-1 showtextdb_2.0 sysfonts_0.7.2
[4] RColorBrewer_1.1-2 ggbeeswarm_0.6.0 kableExtra_0.9.0
[7] bayestestR_0.2.2 brms_2.8.0 Rcpp_1.0.1
[10] forcats_0.4.0 stringr_1.4.0 dplyr_0.8.0.1
[13] purrr_0.3.2 readr_1.1.1 tidyr_0.8.2
[16] tibble_2.0.99.9000 ggplot2_3.1.0 tidyverse_1.2.1
loaded via a namespace (and not attached):
[1] colorspace_1.3-2 ggridges_0.5.0 rsconnect_0.8.8
[4] rprojroot_1.3-2 markdown_0.9 base64enc_0.1-3
[7] fs_1.3.1 rstudioapi_0.10 rstan_2.18.2
[10] DT_0.4 mvtnorm_1.0-8 lubridate_1.7.4
[13] xml2_1.2.0 bridgesampling_0.4-0 knitr_1.22
[16] shinythemes_1.1.1 bayesplot_1.6.0 jsonlite_1.6
[19] workflowr_1.3.0 broom_0.5.0 shiny_1.3.2
[22] compiler_3.5.1 httr_1.4.0 backports_1.1.2
[25] assertthat_0.2.1 Matrix_1.2-14 lazyeval_0.2.2
[28] cli_1.1.0 later_0.8.0 htmltools_0.3.6
[31] prettyunits_1.0.2 tools_3.5.1 igraph_1.2.1
[34] coda_0.19-2 gtable_0.2.0 glue_1.3.1.9000
[37] reshape2_1.4.3 cellranger_1.1.0 nlme_3.1-137
[40] crosstalk_1.0.0 insight_0.3.0 xfun_0.6
[43] ps_1.3.0 rvest_0.3.2 mime_0.6
[46] miniUI_0.1.1.1 gtools_3.8.1 zoo_1.8-3
[49] scales_1.0.0 colourpicker_1.0 hms_0.4.2
[52] promises_1.0.1 Brobdingnag_1.2-5 parallel_3.5.1
[55] inline_0.3.15 shinystan_2.5.0 curl_3.3
[58] yaml_2.2.0 gridExtra_2.3 loo_2.1.0
[61] StanHeaders_2.18.0 stringi_1.4.3 highr_0.8
[64] dygraphs_1.1.1.6 pkgbuild_1.0.2 rlang_0.3.4
[67] pkgconfig_2.0.2 matrixStats_0.54.0 evaluate_0.13
[70] lattice_0.20-35 labeling_0.3 rstantools_1.5.0
[73] htmlwidgets_1.2 tidyselect_0.2.5 processx_3.2.1
[76] plyr_1.8.4 magrittr_1.5 R6_2.4.0
[79] pillar_1.3.1.9000 haven_1.1.2 withr_2.1.2
[82] xts_0.11-0 abind_1.4-5 modelr_0.1.2
[85] crayon_1.3.4 rmarkdown_1.10 grid_3.5.1
[88] readxl_1.1.0 callr_2.0.4 git2r_0.23.0
[91] threejs_0.3.1 digest_0.6.18 xtable_1.8-4
[94] httpuv_1.5.1 stats4_3.5.1 munsell_0.5.0
[97] beeswarm_0.2.3 viridisLite_0.3.0 vipor_0.4.5
[100] shinyjs_1.0