Case Study - Main Results
Pedro L. Baldoni
27 August, 2025
Last updated: 2025-08-27
Checks: 7 0
Knit directory: DTU-code/analysis/
This reproducible R Markdown analysis was created with workflowr (version 1.7.1). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
Great! Since the R Markdown file has been committed to the Git repository, you know the exact version of the code that produced these results.
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
The command set.seed(20240501) was run prior to running
the code in the R Markdown file. Setting a seed ensures that any results
that rely on randomness, e.g. subsampling or permutations, are
reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
Nice! There were no cached chunks for this analysis, so you can be confident that you successfully produced the results during this run.
Great job! Using relative paths to the files within your workflowr project makes it easier to run your code on other machines.
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility.
The results in this page were generated with repository version 5951235. See the Past versions tab to see a history of the changes made to the R Markdown and HTML files.
Note that you need to be careful to ensure that all relevant files for
the analysis have been committed to Git prior to generating the results
(you can use wflow_publish or
wflow_git_commit). workflowr only checks the R Markdown
file, but you know if there are other scripts or data files that it
depends on. Below is the status of the Git repository when the results
were generated:
Ignored files:
Ignored: .Rhistory
Ignored: .Rproj.user/
Ignored: .gitignore
Ignored: .renvignore
Ignored: DTU-code.Rproj
Ignored: code/.RData
Ignored: code/mouse/data/slurm-16691239.out
Ignored: code/mouse/salmon-index/slurm-16693056.out
Ignored: code/mouse/salmon/slurm-16693640.out
Ignored: code/mouse/subread-index/.RData
Ignored: code/mouse/subread-index/buildindex.Rout
Ignored: code/mouse/subread-index/slurm-19350188.out
Ignored: code/mouse/subread/.nextflow.log
Ignored: code/mouse/subread/.nextflow/
Ignored: code/mouse/subread/log/
Ignored: code/mouse/subread/report.html
Ignored: code/mouse/subread/slurm-19356012.out
Ignored: code/mouse/subread/timeline.html
Ignored: code/mouse/subread/trace-20241127-50139746.txt
Ignored: code/pkg/.Rbuildignore
Ignored: code/pkg/.Rhistory
Ignored: code/pkg/.Rproj.user/
Ignored: code/pkg/src/.gitignore
Ignored: code/pkg/src/RcppExports.o
Ignored: code/pkg/src/pkg.so
Ignored: code/pkg/src/rcpparma_hello_world.o
Ignored: code/simulation-no-trend/
Ignored: data/annotation/mm39/GRCm39.genome.fa.gz
Ignored: data/annotation/mm39/gencode.vM35.annotation.gtf.gz
Ignored: data/annotation/mm39/gencode.vM35.metadata.EntrezGene.gz
Ignored: data/annotation/mm39/gencode.vM35.transcripts.fa.gz
Ignored: data/mouse/fastq/
Ignored: ignore/
Ignored: output/mouse/
Ignored: output/simulation-lenient/
Ignored: output/simulation-no-trend/
Ignored: renv/
Untracked files:
Untracked: code/simulation-lenient-no-trend/
Untracked: misc/main-casestudy.Rmd/
Untracked: misc/main-simulation.Rmd/
Untracked: misc/supp-bcv.Rmd/
Untracked: misc/supp-simulation-dividedcounts.Rmd/
Untracked: misc/supp-simulation-lenient.Rmd/
Untracked: misc/supp-simulation-rawcounts.Rmd/
Untracked: output/old-simulation-lenient/
Untracked: output/old-simulation/
Untracked: output/simulation-lenient-no-trend/
Untracked: output/simulation/
Unstaged changes:
Deleted: analysis/about.Rmd
Deleted: analysis/mouse.Rmd
Deleted: analysis/simulation-lenient-supp.Rmd
Deleted: analysis/simulation-paper.Rmd
Deleted: analysis/simulation-supp.Rmd
Modified: code/pkg/R/simulation-summary.R
Modified: code/pkg/R/simulation.R
Deleted: code/readme.txt
Deleted: code/simulation-lenient/main.R
Deleted: code/simulation-lenient/parameters.R
Deleted: code/simulation-lenient/parameters.txt
Deleted: code/simulation-lenient/readme.txt
Deleted: code/simulation-lenient/run.sh
Deleted: code/simulation-lenient/summarize.R
Deleted: code/simulation/main.R
Deleted: code/simulation/parameters.R
Deleted: code/simulation/parameters.txt
Deleted: code/simulation/readme.txt
Deleted: code/simulation/run.sh
Deleted: code/simulation/summarize.R
Deleted: misc/mouse.Rmd/Figure-CaseStudy.pdf
Deleted: misc/simulation-lenient-supp.Rmd/SuppTable-Filtering-Gene.tex
Deleted: misc/simulation-lenient-supp.Rmd/SuppTable-Filtering-Transcript.tex
Deleted: misc/simulation-paper.Rmd/Figure-FDR.pdf
Deleted: misc/simulation-paper.Rmd/Figure-Power.pdf
Deleted: misc/simulation-paper.Rmd/Figure-Pval.pdf
Deleted: misc/simulation-paper.Rmd/Figure-Time.pdf
Deleted: misc/simulation-supp.Rmd/SuppFigure-ErrorRate.pdf
Deleted: misc/simulation-supp.Rmd/SuppFigure-FDR.pdf
Deleted: misc/simulation-supp.Rmd/SuppFigure-FP-SecondaryTranscripts-Unbalanced.pdf
Deleted: misc/simulation-supp.Rmd/SuppFigure-Power.pdf
Deleted: misc/simulation-supp.Rmd/SuppFigure-Pval-Gene-Unbalanced.pdf
Deleted: misc/simulation-supp.Rmd/SuppFigure-Pval-Transcript-Unbalanced.pdf
Deleted: misc/simulation-supp.Rmd/SuppFigure-ROC.pdf
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
These are the previous versions of the repository in which changes were
made to the R Markdown (analysis/main-casestudy.Rmd) and
HTML (docs/main-casestudy.html) files. If you’ve configured
a remote Git repository (see ?wflow_git_remote), click on
the hyperlinks in the table below to view the files as they were in that
past version.
| File | Version | Author | Date | Message |
|---|---|---|---|---|
| Rmd | 5951235 | Pedro Baldoni | 2025-08-27 | DTU paper revision |
Introduction
Setup
knitr::opts_chunk$set(dev = "png",
dpi = 300,
dev.args = list(type = "cairo-png"),
root.dir = '.',
autodep = TRUE)
options(knitr.kable.NA = "-")library(edgeR)
library(rtracklayer)
library(Rsubread)
library(GenomicRanges)
library(GenomicFeatures)
library(data.table)
library(Gviz)
library(TxDb.Mmusculus.UCSC.mm39.refGene)
library(csaw)
library(BiocParallel)
library(ggplot2)
library(stringr)
library(png)
library(patchwork)path.misc <- '../misc/main-casestudy.Rmd'
path.quant <- list.dirs('../output/mouse/salmon',recursive = FALSE)
path.target <- '../data/mouse/misc/targets.txt'
path.entrez <- "../data/annotation/mm39/gencode.vM35.metadata.EntrezGene.gz"
path.fasta <- '../data/annotation/mm39/gencode.vM35.transcripts.fa.gz'
path.gtf <- '../data/annotation/mm39/gencode.vM35.annotation.gtf.gz'
path.bam <- "../output/mouse/subread/alignment"
path.bw <- "../output/mouse/subread/coverage/"
dir.create(path.misc,recursive = TRUE,showWarnings = FALSE)bs <- 8plotSplice2 <- function(fit, coef=ncol(fit), geneid=NULL, genecolname=NULL, rank=1L, FDR=0.05,cex.axis = 1,exonlabel = NULL,fontsize = 8,...)
# This function has been adapted from limma::plotSplice so that axis labels
# uses "transcript" and not "exon"
{
if(is.null(genecolname))
genecolname <- fit$genecolname
else
genecolname <- as.character(genecolname)
if(is.null(geneid)) {
# Find gene from specified rank
if(rank==1L)
i <- which.min(fit$gene.F.p.value[,coef])
else
i <- order(fit$gene.F.p.value[,coef])[rank]
geneid <- paste(fit$gene.genes[i,genecolname], collapse=".")
} else {
# Find gene from specified name
geneid <- as.character(geneid)
i <- which(fit$gene.genes[,genecolname]==geneid)[1]
if(!length(i)) stop(paste("geneid",geneid,"not found"))
}
# Row numbers containing exons
j <- fit$gene.firstexon[i]:fit$gene.lastexon[i]
exoncolname <- fit$exoncolname
if(!is.null(exonlabel)) exoncolname <- exonlabel
# Get strand if possible
strcol <- grepl("strand", colnames(fit$gene.genes), ignore.case=TRUE)
if(any(strcol)) geneid <- paste0(geneid, " (", as.character(fit$gene.genes[i, strcol])[1], ")")
if(is.null(exoncolname)) {
plot(fit$coefficients[j, coef], xlab="Exon", ylab="logFC (this transcript vs rest)", main=geneid, type="b")
} else {
exon.id <- fit$genes[j, exoncolname]
# xlab <- paste("Exon", exoncolname, sep=" ")
plot(fit$coefficients[j, coef], xlab="", ylab="logFC (this transcript vs rest)", main=geneid, type="b", xaxt="n",cex.lab = fontsize/12,cex.main = fontsize/12,cex.axis=fontsize/12,...)
axis(1, at=1:length(j), labels=exon.id, las=2, cex.axis=fontsize/12)
# mtext(xlab, side=1, padj=5.2)
}
# Mark the topSpliced exons
top <- topSplice(fit, coef=coef, number=Inf, test="t", sort.by="none")
m <- which(top[, genecolname] %in% fit$gene.genes[i, genecolname])
fdr <- top$FDR[m]
sig <- fdr < FDR
if(any(sig)){
fdr.sig <- fdr[sig]
if(length(unique(fdr.sig))==1)
cex <- 1.5
else {
abs.fdr <- abs(log10(fdr.sig))
from <- range(abs.fdr)
to <- c(1,2)
cex <- (abs.fdr - from[1])/diff(from) * diff(to) + to[1]
}
points((1:length(j))[sig], fit$coefficients[j[sig], coef], col="red", pch=16, cex=cex)
}
abline(h=0,lty=2)
invisible()
}
plot.barplot <- function(x,top = 10,fontsize = 8){
txt <- x$Term[1:top]
txt <- str_to_sentence(txt)
pval <- x$P.DE[1:top]
df <- data.frame(Term = txt,Value = -log10(pval))
df$Term <- factor(df$Term,levels = txt[order(pval,decreasing = TRUE)])
ggplot(data = df,aes(y = Term,x = Value)) +
geom_col(fill = 'gray',col = 'black') +
theme_bw(base_size = fontsize,base_family = 'sans') +
theme(panel.grid = element_blank(),
axis.text = element_text(colour = 'black',size = fontsize),
axis.title = element_text(colour = 'black',size = fontsize)) +
labs(x = "-log10(P-value)",y = NULL)
}
getGeneRanges <- function(entrezid,type = 'gene',flank = TRUE){
out <-
genes(TxDb.Mmusculus.UCSC.mm39.refGene, filter = list(gene_id = entrezid))
if (type == 'promoter') {
out <- promoters(out)
}
if(flank == TRUE & type == 'gene'){
flank <- ceiling(0.25*width(out))
out <- GRanges(seqnames = as.character(seqnames(out)),IRanges(start = start(out)-flank,end = end(out) + flank))
}
return(out)
}
getCoverage <- function(bam, lib.sizes, gr, param, labels = NULL, group.by = NULL, ...) {
reads <- bplapply(seq_along(bam),function(i){
reads <- extractReads(bam.file = bam[i], gr, param = param)
},...)
if (!is.null(group.by)) {
groups <- unique(group.by)
reads <- bplapply(seq_along(groups),function(i){
subreads <- reads[group.by == groups[i]]
do.call('c',subreads)
},...)
names(reads) <- groups
lib.sizes <- bplapply(seq_along(groups),function(i){
sum(lib.sizes[group.by == groups[i]])
},...)
lib.sizes <- unlist(lib.sizes)
} else{
if (is.null(labels)) {
names(reads) <- bam
} else{
names(reads) <- labels
}
}
cov <- bplapply(seq_along(reads),function(i){
as(coverage(reads[[i]])/(lib.sizes[i]/1e6), "GRanges")
},...)
names(cov) <- names(reads)
return(cov)
}
getTrack <- function(cov, fill = NULL, ylim = NULL,yTicksAt = NULL,isBAM = NULL, ...) {
if (is.null(fill)) {
fill <- rep('gray',length(cov))
}
if (is.null(ylim) & isBAM) {
max.y <- max(unlist(lapply(cov,function(i){max(i$score)})))
ylim <- c(0,round(max.y + 5, -1))
}
out <- bplapply(seq_along(cov),function(i){
DataTrack(cov[[i]],
type = "histogram",
baseline = 0,
col.baseline = 'black',
lwd.baseline = 0.25,
ylim = ylim,
yTicksAt = yTicksAt,
name = names(cov[i]),
fill = fill[i],
col.title = 'black',
background.title = "white",
col.axis = 'black',
col.histogram = NA)
},...)
return(out)
}
plotCoverage <- function(x,lib.sizes,param,anno,fontsize = 8,isBAM = TRUE,
gr = NULL,entrezid = NULL,fill = NULL, labels = NULL, group.by = NULL, flank = TRUE, main = NULL,ylim = NULL, yTicksAt = NULL, ...){
if(!is.null(entrezid)){
gr <- getGeneRanges(entrezid,flank = TRUE)
}
if(isBAM){
cov <- getCoverage(bam = x,lib.sizes = lib.sizes,gr = gr,param = param,group.by = group.by,labels = labels,...)
} else{
cov <- x
names(cov) <- labels
}
if (!is.null(fill) & !is.null(group.by)) {
groups <- unique(group.by)
fill <- lapply(seq_along(groups),function(i){
subfill <- unique(fill[group.by == groups[i]])
if(length(subfill) == 1){
return(subfill)
} else{
stop('fill argument must be unique within groups')
}
})
fill <- unlist(fill)
}
track <- getTrack(cov = cov,fill = fill,ylim = ylim,yTicksAt = yTicksAt,isBAM = isBAM)
itrack <- IdeogramTrack(genome = 'mm39',
chromosome = as.character(seqnames(gr)),
fontcolor = 'black',
cex = fontsize/12)
axisTrack <- GenomeAxisTrack(cex = fontsize/12)
plotTracks(c(itrack,track, anno),
chromosome = as.character(seqnames(gr)),
cex.axis = fontsize/12,
cex.title = fontsize/12,
cex.group = fontsize/12,
sizes = c(0.1,rep(0.4,9), 0.75),
from = start(gr),
to = end(gr))
}
plot.voom <- function(fit,fontsize = 8,...){
plot(x = fit$voom.xy$x,
y = fit$voom.xy$y,
xlab = fit$voom.xy$xlab,
ylab = fit$voom.xy$ylab,
pch = fit$voom.xy$pch,
cex = fit$voom.xy$cex,
cex.lab = fontsize/12,
cex.axis = fontsize/12,
cex.main = fontsize/12,...)
lines(fit$voom.line,col="red",lty=1)
}
# plotBCV returns invisible(), we need to manually export the plot (code from edgeR::plotBCV)
foo.bcv <- function (y,
xlab = "Average log CPM",
ylab = "Biological coefficient of variation",
pch = 16, cex = 0.2,
col.common = "red",
col.trend = "blue",
col.tagwise = "black",
fontsize = 8, ...) {
if (!is(y, "DGEList"))
stop("y must be a DGEList.")
A <- y$AveLogCPM
if (is.null(A))
A <- aveLogCPM(y$counts, offset = getOffset(y))
disp <- getDispersion(y)
if (is.null(disp))
stop("No dispersions to plot")
if (attr(disp, "type") == "common")
disp <- rep_len(disp, length(A))
par(mar = c(3, 3, 0.25, 0.25),mgp = c(1.25,0.5,0))
plot(A, sqrt(disp), xlab = xlab, ylab = ylab, type = "n",
cex.lab = fontsize/12,
cex.axis = fontsize/12,
...)
labels <- cols <- lty <- pt <- NULL
if (!is.null(y$tagwise.dispersion)) {
points(A, sqrt(y$tagwise.dispersion), pch = pch, cex = cex,
col = col.tagwise)
labels <- c(labels, "Tagwise")
cols <- c(cols, col.tagwise)
lty <- c(lty, -1)
pt <- c(pt, pch)
}
if (!is.null(y$common.dispersion)) {
abline(h = sqrt(y$common.dispersion), col = col.common,
lwd = 2)
labels <- c(labels, "Common")
cols <- c(cols, col.common)
lty <- c(lty, 1)
pt <- c(pt, -1)
}
if (!is.null(y$trended.dispersion)) {
o <- order(A)
lines(A[o], sqrt(y$trended.dispersion)[o], col = col.trend,
lwd = 2)
labels <- c(labels, "Trend")
cols <- c(cols, col.trend)
lty <- c(lty, 1)
pt <- c(pt, -1)
}
legend("topright", legend = labels, lty = lty, pch = pt,
pt.cex = cex, lwd = 2, col = cols,cex = fontsize/12)
}Data loading
targets <- read.delim(path.target,sep = ';')
targets$pool <- factor(targets$pool,levels = c('1','2','3'))cs <- catchSalmon(path.quant)Reading ../output/mouse/salmon/GSM7106766, 147556 transcripts, 100 gibbs samples
Reading ../output/mouse/salmon/GSM7106767, 147556 transcripts, 100 gibbs samples
Reading ../output/mouse/salmon/GSM7106768, 147556 transcripts, 100 gibbs samples
Reading ../output/mouse/salmon/GSM7106769, 147556 transcripts, 100 gibbs samples
Reading ../output/mouse/salmon/GSM7106770, 147556 transcripts, 100 gibbs samples
Reading ../output/mouse/salmon/GSM7106771, 147556 transcripts, 100 gibbs samples
Reading ../output/mouse/salmon/GSM7106772, 147556 transcripts, 100 gibbs samples
Reading ../output/mouse/salmon/GSM7106773, 147556 transcripts, 100 gibbs samples
Reading ../output/mouse/salmon/GSM7106774, 147556 transcripts, 100 gibbs samplestx.name <- strsplit2(rownames(cs$annotation),"\\|")
rownames(cs$annotation) <- rownames(cs$counts) <- tx.name[,1]
colnames(cs$counts) <- basename(colnames(cs$counts))entrez <- fread(path.entrez,col.names = c('TranscriptID','EntrezID'))
df.fa <- as.data.table(scanFasta(path.fasta))Warning: 2438 duplicate sequences were found in the input. Please check the summary table.TranscriptID.fa <- strsplit2(df.fa$TranscriptID,"\\|")[,1]
GeneID.fa <- strsplit2(df.fa$TranscriptID,"\\|")[,2]
TranscriptBioType.fa <- strsplit2(df.fa$TranscriptID,"\\|")[,8]
df.fa$TranscriptID <- TranscriptID.fa
df.fa$GeneID <- GeneID.fa
df.fa$TranscriptBioType <- TranscriptBioType.fa
df.fa[,allUnique := all(Unique),by = 'GeneID']
df.fa <- df.fa[order(GeneID,TranscriptID),]
gtf <- import(path.gtf)
gtf.tx <- gtf[gtf$type == 'transcript']
gtf.tx$EntrezID <- entrez$EntrezID[match(gtf.tx$transcript_id,entrez$TranscriptID)]
gtf.tx$keep.gene <- gtf.tx$gene_type %in% c('protein_coding','lncRNA') & #. (1)
as.character(seqnames(gtf.tx)) %in% paste0('chr',c(1:19,'X','Y')) & #. (2)
gtf.tx$gene_id %in% df.fa[allUnique == TRUE,unique(GeneID)] & #. (3)
!is.na(gtf.tx$EntrezID)
gtf.tx$keep.tx <- grepl("protein_coding|lncRNA",gtf.tx$transcript_type)Differential Transcript Usage
dge <- DGEList(counts = cs$counts/cs$annotation$Overdispersion,
samples = targets,
genes = cs$annotation)
dge.raw <- DGEList(counts = cs$counts,
samples = targets,
genes = cs$annotation)
dge$genes[,c('TranscriptID','GeneID')] <-
dge.raw$genes[,c('TranscriptID','GeneID')] <-
df.fa[match(rownames(dge),df.fa$TranscriptID),c('TranscriptID','GeneID')]
dge$genes[,c('TranscriptSymbol','GeneSymbol','EntrezID')] <-
dge.raw$genes[,c('TranscriptSymbol','GeneSymbol','EntrezID')] <-
mcols(gtf.tx)[match(rownames(dge),gtf.tx$transcript_id),c('transcript_name','gene_name','EntrezID')] |>
as.data.frame()keep <- filterByExpr(dge)
keep.gene <- gtf.tx$keep.gene[match(rownames(dge),gtf.tx$transcript_id)]
keep.tx <- gtf.tx$keep.tx[match(rownames(dge),gtf.tx$transcript_id)]
dge.filtr <- dge[keep & keep.gene & keep.tx,,keep.lib.sizes = FALSE]
dge.filtr <- normLibSizes(dge.filtr)
dge.raw.filtr <- dge.raw[keep & keep.gene & keep.tx,,keep.lib.sizes = FALSE]
dge.raw.filtr <- normLibSizes(dge.raw.filtr)par(mfrow = c(2,2))
y <- cpm(dge.filtr,log = TRUE)
plotMDS(y,col = dge.filtr$samples$color,
xlim = c(-6,3),ylim = c(-4,3))
plotMDS(y,labels = dge.filtr$samples$pool,col = dge.filtr$samples$color,
xlim = c(-6,3),ylim = c(-4,3))
y.nobatch <-
removeBatchEffect(x = y,
batch = dge.filtr$samples$pool,
group = dge.filtr$samples$group)
plotMDS(y.nobatch,col = dge.filtr$samples$color,
xlim = c(-6,3),ylim = c(-4,3))
plotMDS(y.nobatch,labels = dge.filtr$samples$pool,col = dge.filtr$samples$color,
xlim = c(-6,3),ylim = c(-4,3))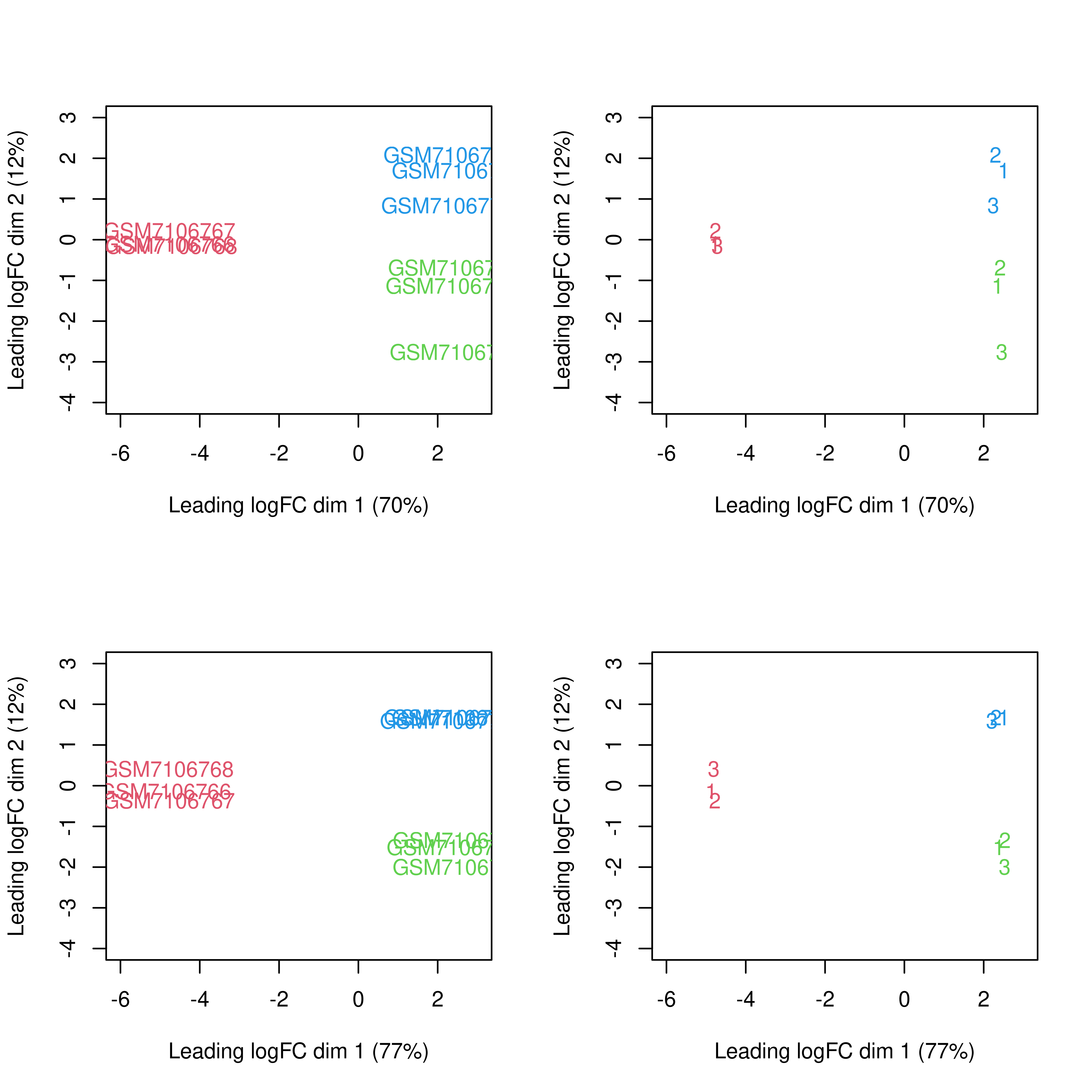
dev.off()null device
1 design <- model.matrix(~0+group,data = dge.filtr$samples)
colnames(design) <- sub("group","",colnames(design))
contr <- makeContrasts(LPvsBa = LP - Basal,
MLvsBa = ML - Basal,
MLvsLP = ML - LP,
LvsBa = 0.5*(LP+ML) - Basal,
levels = design)edgeR
fit.raw <- glmQLFit(y = dge.raw.filtr,design = design)
fit <- glmQLFit(y = dge.filtr,design = design)diffSplice
ds.raw.LPvsBa <- diffSplice(fit.raw, geneid = "GeneID", exonid = "TranscriptID",robust = TRUE,contrast = contr[,'LPvsBa'])Total number of exons: 29954
Total number of genes: 14632
Number of genes with 1 exon: 7067
Mean number of exons in a gene: 2
Max number of exons in a gene: 17 ds.raw.MLvsBa <- diffSplice(fit.raw, geneid = "GeneID", exonid = "TranscriptID",robust = TRUE,contrast = contr[,'MLvsBa'])Total number of exons: 29954
Total number of genes: 14632
Number of genes with 1 exon: 7067
Mean number of exons in a gene: 2
Max number of exons in a gene: 17 ds.raw.MLvsLP <- diffSplice(fit.raw, geneid = "GeneID", exonid = "TranscriptID",robust = TRUE,contrast = contr[,'MLvsLP'])Total number of exons: 29954
Total number of genes: 14632
Number of genes with 1 exon: 7067
Mean number of exons in a gene: 2
Max number of exons in a gene: 17 ds.raw.LvsBa <- diffSplice(fit.raw, geneid = "GeneID", exonid = "TranscriptID",robust = TRUE,contrast = contr[,'LvsBa'])Total number of exons: 29954
Total number of genes: 14632
Number of genes with 1 exon: 7067
Mean number of exons in a gene: 2
Max number of exons in a gene: 17 ds.LPvsBa <- diffSplice(fit, geneid = "GeneID", exonid = "TranscriptID",robust = TRUE,contrast = contr[,'LPvsBa'])Total number of exons: 29954
Total number of genes: 14632
Number of genes with 1 exon: 7067
Mean number of exons in a gene: 2
Max number of exons in a gene: 17 ds.MLvsBa <- diffSplice(fit, geneid = "GeneID", exonid = "TranscriptID",robust = TRUE,contrast = contr[,'MLvsBa'])Total number of exons: 29954
Total number of genes: 14632
Number of genes with 1 exon: 7067
Mean number of exons in a gene: 2
Max number of exons in a gene: 17 ds.MLvsLP <- diffSplice(fit, geneid = "GeneID", exonid = "TranscriptID",robust = TRUE,contrast = contr[,'MLvsLP'])Total number of exons: 29954
Total number of genes: 14632
Number of genes with 1 exon: 7067
Mean number of exons in a gene: 2
Max number of exons in a gene: 17 ds.LvsBa <- diffSplice(fit, geneid = "GeneID", exonid = "TranscriptID",robust = TRUE,contrast = contr[,'LvsBa'])Total number of exons: 29954
Total number of genes: 14632
Number of genes with 1 exon: 7067
Mean number of exons in a gene: 2
Max number of exons in a gene: 17 out.transcript.LPvsBa <- topSplice(ds.LPvsBa, test = "t",number = Inf)
out.gene.LPvsBa <- topSplice(ds.LPvsBa, test = "F", number = Inf)
out.transcript.MLvsBa <- topSplice(ds.MLvsBa, test = "t",number = Inf)
out.gene.MLvsBa <- topSplice(ds.MLvsBa, test = "F", number = Inf)
out.transcript.MLvsLP <- topSplice(ds.MLvsLP, test = "t",number = Inf)
out.gene.MLvsLP <- topSplice(ds.MLvsLP, test = "F", number = Inf)
out.transcript.LvsBa <- topSplice(ds.LvsBa, test = "t",number = Inf)
out.gene.LvsBa <- topSplice(ds.LvsBa, test = "F", number = Inf)summary(ds.raw.LPvsBa$gene.df.prior) Min. 1st Qu. Median Mean 3rd Qu. Max.
4.177 4.177 4.177 4.177 4.177 4.177 summary(ds.LPvsBa$gene.df.prior) Min. 1st Qu. Median Mean 3rd Qu. Max.
1.273 8.230 8.230 8.170 8.230 8.230 head(out.gene.LPvsBa,20) GeneID GeneSymbol EntrezID NExons F
ENSMUSG00000032366.16 ENSMUSG00000032366.16 Tpm1 22003 15 61.57262
ENSMUSG00000026131.22 ENSMUSG00000026131.22 Dst 13518 12 38.92348
ENSMUSG00000022791.20 ENSMUSG00000022791.20 Tnk2 51789 11 41.46390
ENSMUSG00000025290.18 ENSMUSG00000025290.18 Rps24 20088 9 46.63931
ENSMUSG00000005417.18 ENSMUSG00000005417.18 Mprip 26936 8 70.55188
ENSMUSG00000035325.17 ENSMUSG00000035325.17 Sec31a 69162 7 75.27037
ENSMUSG00000000631.21 ENSMUSG00000000631.21 Myo18a 360013 7 60.82077
ENSMUSG00000026987.17 ENSMUSG00000026987.17 Baz2b 407823 11 26.14503
ENSMUSG00000028649.23 ENSMUSG00000028649.23 Macf1 11426 11 23.67257
ENSMUSG00000021870.18 ENSMUSG00000021870.18 Slmap 83997 12 21.46028
ENSMUSG00000031328.17 ENSMUSG00000031328.17 Flna 192176 5 67.08571
ENSMUSG00000016487.16 ENSMUSG00000016487.16 Ppfibp1 67533 5 79.09824
ENSMUSG00000069601.15 ENSMUSG00000069601.15 Ank3 11735 14 16.13991
ENSMUSG00000020758.16 ENSMUSG00000020758.16 Itgb4 192897 7 53.46018
ENSMUSG00000003617.17 ENSMUSG00000003617.17 Cp 12870 6 48.73895
ENSMUSG00000021699.18 ENSMUSG00000021699.18 Pde4d 238871 5 68.42785
ENSMUSG00000025195.10 ENSMUSG00000025195.10 Dnmbp 71972 4 79.68960
ENSMUSG00000017897.19 ENSMUSG00000017897.19 Eya2 14049 4 97.52174
ENSMUSG00000032536.13 ENSMUSG00000032536.13 Trak1 67095 7 36.48084
ENSMUSG00000024589.19 ENSMUSG00000024589.19 Nedd4l 83814 9 20.57101
P.Value FDR
ENSMUSG00000032366.16 3.103767e-42 2.348000e-38
ENSMUSG00000026131.22 7.007902e-28 2.650739e-24
ENSMUSG00000022791.20 1.884195e-26 4.751313e-23
ENSMUSG00000025290.18 2.041619e-23 3.861212e-20
ENSMUSG00000005417.18 3.831376e-23 5.796872e-20
ENSMUSG00000035325.17 8.626840e-23 1.087701e-19
ENSMUSG00000000631.21 8.054858e-22 8.705001e-19
ENSMUSG00000026987.17 1.496884e-20 1.415491e-17
ENSMUSG00000028649.23 7.212320e-20 6.062356e-17
ENSMUSG00000021870.18 9.000020e-20 6.808515e-17
ENSMUSG00000031328.17 5.811140e-19 3.996480e-16
ENSMUSG00000016487.16 7.435928e-19 4.687733e-16
ENSMUSG00000069601.15 1.333623e-18 7.760661e-16
ENSMUSG00000020758.16 1.230927e-17 6.651400e-15
ENSMUSG00000003617.17 1.167510e-16 5.888143e-14
ENSMUSG00000021699.18 1.861145e-16 8.799725e-14
ENSMUSG00000025195.10 2.023675e-16 9.005353e-14
ENSMUSG00000017897.19 3.306905e-16 1.389819e-13
ENSMUSG00000032536.13 5.587118e-16 2.224555e-13
ENSMUSG00000024589.19 2.787341e-15 1.054312e-12head(out.gene.MLvsBa,20) GeneID GeneSymbol EntrezID NExons
ENSMUSG00000032366.16 ENSMUSG00000032366.16 Tpm1 22003 15
ENSMUSG00000026131.22 ENSMUSG00000026131.22 Dst 13518 12
ENSMUSG00000005417.18 ENSMUSG00000005417.18 Mprip 26936 8
ENSMUSG00000069601.15 ENSMUSG00000069601.15 Ank3 11735 14
ENSMUSG00000006345.11 ENSMUSG00000006345.11 Ggt1 14598 9
ENSMUSG00000035325.17 ENSMUSG00000035325.17 Sec31a 69162 7
ENSMUSG00000000631.21 ENSMUSG00000000631.21 Myo18a 360013 7
ENSMUSG00000021870.18 ENSMUSG00000021870.18 Slmap 83997 12
ENSMUSG00000025290.18 ENSMUSG00000025290.18 Rps24 20088 9
ENSMUSG00000028649.23 ENSMUSG00000028649.23 Macf1 11426 11
ENSMUSG00000022791.20 ENSMUSG00000022791.20 Tnk2 51789 11
ENSMUSG00000020758.16 ENSMUSG00000020758.16 Itgb4 192897 7
ENSMUSG00000031328.17 ENSMUSG00000031328.17 Flna 192176 5
ENSMUSG00000026987.17 ENSMUSG00000026987.17 Baz2b 407823 11
ENSMUSG00000025195.10 ENSMUSG00000025195.10 Dnmbp 71972 4
ENSMUSG00000016487.16 ENSMUSG00000016487.16 Ppfibp1 67533 5
ENSMUSG00000022587.16 ENSMUSG00000022587.16 Ly6e 17069 10
ENSMUSG00000002985.17 ENSMUSG00000002985.17 Apoe 11816 6
ENSMUSG00000032536.13 ENSMUSG00000032536.13 Trak1 67095 7
ENSMUSG00000027455.17 ENSMUSG00000027455.17 Nsfl1c 386649 4
F P.Value FDR
ENSMUSG00000032366.16 72.84535 1.902688e-45 1.439383e-41
ENSMUSG00000026131.22 58.25486 5.844034e-34 2.210506e-30
ENSMUSG00000005417.18 85.98632 4.986551e-25 1.257442e-21
ENSMUSG00000069601.15 24.71621 1.065083e-24 2.014338e-21
ENSMUSG00000006345.11 40.43033 1.462226e-22 2.004188e-19
ENSMUSG00000035325.17 73.16849 1.589574e-22 2.004188e-19
ENSMUSG00000000631.21 63.19290 3.384742e-22 3.657939e-19
ENSMUSG00000021870.18 24.79689 1.189489e-21 1.124810e-18
ENSMUSG00000025290.18 38.85818 2.282764e-21 1.918790e-18
ENSMUSG00000028649.23 25.39400 9.461750e-21 7.157814e-18
ENSMUSG00000022791.20 26.27866 1.421162e-20 9.773720e-18
ENSMUSG00000020758.16 70.51814 8.511853e-20 5.366014e-17
ENSMUSG00000031328.17 71.82415 1.466444e-19 8.533576e-17
ENSMUSG00000026987.17 23.97728 1.678805e-19 9.071544e-17
ENSMUSG00000025195.10 119.63230 2.283005e-19 1.151396e-16
ENSMUSG00000016487.16 73.08383 3.122305e-18 1.476265e-15
ENSMUSG00000022587.16 24.41239 4.798830e-18 2.135479e-15
ENSMUSG00000002985.17 48.47224 7.753383e-17 3.258575e-14
ENSMUSG00000032536.13 37.95900 2.637271e-16 1.050050e-13
ENSMUSG00000027455.17 89.89833 1.066914e-15 4.035602e-13head(out.gene.MLvsLP,20) GeneID GeneSymbol EntrezID NExons
ENSMUSG00000006345.11 ENSMUSG00000006345.11 Ggt1 14598 9
ENSMUSG00000021000.18 ENSMUSG00000021000.18 Mia2 338320 8
ENSMUSG00000045636.17 ENSMUSG00000045636.17 Mtus1 102103 6
ENSMUSG00000028337.15 ENSMUSG00000028337.15 Coro2a 107684 2
ENSMUSG00000022791.20 ENSMUSG00000022791.20 Tnk2 51789 11
ENSMUSG00000026131.22 ENSMUSG00000026131.22 Dst 13518 12
ENSMUSG00000024608.12 ENSMUSG00000024608.12 Rps14 20044 4
ENSMUSG00000023017.11 ENSMUSG00000023017.11 Asic1 11419 2
ENSMUSG00000002985.17 ENSMUSG00000002985.17 Apoe 11816 6
ENSMUSG00000029381.17 ENSMUSG00000029381.17 Shroom3 27428 5
ENSMUSG00000031133.13 ENSMUSG00000031133.13 Arhgef6 73341 5
ENSMUSG00000038007.15 ENSMUSG00000038007.15 Acer2 230379 3
ENSMUSG00000004043.15 ENSMUSG00000004043.15 Stat5a 20850 6
ENSMUSG00000044948.18 ENSMUSG00000044948.18 Cfap43 100048534 2
ENSMUSG00000001211.16 ENSMUSG00000001211.16 Agpat3 28169 5
ENSMUSG00000052033.14 ENSMUSG00000052033.14 Pfdn4 109054 5
ENSMUSG00000021710.12 ENSMUSG00000021710.12 Nln 75805 3
ENSMUSG00000028865.15 ENSMUSG00000028865.15 Cd164l2 69655 3
ENSMUSG00000037012.19 ENSMUSG00000037012.19 Hk1 15275 5
ENSMUSG00000043051.19 ENSMUSG00000043051.19 Disc1 244667 3
F P.Value FDR
ENSMUSG00000006345.11 9.671981 9.142872e-09 4.109143e-05
ENSMUSG00000021000.18 10.652731 1.122517e-08 4.109143e-05
ENSMUSG00000045636.17 14.143610 1.629535e-08 4.109143e-05
ENSMUSG00000028337.15 63.092472 1.210010e-07 2.288432e-04
ENSMUSG00000022791.20 6.750281 2.178894e-07 3.114506e-04
ENSMUSG00000026131.22 6.164151 2.824700e-07 3.114506e-04
ENSMUSG00000024608.12 18.269788 2.881895e-07 3.114506e-04
ENSMUSG00000023017.11 54.408588 4.383612e-07 4.145253e-04
ENSMUSG00000002985.17 10.819071 7.808127e-07 6.563164e-04
ENSMUSG00000029381.17 12.296600 1.598942e-06 1.209600e-03
ENSMUSG00000031133.13 11.213043 2.818868e-06 1.938613e-03
ENSMUSG00000038007.15 20.503755 4.386003e-06 2.765009e-03
ENSMUSG00000004043.15 8.957834 6.198328e-06 3.606950e-03
ENSMUSG00000044948.18 35.665474 1.156719e-05 6.115109e-03
ENSMUSG00000001211.16 10.011705 1.216465e-05 6.115109e-03
ENSMUSG00000052033.14 9.654134 1.336658e-05 6.115109e-03
ENSMUSG00000021710.12 16.759582 1.374182e-05 6.115109e-03
ENSMUSG00000028865.15 17.182444 1.715309e-05 7.209064e-03
ENSMUSG00000037012.19 9.289442 2.199436e-05 8.282758e-03
ENSMUSG00000043051.19 17.269158 2.294675e-05 8.282758e-03head(out.gene.LvsBa,20) GeneID GeneSymbol EntrezID NExons
ENSMUSG00000032366.16 ENSMUSG00000032366.16 Tpm1 22003 15
ENSMUSG00000026131.22 ENSMUSG00000026131.22 Dst 13518 12
ENSMUSG00000005417.18 ENSMUSG00000005417.18 Mprip 26936 8
ENSMUSG00000022791.20 ENSMUSG00000022791.20 Tnk2 51789 11
ENSMUSG00000025290.18 ENSMUSG00000025290.18 Rps24 20088 9
ENSMUSG00000021870.18 ENSMUSG00000021870.18 Slmap 83997 12
ENSMUSG00000035325.17 ENSMUSG00000035325.17 Sec31a 69162 7
ENSMUSG00000026987.17 ENSMUSG00000026987.17 Baz2b 407823 11
ENSMUSG00000069601.15 ENSMUSG00000069601.15 Ank3 11735 14
ENSMUSG00000028649.23 ENSMUSG00000028649.23 Macf1 11426 11
ENSMUSG00000000631.21 ENSMUSG00000000631.21 Myo18a 360013 7
ENSMUSG00000020758.16 ENSMUSG00000020758.16 Itgb4 192897 7
ENSMUSG00000016487.16 ENSMUSG00000016487.16 Ppfibp1 67533 5
ENSMUSG00000031328.17 ENSMUSG00000031328.17 Flna 192176 5
ENSMUSG00000025195.10 ENSMUSG00000025195.10 Dnmbp 71972 4
ENSMUSG00000032536.13 ENSMUSG00000032536.13 Trak1 67095 7
ENSMUSG00000022587.16 ENSMUSG00000022587.16 Ly6e 17069 10
ENSMUSG00000051586.18 ENSMUSG00000051586.18 Mical3 194401 8
ENSMUSG00000026721.17 ENSMUSG00000026721.17 Rabgap1l 29809 8
ENSMUSG00000006345.11 ENSMUSG00000006345.11 Ggt1 14598 9
F P.Value FDR
ENSMUSG00000032366.16 89.62684 1.753088e-49 1.326211e-45
ENSMUSG00000026131.22 57.04387 1.236543e-33 4.677222e-30
ENSMUSG00000005417.18 116.48500 5.572209e-28 1.405125e-24
ENSMUSG00000022791.20 42.70691 7.520021e-27 1.422224e-23
ENSMUSG00000025290.18 58.70369 4.397237e-26 6.653020e-23
ENSMUSG00000021870.18 33.37751 9.735968e-26 1.227543e-22
ENSMUSG00000035325.17 101.78473 1.153442e-25 1.246542e-22
ENSMUSG00000026987.17 37.10061 5.068757e-25 4.572634e-22
ENSMUSG00000069601.15 25.19360 5.440014e-25 4.572634e-22
ENSMUSG00000028649.23 32.67393 4.726445e-24 3.575555e-21
ENSMUSG00000000631.21 71.13761 2.250675e-23 1.547850e-20
ENSMUSG00000020758.16 91.43121 7.092377e-22 4.471153e-19
ENSMUSG00000016487.16 113.53720 8.948931e-22 5.207589e-19
ENSMUSG00000031328.17 81.19961 1.191576e-20 6.438768e-18
ENSMUSG00000025195.10 126.09731 9.267966e-20 4.674144e-17
ENSMUSG00000032536.13 52.39220 4.919518e-19 2.326009e-16
ENSMUSG00000022587.16 25.75031 1.238993e-18 5.513521e-16
ENSMUSG00000051586.18 30.71680 2.454190e-18 1.031442e-15
ENSMUSG00000026721.17 30.65362 6.645582e-18 2.645991e-15
ENSMUSG00000006345.11 25.55395 1.899221e-17 7.183803e-15table(out.gene.LPvsBa$FDR < 0.05)
FALSE TRUE
6309 1256 out.gene.LPvsBa[out.gene.LPvsBa$GeneSymbol %in% c('Foxp1','Ezh2','Asap1'),] GeneID GeneSymbol EntrezID NExons
ENSMUSG00000030067.18 ENSMUSG00000030067.18 Foxp1 108655 11
ENSMUSG00000022377.17 ENSMUSG00000022377.17 Asap1 13196 4
ENSMUSG00000029687.17 ENSMUSG00000029687.17 Ezh2 14056 4
F P.Value FDR
ENSMUSG00000030067.18 13.431045 3.321706e-13 7.179629e-11
ENSMUSG00000022377.17 18.623702 2.627593e-07 1.077850e-05
ENSMUSG00000029687.17 4.749363 7.485836e-03 4.644383e-02head(out.transcript.LPvsBa,20) Length EffectiveLength Overdispersion
ENSMUST00000113696.8 1714 1393.417 6.058497
ENSMUST00000097786.10 17212 16891.417 11.289582
ENSMUST00000092887.11 7304 6983.417 18.089224
ENSMUST00000116371.8 8766 8445.417 20.285463
ENSMUST00000113705.8 1759 1438.417 7.934639
ENSMUST00000101454.9 8345 8024.417 62.251681
ENSMUST00000153136.2 890 569.769 2.307391
ENSMUST00000223999.2 525 234.370 5.287670
ENSMUST00000112384.10 468 194.337 6.131152
ENSMUST00000182886.8 4047 3726.417 16.866773
ENSMUST00000182812.8 1093 772.417 2.476381
ENSMUST00000111623.9 4782 4461.417 11.793842
ENSMUST00000108325.9 3183 2862.417 20.002417
ENSMUST00000050905.16 1259 938.417 5.876432
ENSMUST00000106460.9 5919 5598.417 35.672607
ENSMUST00000063433.8 2460 2139.417 4.602893
ENSMUST00000016631.14 4851 4530.417 3.612108
ENSMUST00000238988.2 3353 3032.417 19.736995
ENSMUST00000080933.13 5669 5348.417 20.189698
ENSMUST00000115123.9 4226 3905.417 40.910626
TranscriptID GeneID
ENSMUST00000113696.8 ENSMUST00000113696.8 ENSMUSG00000032366.16
ENSMUST00000097786.10 ENSMUST00000097786.10 ENSMUSG00000026131.22
ENSMUST00000092887.11 ENSMUST00000092887.11 ENSMUSG00000000631.21
ENSMUST00000116371.8 ENSMUST00000116371.8 ENSMUSG00000005417.18
ENSMUST00000113705.8 ENSMUST00000113705.8 ENSMUSG00000032366.16
ENSMUST00000101454.9 ENSMUST00000101454.9 ENSMUSG00000031328.17
ENSMUST00000153136.2 ENSMUST00000153136.2 ENSMUSG00000026987.17
ENSMUST00000223999.2 ENSMUST00000223999.2 ENSMUSG00000025290.18
ENSMUST00000112384.10 ENSMUST00000112384.10 ENSMUSG00000025290.18
ENSMUST00000182886.8 ENSMUST00000182886.8 ENSMUSG00000035325.17
ENSMUST00000182812.8 ENSMUST00000182812.8 ENSMUSG00000035325.17
ENSMUST00000111623.9 ENSMUST00000111623.9 ENSMUSG00000016487.16
ENSMUST00000108325.9 ENSMUST00000108325.9 ENSMUSG00000003617.17
ENSMUST00000050905.16 ENSMUST00000050905.16 ENSMUSG00000032366.16
ENSMUST00000106460.9 ENSMUST00000106460.9 ENSMUSG00000020758.16
ENSMUST00000063433.8 ENSMUST00000063433.8 ENSMUSG00000017897.19
ENSMUST00000016631.14 ENSMUST00000016631.14 ENSMUSG00000016487.16
ENSMUST00000238988.2 ENSMUST00000238988.2 ENSMUSG00000032536.13
ENSMUST00000080933.13 ENSMUST00000080933.13 ENSMUSG00000028284.14
ENSMUST00000115123.9 ENSMUST00000115123.9 ENSMUSG00000022791.20
TranscriptSymbol GeneSymbol EntrezID logFC t
ENSMUST00000113696.8 Tpm1-212 Tpm1 22003 5.725755 16.11515
ENSMUST00000097786.10 Dst-202 Dst 13518 9.537490 14.20699
ENSMUST00000092887.11 Myo18a-203 Myo18a 360013 8.684254 16.82793
ENSMUST00000116371.8 Mprip-204 Mprip 26936 -7.744796 -15.95332
ENSMUST00000113705.8 Tpm1-215 Tpm1 22003 -4.885498 -11.80448
ENSMUST00000101454.9 Flna-202 Flna 192176 7.719149 15.78598
ENSMUST00000153136.2 Baz2b-213 Baz2b 407823 -3.543286 -12.73793
ENSMUST00000223999.2 Rps24-205 Rps24 20088 -4.295030 -13.08691
ENSMUST00000112384.10 Rps24-201 Rps24 20088 4.168845 12.95657
ENSMUST00000182886.8 Sec31a-210 Sec31a 69162 7.856050 14.30556
ENSMUST00000182812.8 Sec31a-208 Sec31a 69162 -7.791285 -14.24490
ENSMUST00000111623.9 Ppfibp1-202 Ppfibp1 67533 -4.762570 -15.07771
ENSMUST00000108325.9 Cp-203 Cp 12870 8.738240 13.49281
ENSMUST00000050905.16 Tpm1-203 Tpm1 22003 3.813449 10.18045
ENSMUST00000106460.9 Itgb4-204 Itgb4 192897 -6.998445 -13.28082
ENSMUST00000063433.8 Eya2-201 Eya2 14049 5.121910 15.03375
ENSMUST00000016631.14 Ppfibp1-201 Ppfibp1 67533 2.542253 12.83323
ENSMUST00000238988.2 Trak1-215 Trak1 67095 -6.585212 -12.01177
ENSMUST00000080933.13 Map3k7-202 Map3k7 26409 -3.367739 -13.08106
ENSMUST00000115123.9 Tnk2-204 Tnk2 51789 -6.924844 -10.10517
P.Value FDR
ENSMUST00000113696.8 2.294375e-29 5.251136e-25
ENSMUST00000097786.10 8.795338e-24 1.006494e-19
ENSMUST00000092887.11 9.687227e-23 7.390385e-19
ENSMUST00000116371.8 7.295853e-21 4.174505e-17
ENSMUST00000113705.8 1.453987e-20 5.785846e-17
ENSMUST00000101454.9 1.516803e-20 5.785846e-17
ENSMUST00000153136.2 1.995921e-20 6.525805e-17
ENSMUST00000223999.2 1.603277e-19 4.586775e-16
ENSMUST00000112384.10 2.534642e-19 6.445595e-16
ENSMUST00000182886.8 5.559523e-19 1.272408e-15
ENSMUST00000182812.8 6.567054e-19 1.366365e-15
ENSMUST00000111623.9 1.941174e-18 3.702304e-15
ENSMUST00000108325.9 4.432966e-17 7.631823e-14
ENSMUST00000050905.16 4.668393e-17 7.631823e-14
ENSMUST00000106460.9 2.653817e-16 4.049194e-13
ENSMUST00000063433.8 4.429212e-16 6.335710e-13
ENSMUST00000016631.14 4.902679e-16 6.600448e-13
ENSMUST00000238988.2 7.509612e-16 9.548471e-13
ENSMUST00000080933.13 1.174446e-15 1.414713e-12
ENSMUST00000115123.9 1.287014e-15 1.472795e-12head(out.transcript.MLvsBa,20) Length EffectiveLength Overdispersion
ENSMUST00000113696.8 1714 1393.417 6.058497
ENSMUST00000097786.10 17212 16891.417 11.289582
ENSMUST00000116371.8 8766 8445.417 20.285463
ENSMUST00000113705.8 1759 1438.417 7.934639
ENSMUST00000092887.11 7304 6983.417 18.089224
ENSMUST00000101454.9 8345 8024.417 62.251681
ENSMUST00000182812.8 1093 772.417 2.476381
ENSMUST00000050905.16 1259 938.417 5.876432
ENSMUST00000187486.7 4539 4218.417 3.716784
ENSMUST00000153136.2 890 569.769 2.307391
ENSMUST00000223999.2 525 234.370 5.287670
ENSMUST00000106460.9 5919 5598.417 35.672607
ENSMUST00000111623.9 4782 4461.417 11.793842
ENSMUST00000112384.10 468 194.337 6.131152
ENSMUST00000182886.8 4047 3726.417 16.866773
ENSMUST00000238988.2 3353 3032.417 19.736995
ENSMUST00000181974.8 3751 3415.417 11.558536
ENSMUST00000097785.10 23251 22930.417 37.491850
ENSMUST00000212396.2 6100 5779.417 3.624965
ENSMUST00000212048.2 4186 3865.417 1.905066
TranscriptID GeneID
ENSMUST00000113696.8 ENSMUST00000113696.8 ENSMUSG00000032366.16
ENSMUST00000097786.10 ENSMUST00000097786.10 ENSMUSG00000026131.22
ENSMUST00000116371.8 ENSMUST00000116371.8 ENSMUSG00000005417.18
ENSMUST00000113705.8 ENSMUST00000113705.8 ENSMUSG00000032366.16
ENSMUST00000092887.11 ENSMUST00000092887.11 ENSMUSG00000000631.21
ENSMUST00000101454.9 ENSMUST00000101454.9 ENSMUSG00000031328.17
ENSMUST00000182812.8 ENSMUST00000182812.8 ENSMUSG00000035325.17
ENSMUST00000050905.16 ENSMUST00000050905.16 ENSMUSG00000032366.16
ENSMUST00000187486.7 ENSMUST00000187486.7 ENSMUSG00000026131.22
ENSMUST00000153136.2 ENSMUST00000153136.2 ENSMUSG00000026987.17
ENSMUST00000223999.2 ENSMUST00000223999.2 ENSMUSG00000025290.18
ENSMUST00000106460.9 ENSMUST00000106460.9 ENSMUSG00000020758.16
ENSMUST00000111623.9 ENSMUST00000111623.9 ENSMUSG00000016487.16
ENSMUST00000112384.10 ENSMUST00000112384.10 ENSMUSG00000025290.18
ENSMUST00000182886.8 ENSMUST00000182886.8 ENSMUSG00000035325.17
ENSMUST00000238988.2 ENSMUST00000238988.2 ENSMUSG00000032536.13
ENSMUST00000181974.8 ENSMUST00000181974.8 ENSMUSG00000069601.15
ENSMUST00000097785.10 ENSMUST00000097785.10 ENSMUSG00000026131.22
ENSMUST00000212396.2 ENSMUST00000212396.2 ENSMUSG00000025195.10
ENSMUST00000212048.2 ENSMUST00000212048.2 ENSMUSG00000025195.10
TranscriptSymbol GeneSymbol EntrezID logFC t
ENSMUST00000113696.8 Tpm1-212 Tpm1 22003 5.843001 16.40302
ENSMUST00000097786.10 Dst-202 Dst 13518 9.982264 15.31755
ENSMUST00000116371.8 Mprip-204 Mprip 26936 -7.802606 -18.17166
ENSMUST00000113705.8 Tpm1-215 Tpm1 22003 -4.639910 -12.64791
ENSMUST00000092887.11 Myo18a-203 Myo18a 360013 8.387651 16.09720
ENSMUST00000101454.9 Flna-202 Flna 192176 7.768713 16.05892
ENSMUST00000182812.8 Sec31a-208 Sec31a 69162 -4.428908 -14.95384
ENSMUST00000050905.16 Tpm1-203 Tpm1 22003 3.997142 11.13444
ENSMUST00000187486.7 Dst-217 Dst 13518 7.136132 11.56893
ENSMUST00000153136.2 Baz2b-213 Baz2b 407823 -2.426423 -11.75416
ENSMUST00000223999.2 Rps24-205 Rps24 20088 -3.767190 -12.22724
ENSMUST00000106460.9 Itgb4-204 Itgb4 192897 -6.958211 -15.08806
ENSMUST00000111623.9 Ppfibp1-202 Ppfibp1 67533 -3.249190 -14.42305
ENSMUST00000112384.10 Rps24-201 Rps24 20088 3.591507 11.48674
ENSMUST00000182886.8 Sec31a-210 Sec31a 69162 7.220616 12.43882
ENSMUST00000238988.2 Trak1-215 Trak1 67095 -6.255212 -12.62630
ENSMUST00000181974.8 Ank3-207 Ank3 11735 4.134726 10.03677
ENSMUST00000097785.10 Dst-201 Dst 13518 -4.190308 -10.29581
ENSMUST00000212396.2 Dnmbp-205 Dnmbp 71972 -3.312801 -13.82316
ENSMUST00000212048.2 Dnmbp-203 Dnmbp 71972 3.331550 13.12552
P.Value FDR
ENSMUST00000113696.8 6.410959e-30 1.467276e-25
ENSMUST00000097786.10 9.878200e-26 1.130412e-21
ENSMUST00000116371.8 3.305327e-23 2.521634e-19
ENSMUST00000113705.8 2.358254e-22 1.349334e-18
ENSMUST00000092887.11 6.811969e-22 3.118111e-18
ENSMUST00000101454.9 7.611756e-21 2.903504e-17
ENSMUST00000182812.8 9.635903e-20 3.150527e-16
ENSMUST00000050905.16 3.997520e-19 1.143641e-15
ENSMUST00000187486.7 6.862194e-19 1.745056e-15
ENSMUST00000153136.2 1.131050e-18 2.588634e-15
ENSMUST00000223999.2 3.421923e-18 6.885706e-15
ENSMUST00000106460.9 3.610280e-18 6.885706e-15
ENSMUST00000111623.9 9.178506e-18 1.615911e-14
ENSMUST00000112384.10 5.147343e-17 8.414803e-14
ENSMUST00000182886.8 1.152067e-16 1.757824e-13
ENSMUST00000238988.2 1.259495e-16 1.801629e-13
ENSMUST00000181974.8 1.605422e-16 2.161370e-13
ENSMUST00000097785.10 2.037339e-16 2.590477e-13
ENSMUST00000212396.2 2.233102e-16 2.689947e-13
ENSMUST00000212048.2 1.141003e-15 1.305706e-12head(out.transcript.MLvsLP,20) Length EffectiveLength Overdispersion
ENSMUST00000059115.13 6548 6227.417 9.176039
ENSMUST00000219140.3 4194 3873.417 3.283965
ENSMUST00000236652.2 367 131.390 1.373704
ENSMUST00000145079.8 676 361.546 1.819521
ENSMUST00000176986.8 1604 1283.417 2.952235
ENSMUST00000107756.4 4154 3833.417 3.221990
ENSMUST00000127304.2 642 330.598 1.000000
ENSMUST00000115124.9 4516 4195.417 40.643195
ENSMUST00000023758.9 4114 3793.417 2.843403
ENSMUST00000227670.2 2788 2467.417 4.264119
ENSMUST00000075087.8 816 496.416 4.785076
ENSMUST00000152761.8 784 464.953 2.508438
ENSMUST00000105389.8 3678 3357.417 6.816707
ENSMUST00000135874.2 2943 2622.417 1.184062
ENSMUST00000069430.15 2869 2548.417 18.887385
ENSMUST00000105910.2 986 665.431 1.056655
ENSMUST00000088132.13 2447 2126.417 5.137227
ENSMUST00000135803.8 801 481.644 1.400323
ENSMUST00000122389.8 1694 1354.417 1.214908
ENSMUST00000115672.2 2909 2588.417 1.597177
TranscriptID GeneID
ENSMUST00000059115.13 ENSMUST00000059115.13 ENSMUSG00000045636.17
ENSMUST00000219140.3 ENSMUST00000219140.3 ENSMUSG00000021000.18
ENSMUST00000236652.2 ENSMUST00000236652.2 ENSMUSG00000024608.12
ENSMUST00000145079.8 ENSMUST00000145079.8 ENSMUSG00000006345.11
ENSMUST00000176986.8 ENSMUST00000176986.8 ENSMUSG00000031133.13
ENSMUST00000107756.4 ENSMUST00000107756.4 ENSMUSG00000028337.15
ENSMUST00000127304.2 ENSMUST00000127304.2 ENSMUSG00000028337.15
ENSMUST00000115124.9 ENSMUST00000115124.9 ENSMUSG00000022791.20
ENSMUST00000023758.9 ENSMUST00000023758.9 ENSMUSG00000023017.11
ENSMUST00000227670.2 ENSMUST00000227670.2 ENSMUSG00000023017.11
ENSMUST00000075087.8 ENSMUST00000075087.8 ENSMUSG00000052033.14
ENSMUST00000152761.8 ENSMUST00000152761.8 ENSMUSG00000037012.19
ENSMUST00000105389.8 ENSMUST00000105389.8 ENSMUSG00000001211.16
ENSMUST00000135874.2 ENSMUST00000135874.2 ENSMUSG00000038007.15
ENSMUST00000069430.15 ENSMUST00000069430.15 ENSMUSG00000021000.18
ENSMUST00000105910.2 ENSMUST00000105910.2 ENSMUSG00000028865.15
ENSMUST00000088132.13 ENSMUST00000088132.13 ENSMUSG00000017897.19
ENSMUST00000135803.8 ENSMUST00000135803.8 ENSMUSG00000026821.17
ENSMUST00000122389.8 ENSMUST00000122389.8 ENSMUSG00000043051.19
ENSMUST00000115672.2 ENSMUST00000115672.2 ENSMUSG00000032000.18
TranscriptSymbol GeneSymbol EntrezID logFC t
ENSMUST00000059115.13 Mtus1-202 Mtus1 102103 -1.3865726 -6.927530
ENSMUST00000219140.3 Mia2-222 Mia2 338320 1.8757371 6.591628
ENSMUST00000236652.2 Rps14-210 Rps14 20044 1.7810520 7.321235
ENSMUST00000145079.8 Ggt1-214 Ggt1 14598 -2.8654992 -6.360829
ENSMUST00000176986.8 Arhgef6-208 Arhgef6 73341 2.3476637 6.613627
ENSMUST00000107756.4 Coro2a-202 Coro2a 107684 2.5885581 7.943077
ENSMUST00000127304.2 Coro2a-204 Coro2a 107684 -2.5885581 -7.943077
ENSMUST00000115124.9 Tnk2-205 Tnk2 51789 2.4853317 5.581714
ENSMUST00000023758.9 Asic1-201 Asic1 11419 -3.1208505 -7.376218
ENSMUST00000227670.2 Asic1-203 Asic1 11419 3.1208505 7.376218
ENSMUST00000075087.8 Pfdn4-202 Pfdn4 109054 -2.3464356 -5.971252
ENSMUST00000152761.8 Hk1-218 Hk1 15275 -1.4232565 -5.989003
ENSMUST00000105389.8 Agpat3-204 Agpat3 28169 -1.3075798 -5.707978
ENSMUST00000135874.2 Acer2-204 Acer2 230379 1.2610295 6.063741
ENSMUST00000069430.15 Mia2-201 Mia2 338320 -1.3252988 -5.137141
ENSMUST00000105910.2 Cd164l2-201 Cd164l2 69655 1.5111046 5.826615
ENSMUST00000088132.13 Eya2-202 Eya2 14049 1.9515783 5.565517
ENSMUST00000135803.8 Ralgds-206 Ralgds 19730 0.9286164 5.234545
ENSMUST00000122389.8 Disc1-206 Disc1 244667 2.2795145 5.871323
ENSMUST00000115672.2 Birc3-202 Birc3 11796 0.8475912 5.243062
P.Value FDR
ENSMUST00000059115.13 9.351224e-09 0.0001240438
ENSMUST00000219140.3 1.160030e-08 0.0001240438
ENSMUST00000236652.2 1.625951e-08 0.0001240438
ENSMUST00000145079.8 2.171401e-08 0.0001242421
ENSMUST00000176986.8 5.240045e-08 0.0002398578
ENSMUST00000107756.4 1.210010e-07 0.0003956215
ENSMUST00000127304.2 1.210010e-07 0.0003956215
ENSMUST00000115124.9 3.660861e-07 0.0009776584
ENSMUST00000023758.9 4.383612e-07 0.0009776584
ENSMUST00000227670.2 4.383612e-07 0.0009776584
ENSMUST00000075087.8 4.698843e-07 0.0009776584
ENSMUST00000152761.8 5.197076e-07 0.0009912124
ENSMUST00000105389.8 1.409573e-06 0.0024816072
ENSMUST00000135874.2 2.030865e-06 0.0033200284
ENSMUST00000069430.15 3.094822e-06 0.0047220795
ENSMUST00000105910.2 3.760475e-06 0.0051152560
ENSMUST00000088132.13 3.799509e-06 0.0051152560
ENSMUST00000135803.8 4.374556e-06 0.0055622477
ENSMUST00000122389.8 4.766821e-06 0.0057420123
ENSMUST00000115672.2 6.105390e-06 0.0067086253head(out.transcript.LvsBa,20) Length EffectiveLength Overdispersion
ENSMUST00000113696.8 1714 1393.417 6.058497
ENSMUST00000097786.10 17212 16891.417 11.289582
ENSMUST00000116371.8 8766 8445.417 20.285463
ENSMUST00000113705.8 1759 1438.417 7.934639
ENSMUST00000153136.2 890 569.769 2.307391
ENSMUST00000223999.2 525 234.370 5.287670
ENSMUST00000092887.11 7304 6983.417 18.089224
ENSMUST00000050905.16 1259 938.417 5.876432
ENSMUST00000182812.8 1093 772.417 2.476381
ENSMUST00000101454.9 8345 8024.417 62.251681
ENSMUST00000111623.9 4782 4461.417 11.793842
ENSMUST00000106460.9 5919 5598.417 35.672607
ENSMUST00000112384.10 468 194.337 6.131152
ENSMUST00000182886.8 4047 3726.417 16.866773
ENSMUST00000238988.2 3353 3032.417 19.736995
ENSMUST00000115123.9 4226 3905.417 40.910626
ENSMUST00000016631.14 4851 4530.417 3.612108
ENSMUST00000151602.4 992 671.427 2.894139
ENSMUST00000080933.13 5669 5348.417 20.189698
ENSMUST00000108325.9 3183 2862.417 20.002417
TranscriptID GeneID
ENSMUST00000113696.8 ENSMUST00000113696.8 ENSMUSG00000032366.16
ENSMUST00000097786.10 ENSMUST00000097786.10 ENSMUSG00000026131.22
ENSMUST00000116371.8 ENSMUST00000116371.8 ENSMUSG00000005417.18
ENSMUST00000113705.8 ENSMUST00000113705.8 ENSMUSG00000032366.16
ENSMUST00000153136.2 ENSMUST00000153136.2 ENSMUSG00000026987.17
ENSMUST00000223999.2 ENSMUST00000223999.2 ENSMUSG00000025290.18
ENSMUST00000092887.11 ENSMUST00000092887.11 ENSMUSG00000000631.21
ENSMUST00000050905.16 ENSMUST00000050905.16 ENSMUSG00000032366.16
ENSMUST00000182812.8 ENSMUST00000182812.8 ENSMUSG00000035325.17
ENSMUST00000101454.9 ENSMUST00000101454.9 ENSMUSG00000031328.17
ENSMUST00000111623.9 ENSMUST00000111623.9 ENSMUSG00000016487.16
ENSMUST00000106460.9 ENSMUST00000106460.9 ENSMUSG00000020758.16
ENSMUST00000112384.10 ENSMUST00000112384.10 ENSMUSG00000025290.18
ENSMUST00000182886.8 ENSMUST00000182886.8 ENSMUSG00000035325.17
ENSMUST00000238988.2 ENSMUST00000238988.2 ENSMUSG00000032536.13
ENSMUST00000115123.9 ENSMUST00000115123.9 ENSMUSG00000022791.20
ENSMUST00000016631.14 ENSMUST00000016631.14 ENSMUSG00000016487.16
ENSMUST00000151602.4 ENSMUST00000151602.4 ENSMUSG00000051586.18
ENSMUST00000080933.13 ENSMUST00000080933.13 ENSMUSG00000028284.14
ENSMUST00000108325.9 ENSMUST00000108325.9 ENSMUSG00000003617.17
TranscriptSymbol GeneSymbol EntrezID logFC t
ENSMUST00000113696.8 Tpm1-212 Tpm1 22003 6.486538 18.06053
ENSMUST00000097786.10 Dst-202 Dst 13518 10.053352 15.76129
ENSMUST00000116371.8 Mprip-204 Mprip 26936 -7.680833 -21.66116
ENSMUST00000113705.8 Tpm1-215 Tpm1 22003 -4.060545 -13.81722
ENSMUST00000153136.2 Baz2b-213 Baz2b 407823 -2.905632 -15.32018
ENSMUST00000223999.2 Rps24-205 Rps24 20088 -3.877061 -16.05459
ENSMUST00000092887.11 Myo18a-203 Myo18a 360013 8.619093 17.58371
ENSMUST00000050905.16 Tpm1-203 Tpm1 22003 4.607455 13.18956
ENSMUST00000182812.8 Sec31a-208 Sec31a 69162 -5.960175 -18.31754
ENSMUST00000101454.9 Flna-202 Flna 192176 7.877288 17.04576
ENSMUST00000111623.9 Ppfibp1-202 Ppfibp1 67533 -3.909690 -18.05886
ENSMUST00000106460.9 Itgb4-204 Itgb4 192897 -6.913488 -17.85856
ENSMUST00000112384.10 Rps24-201 Rps24 20088 4.124033 13.39067
ENSMUST00000182886.8 Sec31a-210 Sec31a 69162 7.691280 14.68359
ENSMUST00000238988.2 Trak1-215 Trak1 67095 -6.389912 -14.97343
ENSMUST00000115123.9 Tnk2-204 Tnk2 51789 -6.480377 -11.56945
ENSMUST00000016631.14 Ppfibp1-201 Ppfibp1 67533 2.427456 14.72839
ENSMUST00000151602.4 Mical3-206 Mical3 194401 -3.236574 -11.39845
ENSMUST00000080933.13 Map3k7-202 Map3k7 26409 -2.728790 -14.30489
ENSMUST00000108325.9 Cp-203 Cp 12870 8.427299 13.29319
P.Value FDR
ENSMUST00000113696.8 5.097375e-33 1.166636e-28
ENSMUST00000097786.10 1.716932e-26 1.316074e-22
ENSMUST00000116371.8 1.725094e-26 1.316074e-22
ENSMUST00000113705.8 8.722972e-25 4.021458e-21
ENSMUST00000153136.2 8.785463e-25 4.021458e-21
ENSMUST00000223999.2 8.511164e-24 3.246583e-20
ENSMUST00000092887.11 1.369562e-23 4.477880e-20
ENSMUST00000050905.16 1.731103e-23 4.952470e-20
ENSMUST00000182812.8 2.285726e-23 5.812601e-20
ENSMUST00000101454.9 6.726396e-22 1.539470e-18
ENSMUST00000111623.9 2.871907e-21 5.975394e-18
ENSMUST00000106460.9 9.647834e-21 1.840083e-17
ENSMUST00000112384.10 5.560696e-20 9.789819e-17
ENSMUST00000182886.8 1.988681e-19 3.235780e-16
ENSMUST00000238988.2 2.120711e-19 3.235780e-16
ENSMUST00000115123.9 2.582983e-18 3.694796e-15
ENSMUST00000016631.14 4.420876e-18 5.951800e-15
ENSMUST00000151602.4 3.581388e-17 4.553735e-14
ENSMUST00000080933.13 6.851047e-17 8.252628e-14
ENSMUST00000108325.9 7.463886e-17 8.541298e-14table(out.transcript.LPvsBa$FDR < 0.05)
FALSE TRUE
20409 2478 out.transcript.LPvsBa[out.transcript.LPvsBa$GeneSymbol %in% c('Foxp1','Ezh2','Asap1'),] Length EffectiveLength Overdispersion
ENSMUST00000175838.2 420 163.758 1.159129
ENSMUST00000177229.8 1848 1527.417 16.866677
ENSMUST00000177083.8 4415 4094.417 13.839170
ENSMUST00000110115.9 4535 4214.417 18.135542
ENSMUST00000124058.8 1213 892.417 3.563683
ENSMUST00000081721.13 2787 2466.417 6.653742
ENSMUST00000154163.9 853 533.020 1.566080
ENSMUST00000092648.13 2115 1794.417 4.728582
ENSMUST00000176565.8 3727 3406.417 8.946068
ENSMUST00000177371.8 6164 5843.417 3.344945
ENSMUST00000177410.2 618 309.424 1.435156
ENSMUST00000114618.8 2775 2454.417 9.490129
ENSMUST00000177437.8 2468 2147.417 5.693538
ENSMUST00000177307.8 2121 1800.417 6.298976
ENSMUST00000113322.9 7177 6856.417 5.676970
ENSMUST00000177227.8 967 646.467 1.373630
ENSMUST00000113321.7 1176 855.417 1.235459
ENSMUST00000175799.8 4237 3916.417 12.753683
ENSMUST00000170327.3 834 514.204 1.613701
TranscriptID GeneID
ENSMUST00000175838.2 ENSMUST00000175838.2 ENSMUSG00000030067.18
ENSMUST00000177229.8 ENSMUST00000177229.8 ENSMUSG00000030067.18
ENSMUST00000177083.8 ENSMUST00000177083.8 ENSMUSG00000022377.17
ENSMUST00000110115.9 ENSMUST00000110115.9 ENSMUSG00000022377.17
ENSMUST00000124058.8 ENSMUST00000124058.8 ENSMUSG00000030067.18
ENSMUST00000081721.13 ENSMUST00000081721.13 ENSMUSG00000029687.17
ENSMUST00000154163.9 ENSMUST00000154163.9 ENSMUSG00000030067.18
ENSMUST00000092648.13 ENSMUST00000092648.13 ENSMUSG00000029687.17
ENSMUST00000176565.8 ENSMUST00000176565.8 ENSMUSG00000030067.18
ENSMUST00000177371.8 ENSMUST00000177371.8 ENSMUSG00000022377.17
ENSMUST00000177410.2 ENSMUST00000177410.2 ENSMUSG00000030067.18
ENSMUST00000114618.8 ENSMUST00000114618.8 ENSMUSG00000029687.17
ENSMUST00000177437.8 ENSMUST00000177437.8 ENSMUSG00000030067.18
ENSMUST00000177307.8 ENSMUST00000177307.8 ENSMUSG00000030067.18
ENSMUST00000113322.9 ENSMUST00000113322.9 ENSMUSG00000030067.18
ENSMUST00000177227.8 ENSMUST00000177227.8 ENSMUSG00000030067.18
ENSMUST00000113321.7 ENSMUST00000113321.7 ENSMUSG00000030067.18
ENSMUST00000175799.8 ENSMUST00000175799.8 ENSMUSG00000022377.17
ENSMUST00000170327.3 ENSMUST00000170327.3 ENSMUSG00000029687.17
TranscriptSymbol GeneSymbol EntrezID logFC
ENSMUST00000175838.2 Foxp1-216 Foxp1 108655 2.34251465
ENSMUST00000177229.8 Foxp1-225 Foxp1 108655 -4.98978294
ENSMUST00000177083.8 Asap1-216 Asap1 13196 1.79727237
ENSMUST00000110115.9 Asap1-203 Asap1 13196 -2.48945656
ENSMUST00000124058.8 Foxp1-210 Foxp1 108655 -2.24576581
ENSMUST00000081721.13 Ezh2-201 Ezh2 14056 -0.75797128
ENSMUST00000154163.9 Foxp1-214 Foxp1 108655 0.77794988
ENSMUST00000092648.13 Ezh2-202 Ezh2 14056 0.59402664
ENSMUST00000176565.8 Foxp1-220 Foxp1 108655 0.66360968
ENSMUST00000177371.8 Asap1-217 Asap1 13196 -0.43202853
ENSMUST00000177410.2 Foxp1-229 Foxp1 108655 -0.59259459
ENSMUST00000114618.8 Ezh2-204 Ezh2 14056 0.37909712
ENSMUST00000177437.8 Foxp1-230 Foxp1 108655 -0.22543910
ENSMUST00000177307.8 Foxp1-228 Foxp1 108655 0.27214486
ENSMUST00000113322.9 Foxp1-204 Foxp1 108655 -0.14755876
ENSMUST00000177227.8 Foxp1-224 Foxp1 108655 -0.21548349
ENSMUST00000113321.7 Foxp1-203 Foxp1 108655 -0.29769900
ENSMUST00000175799.8 Asap1-206 Asap1 13196 0.28839692
ENSMUST00000170327.3 Ezh2-211 Ezh2 14056 -0.08994926
t P.Value FDR
ENSMUST00000175838.2 7.2168036 3.491649e-10 7.264852e-08
ENSMUST00000177229.8 -6.8464304 1.737724e-09 2.924360e-07
ENSMUST00000177083.8 5.8461597 1.393955e-06 7.800355e-05
ENSMUST00000110115.9 -5.7119886 2.080546e-06 1.082215e-04
ENSMUST00000124058.8 -3.7360428 3.605017e-04 6.980374e-03
ENSMUST00000081721.13 -3.4252951 1.696166e-03 2.227118e-02
ENSMUST00000154163.9 2.8769595 5.214811e-03 4.867511e-02
ENSMUST00000092648.13 2.6647326 1.194679e-02 8.705066e-02
ENSMUST00000176565.8 2.3047633 2.392405e-02 1.386553e-01
ENSMUST00000177371.8 -1.6523316 1.077297e-01 3.518754e-01
ENSMUST00000177410.2 -1.3924629 1.678613e-01 4.482622e-01
ENSMUST00000114618.8 1.3918968 1.735053e-01 4.569113e-01
ENSMUST00000177437.8 -1.3275143 1.883304e-01 4.774180e-01
ENSMUST00000177307.8 1.2858646 2.024119e-01 4.940746e-01
ENSMUST00000113322.9 -0.9334122 3.535766e-01 6.568431e-01
ENSMUST00000177227.8 -0.8974528 3.723243e-01 6.748010e-01
ENSMUST00000113321.7 -0.8811142 3.810466e-01 6.813697e-01
ENSMUST00000175799.8 0.8257307 4.147483e-01 7.097249e-01
ENSMUST00000170327.3 -0.3597590 7.213790e-01 8.894186e-01gene.unique <- out.gene.LPvsBa[out.gene.LPvsBa$FDR < 0.05,'GeneSymbol']
tx.unique <-
out.transcript.LPvsBa[out.transcript.LPvsBa$FDR < 0.05 &
out.transcript.LPvsBa$GeneSymbol %in% gene.unique,"GeneSymbol"]
table(table(tx.unique))
1 2 3 4 5 6 7 8 9 14
356 671 92 24 17 10 2 2 2 1 x <- as.data.table(out.transcript.LPvsBa[out.transcript.LPvsBa$FDR < 0.05 &
out.transcript.LPvsBa$GeneSymbol %in% gene.unique,])
x.sign <- x[GeneSymbol %in% names(table(tx.unique)[table(tx.unique) == 2]),][,.(min = sign(min(logFC)),max = sign(max(logFC))),by = 'GeneSymbol']
x.sign[,table(min,max)] max
min -1 1
-1 8 656
1 0 7out.gene.LPvsBa[out.gene.LPvsBa$GeneSymbol %in% c('Eya2'),] GeneID GeneSymbol EntrezID NExons F
ENSMUSG00000017897.19 ENSMUSG00000017897.19 Eya2 14049 4 97.52174
P.Value FDR
ENSMUSG00000017897.19 3.306905e-16 1.389819e-13out.transcript.LPvsBa[out.transcript.LPvsBa$GeneSymbol %in% c('Eya2'),] Length EffectiveLength Overdispersion
ENSMUST00000063433.8 2460 2139.417 4.602893
ENSMUST00000088132.13 2447 2126.417 5.137227
ENSMUST00000150638.8 679 364.332 3.585353
ENSMUST00000150669.2 959 638.483 1.635973
TranscriptID GeneID
ENSMUST00000063433.8 ENSMUST00000063433.8 ENSMUSG00000017897.19
ENSMUST00000088132.13 ENSMUST00000088132.13 ENSMUSG00000017897.19
ENSMUST00000150638.8 ENSMUST00000150638.8 ENSMUSG00000017897.19
ENSMUST00000150669.2 ENSMUST00000150669.2 ENSMUSG00000017897.19
TranscriptSymbol GeneSymbol EntrezID logFC
ENSMUST00000063433.8 Eya2-201 Eya2 14049 5.12190973
ENSMUST00000088132.13 Eya2-202 Eya2 14049 -5.05849875
ENSMUST00000150638.8 Eya2-205 Eya2 14049 -0.54906758
ENSMUST00000150669.2 Eya2-206 Eya2 14049 0.09507216
t P.Value FDR
ENSMUST00000063433.8 15.0337548 4.429212e-16 6.335710e-13
ENSMUST00000088132.13 -14.2480934 1.993491e-15 2.172620e-12
ENSMUST00000150638.8 -1.2311373 2.272202e-01 5.255040e-01
ENSMUST00000150669.2 0.2523196 8.024035e-01 9.303246e-01go <- goana(de = as.character(out.gene.LPvsBa[out.gene.LPvsBa$FDR < 0.05,"EntrezID"]),species = "Mm")
top.go <- topGO(go,number = Inf,ontology = "BP")
head(top.go,20) Term Ont N DE
GO:0009987 cellular process BP 17623 1092
GO:0016043 cellular component organization BP 6506 562
GO:0071840 cellular component organization or biogenesis BP 6717 573
GO:0065007 biological regulation BP 12967 847
GO:0008152 metabolic process BP 11602 781
GO:0050789 regulation of biological process BP 12570 820
GO:0050794 regulation of cellular process BP 11938 793
GO:0044238 primary metabolic process BP 10134 706
GO:0051179 localization BP 5383 466
GO:0044237 cellular metabolic process BP 10118 701
GO:0051641 cellular localization BP 3560 358
GO:0006807 nitrogen compound metabolic process BP 9563 673
GO:0071704 organic substance metabolic process BP 11172 742
GO:0006996 organelle organization BP 3584 355
GO:0048518 positive regulation of biological process BP 6679 526
GO:0048522 positive regulation of cellular process BP 6141 495
GO:0051128 regulation of cellular component organization BP 2632 286
GO:0032502 developmental process BP 6904 520
GO:0048856 anatomical structure development BP 6478 496
GO:0048523 negative regulation of cellular process BP 5339 433
P.DE
GO:0009987 2.060894e-110
GO:0016043 1.538853e-77
GO:0071840 1.880709e-77
GO:0065007 7.639252e-70
GO:0008152 3.071768e-66
GO:0050789 3.371151e-65
GO:0050794 3.817895e-65
GO:0044238 4.059325e-62
GO:0051179 7.159048e-61
GO:0044237 2.472169e-60
GO:0051641 6.215446e-60
GO:0006807 2.504505e-59
GO:0071704 9.401887e-58
GO:0006996 1.062420e-57
GO:0048518 7.026043e-57
GO:0048522 1.229316e-55
GO:0051128 2.523879e-53
GO:0032502 1.018889e-49
GO:0048856 7.848785e-49
GO:0048523 1.272948e-47Plots
bcv <- estimateDisp(dge.filtr,design = design)
fig.bcv <- wrap_elements(full = ~ foo.bcv(bcv,ylim = c(0,3)))
file.bcv <- tempfile("bcv",fileext = '.png')
png(file.bcv,width = 4,height = 3,units = 'in',res = 300)
par(mar = c(3, 3, 2, 0.25),mgp = c(2,1,0))
fig.bcv
dev.off()png
2 fig.bcv <- readPNG(file.bcv, native = TRUE)
foo.bcv(bcv,ylim = c(0,3))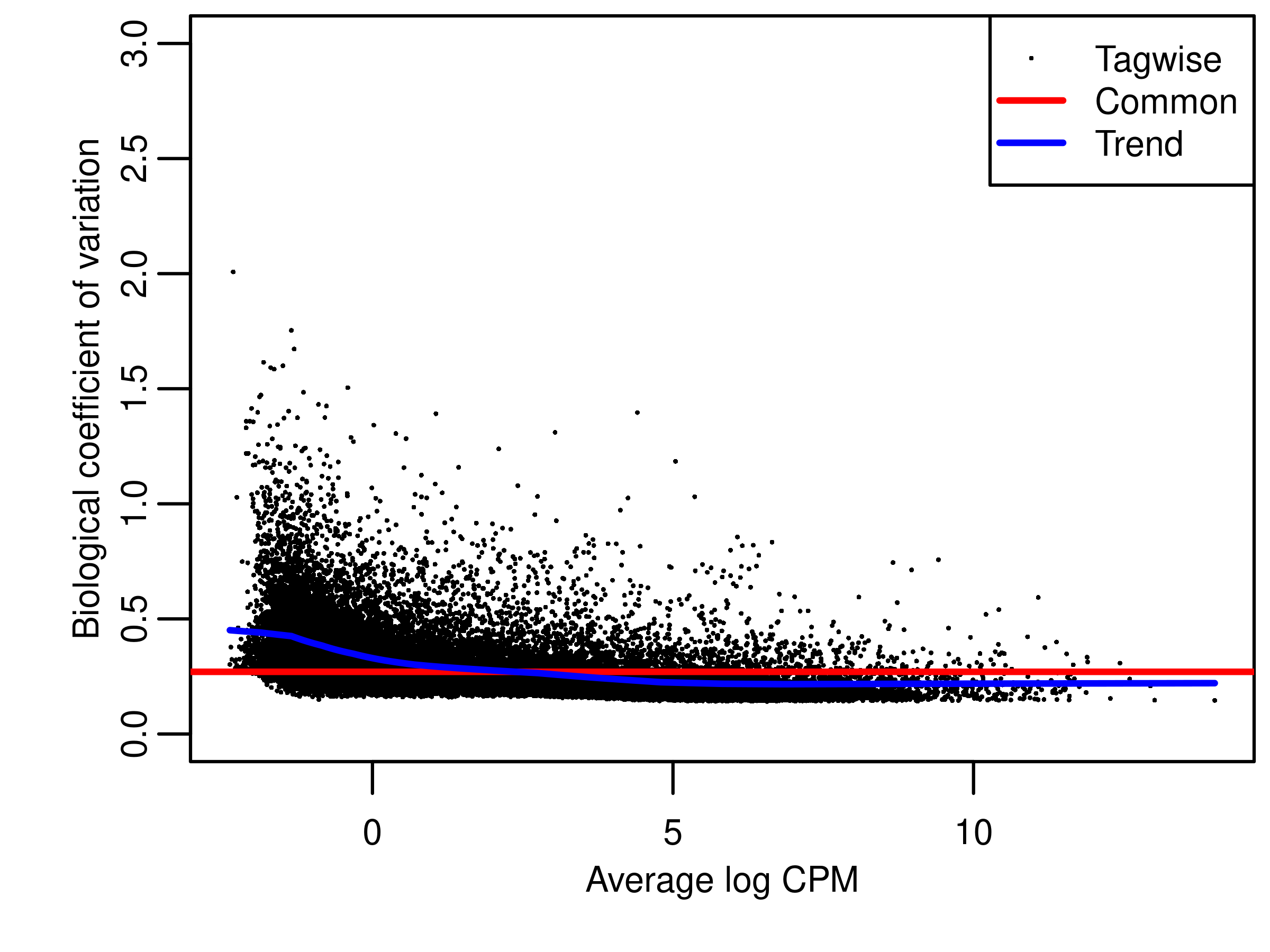
bcv.raw <- estimateDisp(dge.raw.filtr,design = design)
fig.bcv.raw <- wrap_elements(full = ~ foo.bcv(bcv.raw,ylim = c(0,3)))
file.bcv.raw <- tempfile("bcvraw",fileext = '.png')
png(file.bcv.raw,width = 4,height = 3,units = 'in',res = 300)
par(mar = c(3, 3, 2, 0.25),mgp = c(2,1,0))
fig.bcv.raw
dev.off()png
2 fig.bcv.raw <- readPNG(file.bcv.raw, native = TRUE)
foo.bcv(bcv.raw,ylim = c(0,3))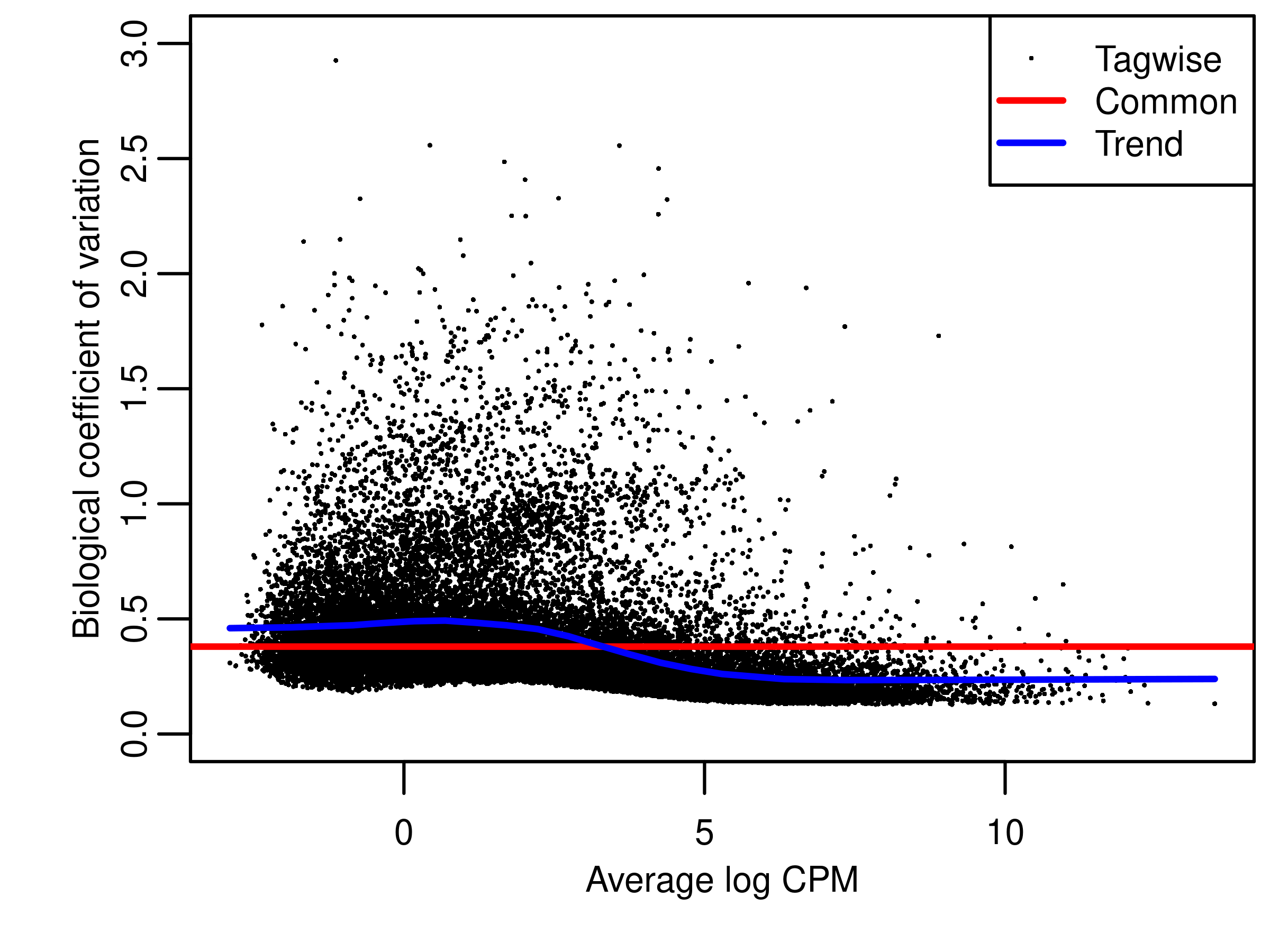
fig.splice <-
wrap_elements(full = ~ plotSplice2(ds.LPvsBa,geneid = 'Foxp1',
genecolname = 'GeneSymbol',
coef = '-1*Basal 1*LP',
exonlabel = 'TranscriptSymbol',
ylim = c(-6,4)))
file.splice <- tempfile("splice",fileext = '.png')
png(file.splice,width = 4,height = 3,units = 'in',res = 300)
par(mar = c(4, 3, 2, 0.25),mgp = c(2,1,0))
fig.splice
dev.off()png
2 fig.splice <- readPNG(file.splice, native = TRUE)
plotSplice2(ds.LPvsBa,geneid = 'Foxp1',
genecolname = 'GeneSymbol',
coef = '-1*Basal 1*LP',
exonlabel = 'TranscriptSymbol',
ylim = c(-6,4))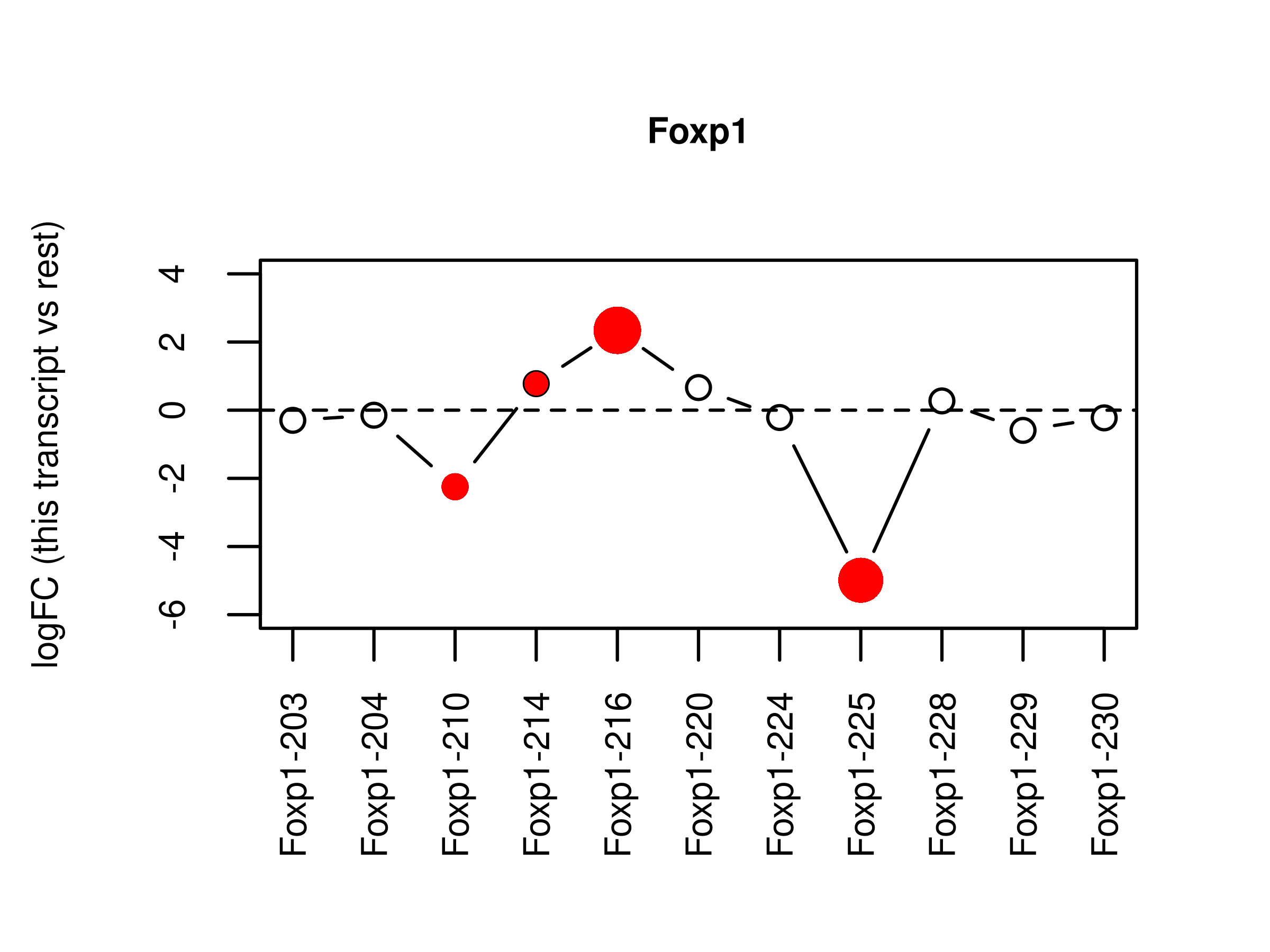
file.barplot <- tempfile("barplot",fileext = '.png')
png(file.barplot,width = 4,height = 3,units = 'in',res = 300)
plot.barplot(top.go,top = 10)
dev.off()png
2 fig.barplot <- readPNG(file.barplot, native = TRUE)
plot.barplot(top.go,top = 10)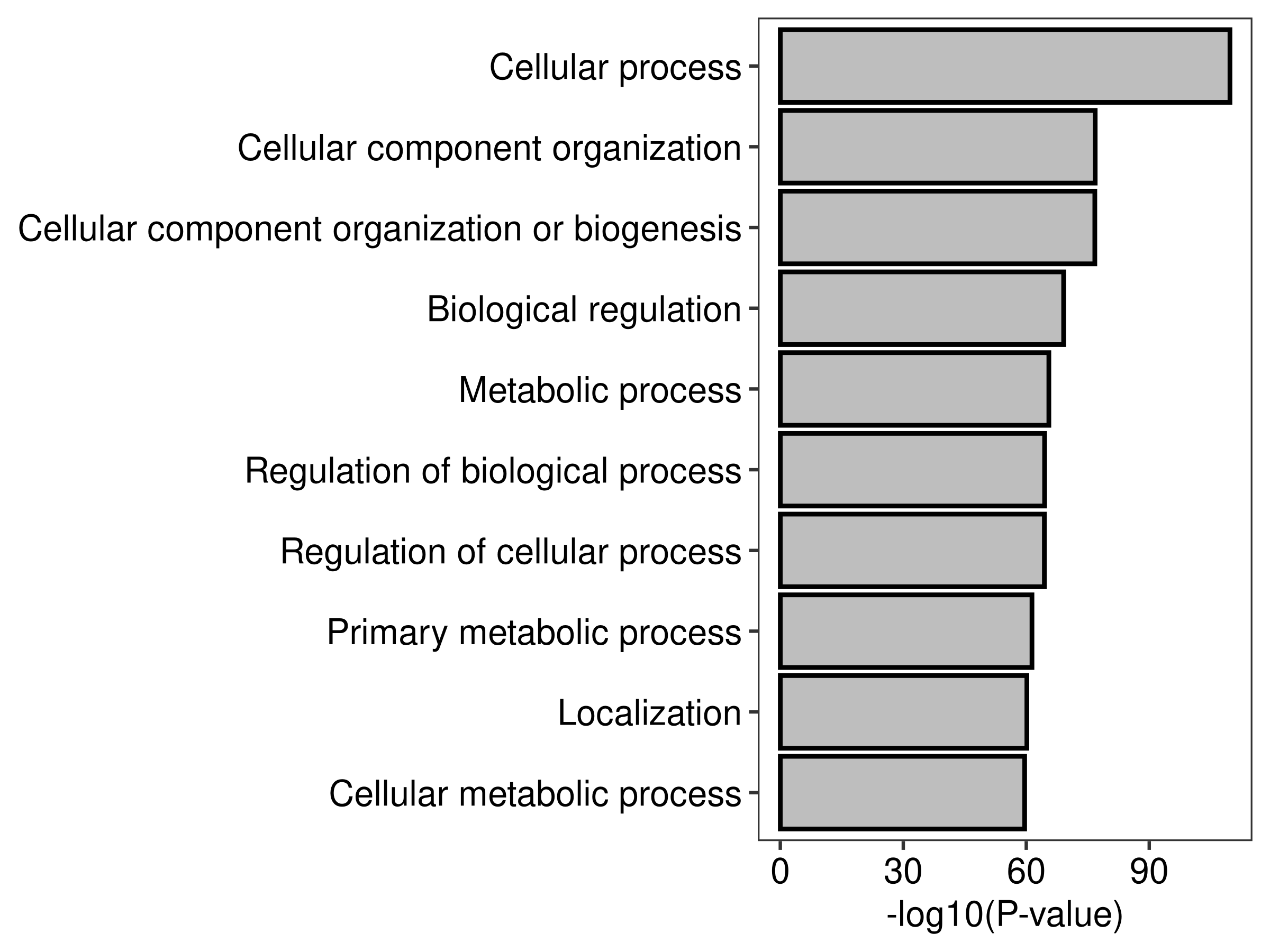
BAM <- file.path(path.bam,paste0(dge.filtr$samples$sample,'.bam'))
BW <- file.path(path.bw,paste0(dge.filtr$samples$sample,'.bw'))
txdb <- makeTxDbFromGRanges(gtf[(gtf$transcript_id %in% gtf.tx$transcript_id) &
(gtf$transcript_id %in% ds.LPvsBa$genes$TranscriptID)])Warning in call_fun_in_txdbmaker("makeTxDbFromGRanges", ...): makeTxDbFromGRanges() has moved to the txdbmaker package. Please call
txdbmaker::makeTxDbFromGRanges() to get rid of this warning.Warning in .get_cds_IDX(mcols0$type, mcols0$phase): The "phase" metadata column contains non-NA values for features of type
stop_codon. This information was ignored.geneTrack <-
GeneRegionTrack(txdb,showId=TRUE,
just.group = 'right',
background.title = "white",
min.height=10,
fill = 'grey',
name = 'Transcripts',
col.title = 'black',
fontcolor.group = 'black')
ranges(geneTrack)$symbol <-
gtf.tx$transcript_name[match(ranges(geneTrack)$symbol,gtf.tx$transcript_id)]
param <- readParam(pe = 'both',restrict = paste0("chr", c(1:19, "X", "Y")))
geneTrack.left <- geneTrack
geneTrack.left@dp@pars$just.group <- 'left'
Eya2 <- tempfile("Eya2",fileext = 'png')
png(Eya2,width = 4,height = 6,res = 300,units = 'in')
plotCoverage(gr = GRanges('chr2',IRanges(165415686,165545256)),
x = BAM,
fontsize = 8,
lib.sizes = dge.filtr$samples$lib.size*dge.filtr$samples$norm.factors,
param = param,
anno = geneTrack.left,
fill = c(rep('#df5b6d',3),rep('#5ece5a',3),rep("#2f95e2",3)),
ylim = c(-0.75,6.75),
yTicksAt = c(0,3,6),
labels = with(dge.filtr$samples,paste(group,pool,sep = '.')))
dev.off()png
2 fig.Eya2 <- readPNG(Eya2, native = TRUE)
plotCoverage(gr = GRanges('chr2',IRanges(165415686,165545256)),
x = BAM,
fontsize = 8,
lib.sizes = dge.filtr$samples$lib.size*dge.filtr$samples$norm.factors,
param = param,
anno = geneTrack.left,
fill = c(rep('#df5b6d',3),rep('#5ece5a',3),rep("#2f95e2",3)),
ylim = c(-0.75,6.75),
yTicksAt = c(0,3,6),
labels = with(dge.filtr$samples,paste(group,pool,sep = '.')))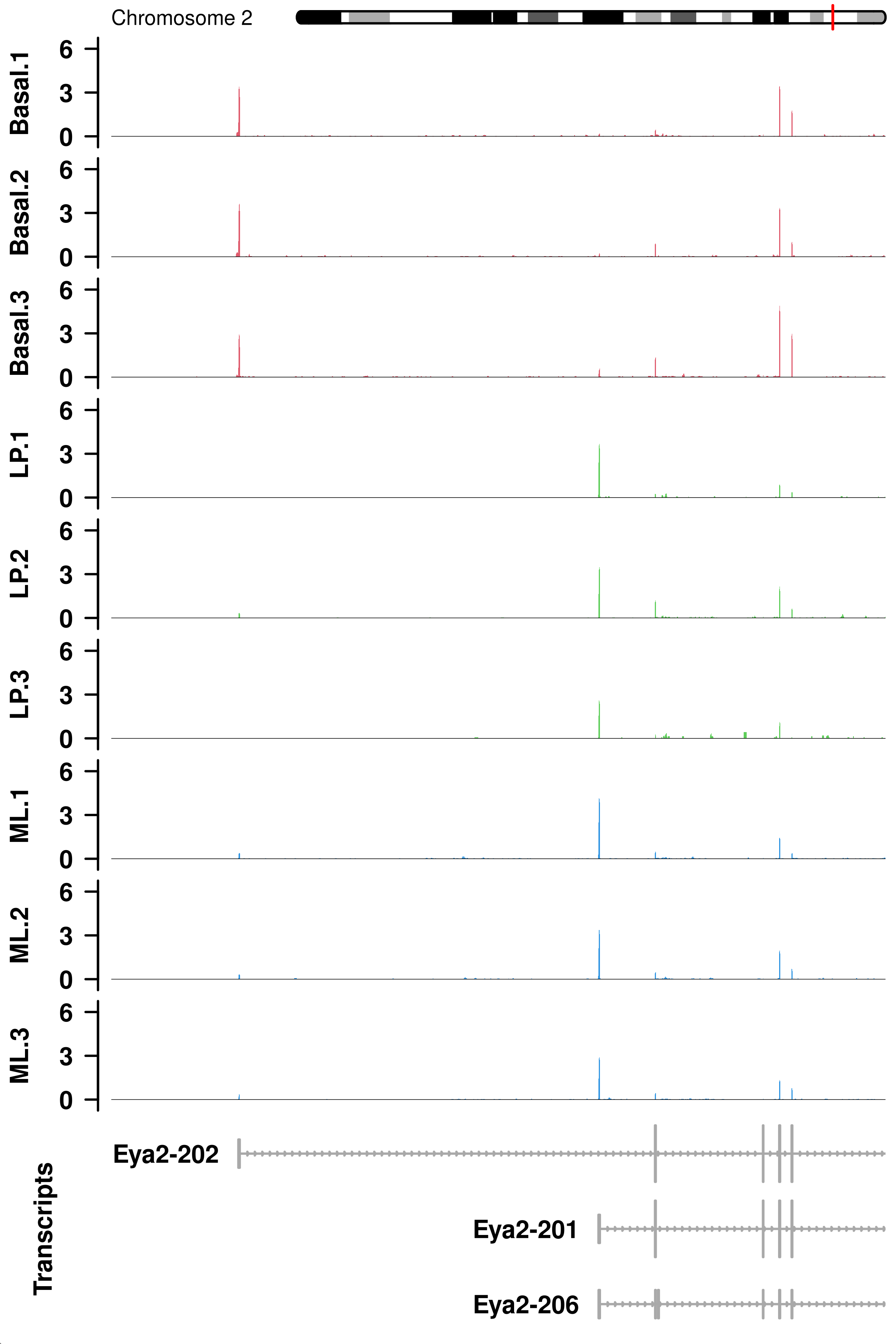
Output files
fig.ds <- wrap_plots(A = wrap_elements(fig.bcv.raw),
B = wrap_elements(fig.bcv),
C = wrap_elements(fig.barplot),
D = wrap_elements(fig.splice),
E = wrap_elements(fig.Eya2),
design = c(area(1,1),area(1,2),
area(2,1),area(3,1),area(2,2,3,2)),
heights = c(3,3,3)/8) +
plot_annotation(tag_levels = 'a',
theme = theme(plot.tag = element_text(size = 8)))
fig.ds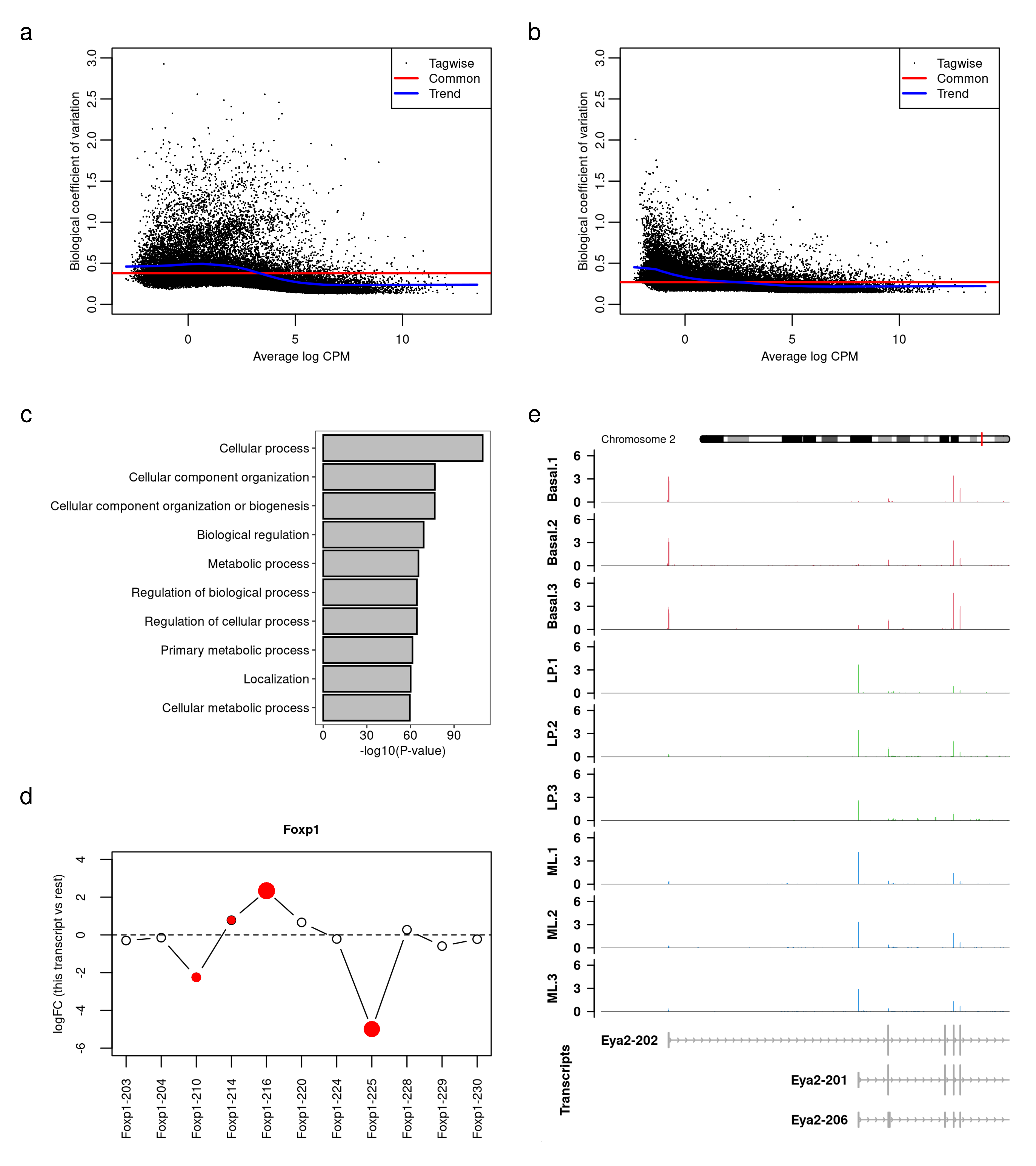
ggsave(plot = fig.ds,
filename = file.path(path.misc,'Figure-CaseStudy.pdf'),
device = 'pdf',width = 8,height = 9,units = 'in',dpi = 300)
sessionInfo()R version 4.4.1 (2024-06-14)
Platform: x86_64-pc-linux-gnu
Running under: Red Hat Enterprise Linux 9.3 (Plow)
Matrix products: default
BLAS: /stornext/System/data/software/rhel/9/base/tools/R/4.4.1/lib64/R/lib/libRblas.so
LAPACK: /stornext/System/data/software/rhel/9/base/tools/R/4.4.1/lib64/R/lib/libRlapack.so; LAPACK version 3.12.0
locale:
[1] LC_CTYPE=en_US.UTF-8 LC_NUMERIC=C
[3] LC_TIME=en_US.UTF-8 LC_COLLATE=en_US.UTF-8
[5] LC_MONETARY=en_US.UTF-8 LC_MESSAGES=en_US.UTF-8
[7] LC_PAPER=en_US.UTF-8 LC_NAME=C
[9] LC_ADDRESS=C LC_TELEPHONE=C
[11] LC_MEASUREMENT=en_US.UTF-8 LC_IDENTIFICATION=C
time zone: Australia/Melbourne
tzcode source: system (glibc)
attached base packages:
[1] grid stats4 stats graphics grDevices datasets utils
[8] methods base
other attached packages:
[1] patchwork_1.3.0
[2] png_0.1-8
[3] stringr_1.5.1
[4] ggplot2_3.5.1
[5] BiocParallel_1.38.0
[6] csaw_1.38.0
[7] SummarizedExperiment_1.34.0
[8] MatrixGenerics_1.16.0
[9] matrixStats_1.5.0
[10] TxDb.Mmusculus.UCSC.mm39.refGene_3.19.0
[11] Gviz_1.48.0
[12] data.table_1.17.0
[13] GenomicFeatures_1.56.0
[14] AnnotationDbi_1.66.0
[15] Biobase_2.64.0
[16] Rsubread_2.18.0
[17] rtracklayer_1.64.0
[18] GenomicRanges_1.56.2
[19] GenomeInfoDb_1.40.1
[20] IRanges_2.38.1
[21] S4Vectors_0.42.1
[22] BiocGenerics_0.50.0
[23] edgeR_4.5.9
[24] limma_3.63.9
[25] workflowr_1.7.1
loaded via a namespace (and not attached):
[1] later_1.4.1 BiocIO_1.14.0 bitops_1.0-9
[4] filelock_1.0.3 tibble_3.2.1 R.oo_1.27.0
[7] XML_3.99-0.18 rpart_4.1.23 lifecycle_1.0.4
[10] httr2_1.1.0 rprojroot_2.0.4 processx_3.8.6
[13] lattice_0.22-6 vroom_1.6.5 ensembldb_2.28.1
[16] backports_1.5.0 magrittr_2.0.3 Hmisc_5.2-2
[19] sass_0.4.9 rmarkdown_2.29 jquerylib_0.1.4
[22] yaml_2.3.10 metapod_1.12.0 httpuv_1.6.15
[25] DBI_1.2.3 RColorBrewer_1.1-3 abind_1.4-8
[28] zlibbioc_1.50.0 R.utils_2.13.0 AnnotationFilter_1.28.0
[31] biovizBase_1.52.0 RCurl_1.98-1.16 nnet_7.3-19
[34] VariantAnnotation_1.50.0 rappdirs_0.3.3 git2r_0.35.0
[37] GenomeInfoDbData_1.2.12 codetools_0.2-20 DelayedArray_0.30.1
[40] xml2_1.3.7 tidyselect_1.2.1 UCSC.utils_1.0.0
[43] farver_2.1.2 BiocFileCache_2.12.0 base64enc_0.1-3
[46] GenomicAlignments_1.40.0 jsonlite_1.9.1 Formula_1.2-5
[49] systemfonts_1.2.1 tools_4.4.1 progress_1.2.3
[52] ragg_1.3.3 Rcpp_1.0.14 glue_1.8.0
[55] gridExtra_2.3 SparseArray_1.4.8 xfun_0.51
[58] dplyr_1.1.4 withr_3.0.2 BiocManager_1.30.25
[61] fastmap_1.2.0 latticeExtra_0.6-30 callr_3.7.6
[64] digest_0.6.37 R6_2.6.1 gridGraphics_0.5-1
[67] textshaping_1.0.0 colorspace_2.1-1 GO.db_3.19.1
[70] jpeg_0.1-10 dichromat_2.0-0.1 biomaRt_2.60.1
[73] RSQLite_2.3.9 R.methodsS3_1.8.2 generics_0.1.3
[76] renv_1.1.2 prettyunits_1.2.0 httr_1.4.7
[79] htmlwidgets_1.6.4 S4Arrays_1.4.1 org.Mm.eg.db_3.19.1
[82] whisker_0.4.1 pkgconfig_2.0.3 gtable_0.3.6
[85] blob_1.2.4 XVector_0.44.0 htmltools_0.5.8.1
[88] ProtGenerics_1.36.0 scales_1.3.0 knitr_1.49
[91] rstudioapi_0.17.1 tzdb_0.4.0 rjson_0.2.23
[94] checkmate_2.3.2 curl_6.2.1 cachem_1.1.0
[97] parallel_4.4.1 foreign_0.8-86 restfulr_0.0.15
[100] pillar_1.10.1 vctrs_0.6.5 promises_1.3.2
[103] dbplyr_2.5.0 cluster_2.1.6 htmlTable_2.4.3
[106] evaluate_1.0.3 readr_2.1.5 cli_3.6.4
[109] locfit_1.5-9.12 compiler_4.4.1 Rsamtools_2.20.0
[112] rlang_1.1.5 crayon_1.5.3 labeling_0.4.3
[115] interp_1.1-6 ps_1.9.0 getPass_0.2-4
[118] fs_1.6.5 stringi_1.8.4 deldir_2.0-4
[121] txdbmaker_1.0.1 munsell_0.5.1 Biostrings_2.72.1
[124] lazyeval_0.2.2 Matrix_1.7-0 BSgenome_1.72.0
[127] hms_1.1.3 bit64_4.6.0-1 KEGGREST_1.44.1
[130] statmod_1.5.0 memoise_2.0.1 bslib_0.9.0
[133] bit_4.6.0