SCZ - Brain Caudate basal ganglia
sheng Qian
2021-2-6
Last updated: 2022-03-16
Checks: 5 2
Knit directory: cTWAS_analysis/
This reproducible R Markdown analysis was created with workflowr (version 1.7.0). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
The R Markdown is untracked by Git. To know which version of the R Markdown file created these results, you’ll want to first commit it to the Git repo. If you’re still working on the analysis, you can ignore this warning. When you’re finished, you can run wflow_publish to commit the R Markdown file and build the HTML.
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
The command set.seed(20211220) was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
Nice! There were no cached chunks for this analysis, so you can be confident that you successfully produced the results during this run.
Using absolute paths to the files within your workflowr project makes it difficult for you and others to run your code on a different machine. Change the absolute path(s) below to the suggested relative path(s) to make your code more reproducible.
| absolute | relative |
|---|---|
| /project2/xinhe/shengqian/cTWAS/cTWAS_analysis/data/ | data |
| /project2/xinhe/shengqian/cTWAS/cTWAS_analysis/code/ctwas_config.R | code/ctwas_config.R |
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility.
The results in this page were generated with repository version d57314b. See the Past versions tab to see a history of the changes made to the R Markdown and HTML files.
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can use wflow_publish or wflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Ignored files:
Ignored: .ipynb_checkpoints/
Ignored: data/AF/
Untracked files:
Untracked: Rplot.png
Untracked: analysis/.ipynb_checkpoints/
Untracked: analysis/SCZ_2020_Brain_Amygdala.Rmd
Untracked: analysis/SCZ_2020_Brain_Anterior_cingulate_cortex_BA24.Rmd
Untracked: analysis/SCZ_2020_Brain_Caudate_basal_ganglia.Rmd
Untracked: analysis/SCZ_2020_Brain_Cerebellar_Hemisphere.Rmd
Untracked: analysis/SCZ_2020_Brain_Cerebellum.Rmd
Untracked: analysis/SCZ_2020_Brain_Hippocampus.Rmd
Untracked: analysis/SCZ_2020_Brain_Nucleus_accumbens_basal_ganglia.Rmd
Untracked: analysis/SCZ_2020_Brain_Spinal_cord_cervical_c-1.Rmd
Untracked: analysis/SCZ_2020_Brain_Substantia_nigra.Rmd
Untracked: code/.ipynb_checkpoints/
Untracked: code/AF_out/
Untracked: code/Autism_out/
Untracked: code/BMI_S_out/
Untracked: code/BMI_out/
Untracked: code/Glucose_out/
Untracked: code/LDL_S_out/
Untracked: code/SCZ_2014_EUR_out/
Untracked: code/SCZ_2020_out/
Untracked: code/SCZ_S_out/
Untracked: code/SCZ_out/
Untracked: code/T2D_out/
Untracked: code/ctwas_config.R
Untracked: code/mapping.R
Untracked: code/out/
Untracked: code/run_AF_analysis.sbatch
Untracked: code/run_AF_analysis.sh
Untracked: code/run_AF_ctwas_rss_LDR.R
Untracked: code/run_Autism_analysis.sbatch
Untracked: code/run_Autism_analysis.sh
Untracked: code/run_Autism_ctwas_rss_LDR.R
Untracked: code/run_BMI_analysis.sbatch
Untracked: code/run_BMI_analysis.sh
Untracked: code/run_BMI_analysis_S.sbatch
Untracked: code/run_BMI_analysis_S.sh
Untracked: code/run_BMI_ctwas_rss_LDR.R
Untracked: code/run_BMI_ctwas_rss_LDR_S.R
Untracked: code/run_Glucose_analysis.sbatch
Untracked: code/run_Glucose_analysis.sh
Untracked: code/run_Glucose_ctwas_rss_LDR.R
Untracked: code/run_LDL_analysis_S.sbatch
Untracked: code/run_LDL_analysis_S.sh
Untracked: code/run_LDL_ctwas_rss_LDR_S.R
Untracked: code/run_SCZ_2014_EUR_analysis.sbatch
Untracked: code/run_SCZ_2014_EUR_analysis.sh
Untracked: code/run_SCZ_2014_EUR_ctwas_rss_LDR.R
Untracked: code/run_SCZ_2020_analysis.sbatch
Untracked: code/run_SCZ_2020_analysis.sh
Untracked: code/run_SCZ_2020_ctwas_rss_LDR.R
Untracked: code/run_SCZ_analysis.sbatch
Untracked: code/run_SCZ_analysis.sh
Untracked: code/run_SCZ_analysis_S.sbatch
Untracked: code/run_SCZ_analysis_S.sh
Untracked: code/run_SCZ_ctwas_rss_LDR.R
Untracked: code/run_SCZ_ctwas_rss_LDR_S.R
Untracked: code/run_T2D_analysis.sbatch
Untracked: code/run_T2D_analysis.sh
Untracked: code/run_T2D_ctwas_rss_LDR.R
Untracked: code/wflow_build.R
Untracked: code/wflow_build.sbatch
Untracked: data/.ipynb_checkpoints/
Untracked: data/BMI/
Untracked: data/PGC3_SCZ_wave3_public.v2.tsv
Untracked: data/SCZ/
Untracked: data/SCZ_2014_EUR/
Untracked: data/SCZ_2020/
Untracked: data/SCZ_S/
Untracked: data/T2D/
Untracked: data/UKBB/
Untracked: data/UKBB_SNPs_Info.text
Untracked: data/gene_OMIM.txt
Untracked: data/gene_pip_0.8.txt
Untracked: data/mashr_Heart_Atrial_Appendage.db
Untracked: data/mashr_sqtl/
Untracked: data/summary_known_genes_annotations.xlsx
Untracked: data/untitled.txt
Unstaged changes:
Modified: analysis/SCZ_2020_Brain_Cortex.Rmd
Modified: analysis/SCZ_2020_Brain_Frontal_Cortex_BA9.Rmd
Modified: analysis/SCZ_2020_Brain_Hypothalamus.Rmd
Modified: analysis/SCZ_2020_Brain_Putamen_basal_ganglia.Rmd
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
There are no past versions. Publish this analysis with wflow_publish() to start tracking its development.
Weight QC
#number of imputed weights
nrow(qclist_all)[1] 11549#number of imputed weights by chromosome
table(qclist_all$chr)
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
1113 836 688 449 569 642 534 428 425 443 690 658 238 386 385 524
17 18 19 20 21 22
714 185 888 345 129 280 #number of imputed weights without missing variants
sum(qclist_all$nmiss==0)[1] 8945#proportion of imputed weights without missing variants
mean(qclist_all$nmiss==0)[1] 0.7745Check convergence of parameters
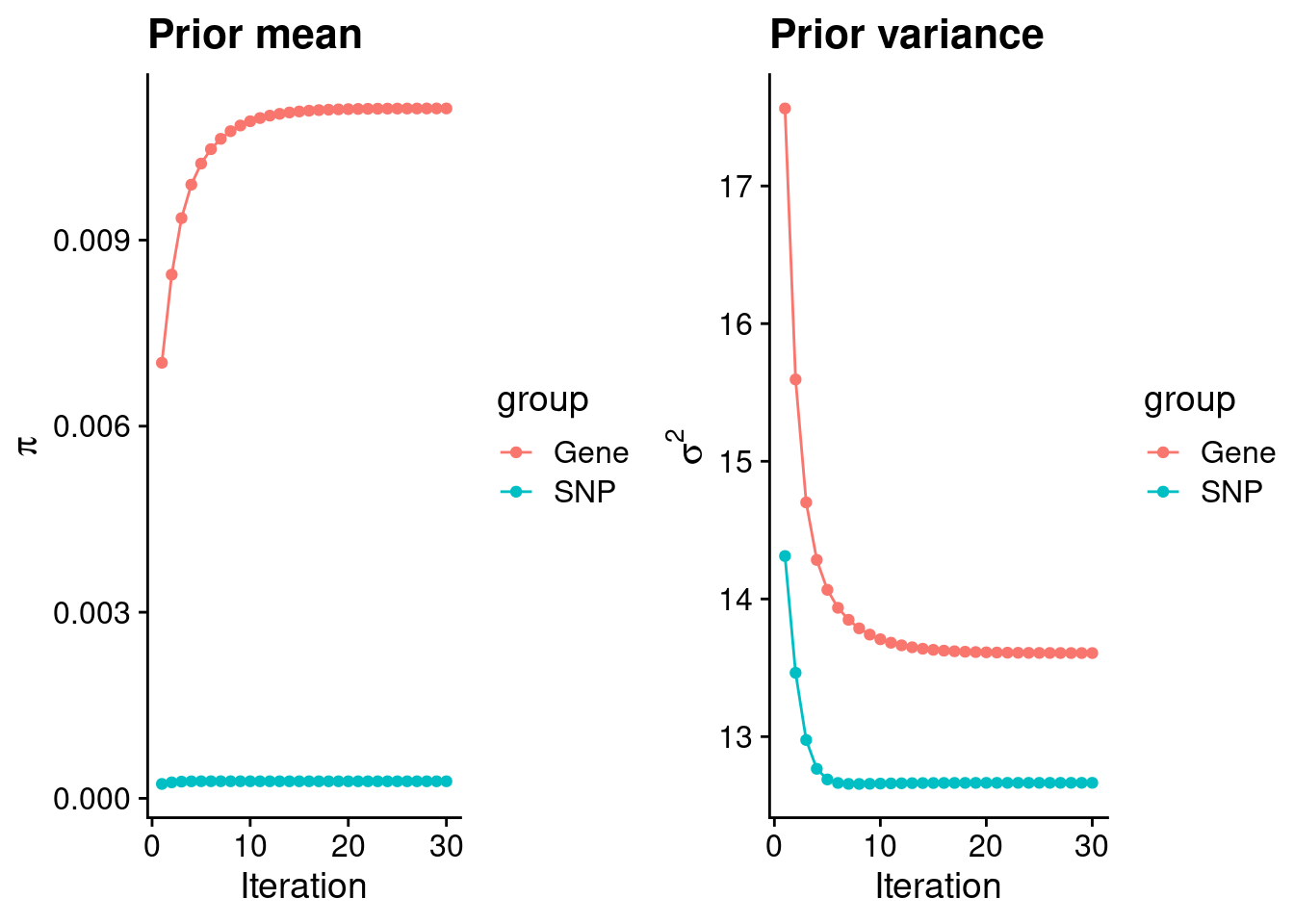
#estimated group prior
estimated_group_prior <- group_prior_rec[,ncol(group_prior_rec)]
names(estimated_group_prior) <- c("gene", "snp")
estimated_group_prior["snp"] <- estimated_group_prior["snp"]*thin #adjust parameter to account for thin argument
print(estimated_group_prior) gene snp
0.0111227 0.0002745 #estimated group prior variance
estimated_group_prior_var <- group_prior_var_rec[,ncol(group_prior_var_rec)]
names(estimated_group_prior_var) <- c("gene", "snp")
print(estimated_group_prior_var) gene snp
13.61 12.66 #report sample size
print(sample_size)[1] 161405#report group size
group_size <- c(nrow(ctwas_gene_res), n_snps)
print(group_size)[1] 11549 7394310#estimated group PVE
estimated_group_pve <- estimated_group_prior_var*estimated_group_prior*group_size/sample_size #check PVE calculation
names(estimated_group_pve) <- c("gene", "snp")
print(estimated_group_pve) gene snp
0.01083 0.15925 #compare sum(PIP*mu2/sample_size) with above PVE calculation
c(sum(ctwas_gene_res$PVE),sum(ctwas_snp_res$PVE))[1] 0.04685 0.80048Genes with highest PIPs
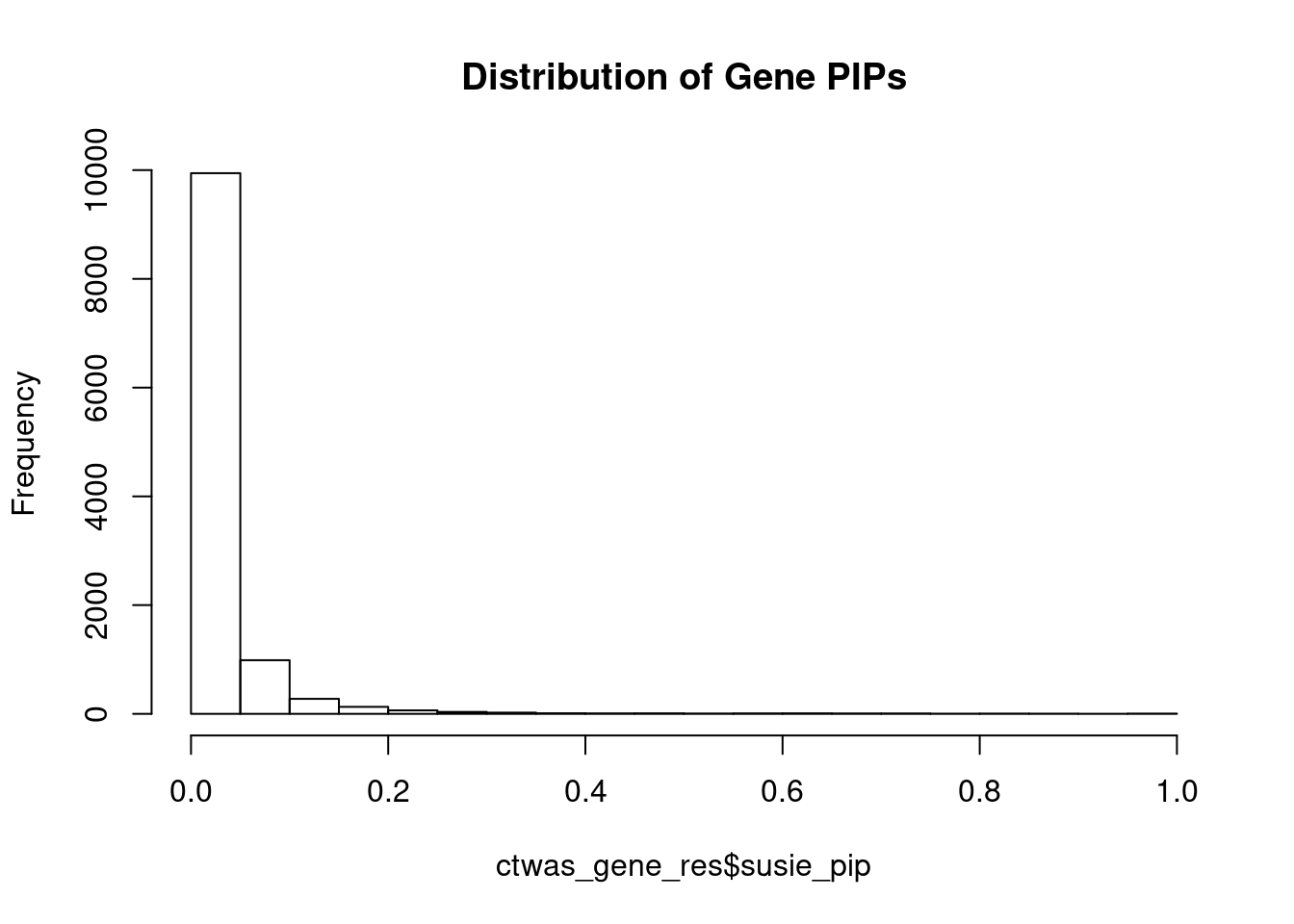
genename region_tag susie_pip mu2 PVE z num_eqtl
8043 PACSIN3 11_29 0.9920 48.73 0.0002995 7.013 2
6997 SPPL3 12_74 0.9891 34.38 0.0002107 -5.663 2
10518 MPPED1 22_19 0.9889 29.58 0.0001812 -5.318 2
11135 ZNF823 19_10 0.9774 40.41 0.0002447 6.308 2
12311 AC012074.2 2_16 0.9758 30.43 0.0001840 5.469 1
101 FARP2 2_144 0.9020 22.41 0.0001253 4.355 3
2677 TRPV4 12_66 0.8820 24.20 0.0001322 4.416 1
110 ELAC2 17_11 0.8757 24.49 0.0001329 5.507 1
12520 HLA-DMB 6_27 0.8715 79.57 0.0004296 -9.679 1
14012 MYO19 17_22 0.8505 26.46 0.0001394 -4.886 1
7145 ACE 17_37 0.8389 33.79 0.0001756 -5.876 1
6200 FAM135B 8_91 0.8356 22.25 0.0001152 -3.461 1
11142 RPL12 9_66 0.8267 23.97 0.0001228 4.663 2
7959 GTF2A1 14_39 0.8119 24.39 0.0001227 -4.850 1
5922 METTL21A 2_122 0.8042 25.32 0.0001261 -4.404 1
12134 AC008269.2 2_122 0.7729 22.36 0.0001071 4.336 1
5830 RIT1 1_76 0.7673 23.85 0.0001134 -4.023 1
11616 ITSN1 21_14 0.7568 22.63 0.0001061 4.315 2
8810 FOXN2 2_31 0.7510 26.11 0.0001215 -5.260 2
6653 SLC25A27 6_35 0.7493 25.86 0.0001200 -3.899 3Genes with largest effect sizes

genename region_tag susie_pip mu2 PVE z num_eqtl
10 SEMA3F 3_35 6.666e-02 769.81 3.179e-04 0.2075 1
38 RBM6 3_35 3.483e-01 456.56 9.852e-04 4.4688 1
9765 LSMEM2 3_35 5.565e-01 399.87 1.379e-03 4.2709 1
7736 MST1R 3_35 1.603e-05 382.04 3.794e-08 -3.4420 2
12539 NAT6 3_35 1.332e-05 360.61 2.975e-08 1.6917 3
10521 HYAL3 3_35 3.000e-05 337.77 6.278e-08 -2.5066 1
712 RASSF1 3_35 1.092e-05 322.64 2.184e-08 4.3268 1
7732 RNF123 3_35 9.944e-06 272.41 1.678e-08 -2.3622 1
130 CACNA2D2 3_35 4.435e-05 113.02 3.106e-08 -0.1392 1
10491 BTN3A2 6_20 1.445e-02 111.96 1.002e-05 9.0087 2
702 RHOA 3_35 1.187e-05 101.93 7.499e-09 -1.9997 1
1222 C3orf18 3_35 1.169e-04 99.42 7.203e-08 -0.4441 1
2990 HEMK1 3_35 1.169e-04 99.42 7.203e-08 0.4441 1
11731 CLIC1 6_26 4.557e-01 82.93 2.341e-04 10.7310 2
5119 PGBD1 6_22 2.865e-02 82.31 1.461e-05 -8.4603 1
11478 APOM 6_26 2.228e-01 81.12 1.120e-04 10.6484 1
2958 USP4 3_35 1.441e-04 80.77 7.213e-08 2.9885 2
12520 HLA-DMB 6_27 8.715e-01 79.57 4.296e-04 -9.6790 1
5990 AMT 3_35 1.828e-05 78.60 8.901e-09 -1.5571 1
12582 C4A 6_26 6.111e-02 78.41 2.968e-05 10.4799 2Genes with highest PVE
genename region_tag susie_pip mu2 PVE z num_eqtl
9765 LSMEM2 3_35 0.55648 399.87 0.0013786 4.2709 1
38 RBM6 3_35 0.34830 456.56 0.0009852 4.4688 1
12520 HLA-DMB 6_27 0.87145 79.57 0.0004296 -9.6790 1
10 SEMA3F 3_35 0.06666 769.81 0.0003179 0.2075 1
8043 PACSIN3 11_29 0.99198 48.73 0.0002995 7.0133 2
11135 ZNF823 19_10 0.97742 40.41 0.0002447 6.3077 2
7700 PBRM1 3_36 0.57790 66.13 0.0002368 -9.4285 1
11731 CLIC1 6_26 0.45566 82.93 0.0002341 10.7310 2
6997 SPPL3 12_74 0.98914 34.38 0.0002107 -5.6634 2
3091 SF3B1 2_117 0.63631 51.76 0.0002041 7.6053 1
8307 GATAD2A 19_15 0.60643 52.85 0.0001986 -7.4194 1
12090 HCG11 6_20 0.74660 40.25 0.0001862 -0.5804 1
12311 AC012074.2 2_16 0.97584 30.43 0.0001840 5.4694 1
10518 MPPED1 22_19 0.98894 29.58 0.0001812 -5.3176 2
1226 PPP1R13B 14_54 0.59527 48.93 0.0001805 7.4786 2
8639 INO80E 16_24 0.54928 51.79 0.0001762 7.5514 1
7145 ACE 17_37 0.83886 33.79 0.0001756 -5.8759 1
9836 HARBI1 11_28 0.43587 58.70 0.0001585 8.0462 1
11474 CSNK2B 6_26 0.73898 32.14 0.0001472 -6.9161 1
8241 PDIA3 15_16 0.62403 37.68 0.0001457 6.3137 1Genes with largest z scores
genename region_tag susie_pip mu2 PVE z num_eqtl
11731 CLIC1 6_26 0.4556596 82.93 2.341e-04 10.731 2
11478 APOM 6_26 0.2228140 81.12 1.120e-04 10.648 1
12582 C4A 6_26 0.0611069 78.41 2.968e-05 10.480 2
6289 CNNM2 10_66 0.1236064 41.57 3.184e-05 -9.914 2
12520 HLA-DMB 6_27 0.8714547 79.57 4.296e-04 -9.679 1
7700 PBRM1 3_36 0.5779011 66.13 2.368e-04 -9.429 1
11431 HLA-DMA 6_27 0.0804636 75.86 3.782e-05 -9.408 1
11444 PRRT1 6_26 0.0105908 56.70 3.721e-06 9.276 1
11469 MSH5 6_26 0.0057968 67.44 2.422e-06 9.136 2
7699 GNL3 3_36 0.1773205 63.53 6.980e-05 9.127 3
10491 BTN3A2 6_20 0.0144493 111.96 1.002e-05 9.009 2
6424 ABCB9 12_75 0.0006627 63.47 2.606e-07 8.638 1
9986 ARL6IP4 12_75 0.0006047 63.06 2.362e-07 8.615 1
2697 OGFOD2 12_75 0.0005826 62.95 2.272e-07 8.602 1
8450 SMIM4 3_36 0.0174944 56.68 6.144e-06 -8.494 1
5119 PGBD1 6_22 0.0286547 82.31 1.461e-05 -8.460 1
11440 AGER 6_26 0.0043172 40.24 1.076e-06 -8.380 2
9836 HARBI1 11_28 0.4358733 58.70 1.585e-04 8.046 1
2630 MDK 11_28 0.1542184 56.23 5.372e-05 -7.898 1
3012 NEK4 3_36 0.0107185 48.35 3.211e-06 7.898 1Comparing z scores and PIPs
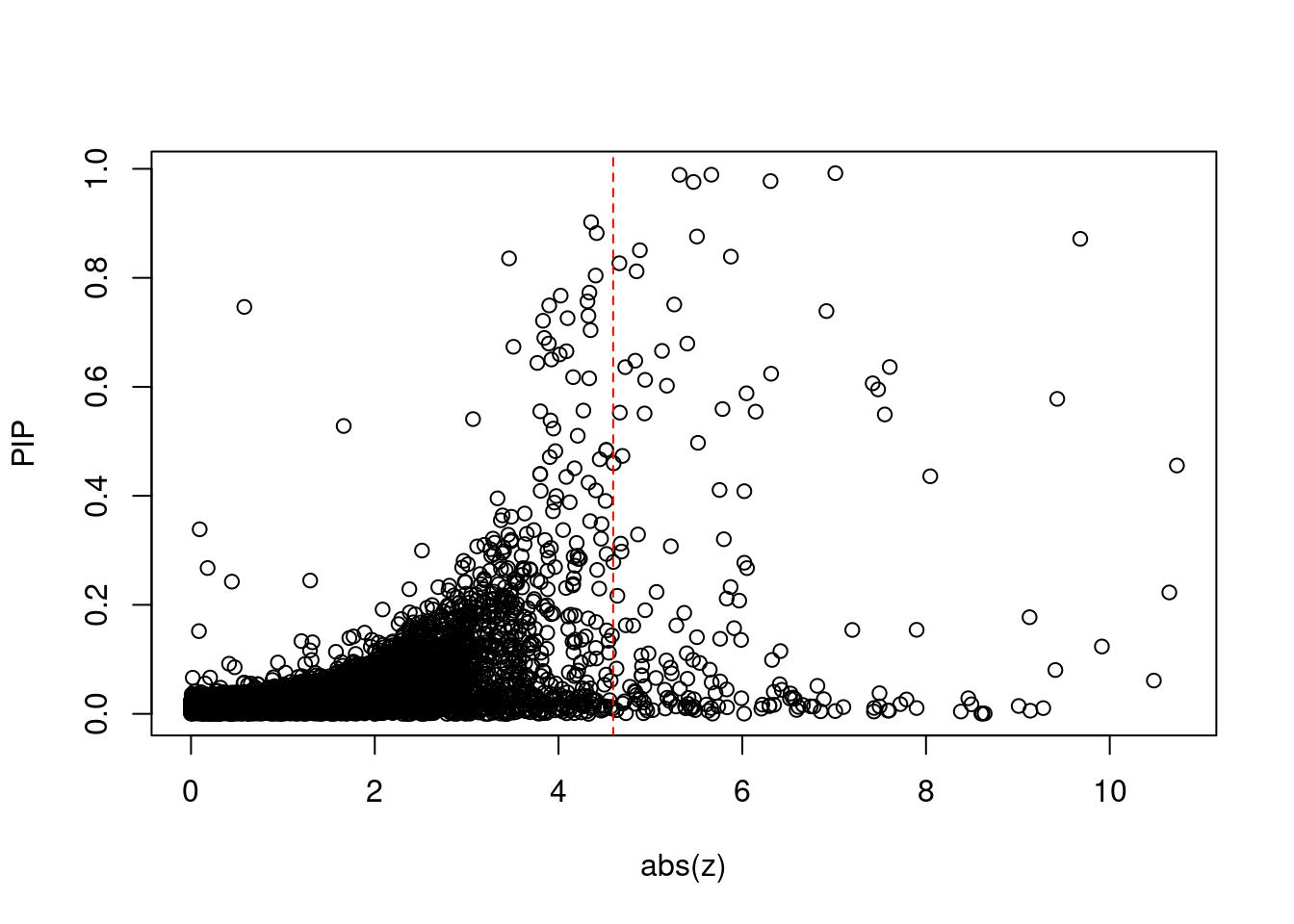
[1] 0.01455GO enrichment analysis for genes with PIP>0.5
#number of genes for gene set enrichment
length(genes)[1] 59Uploading data to Enrichr... Done.
Querying GO_Biological_Process_2021... Done.
Querying GO_Cellular_Component_2021... Done.
Querying GO_Molecular_Function_2021... Done.
Parsing results... Done.
[1] "GO_Biological_Process_2021"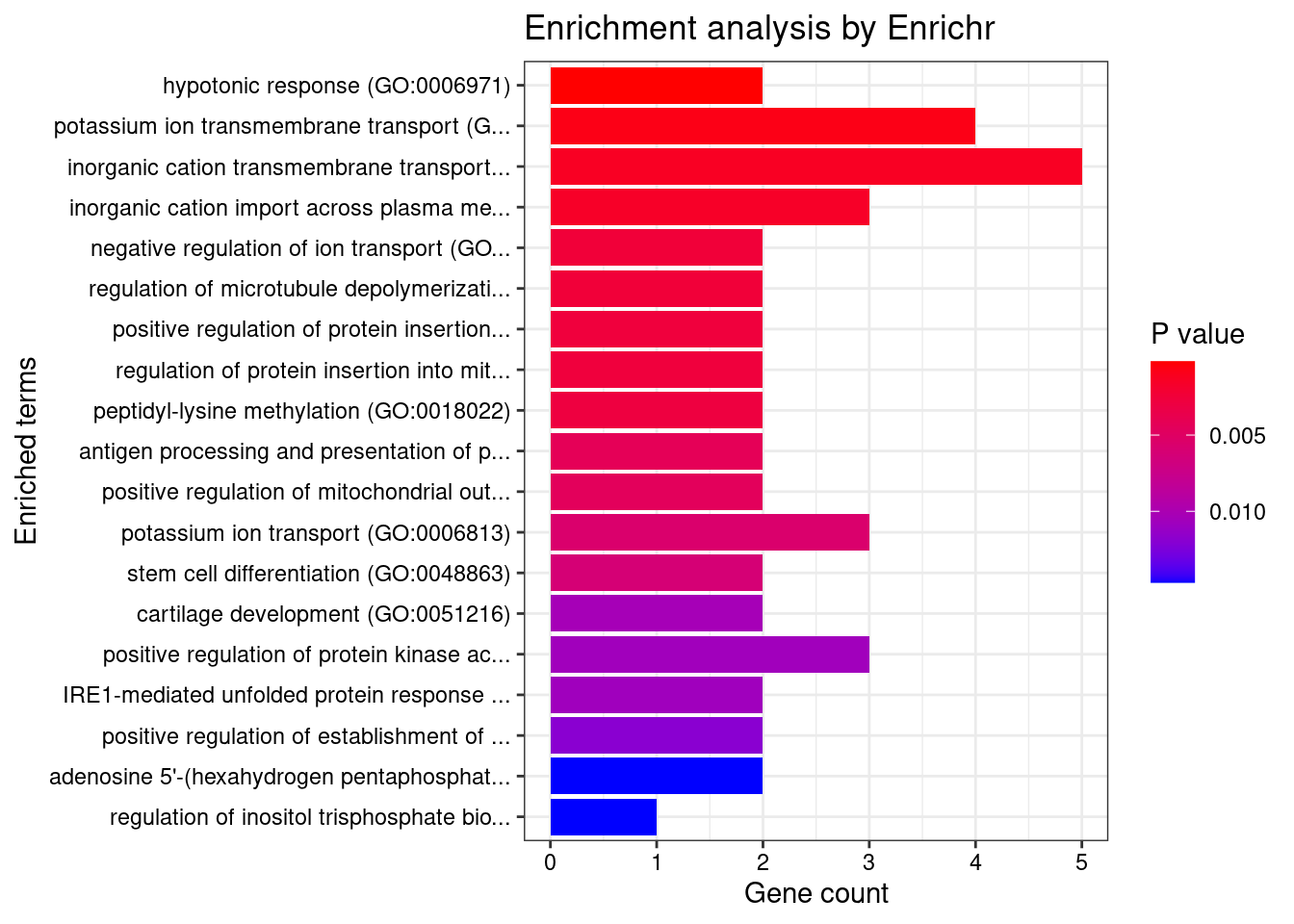
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)
[1] "GO_Cellular_Component_2021"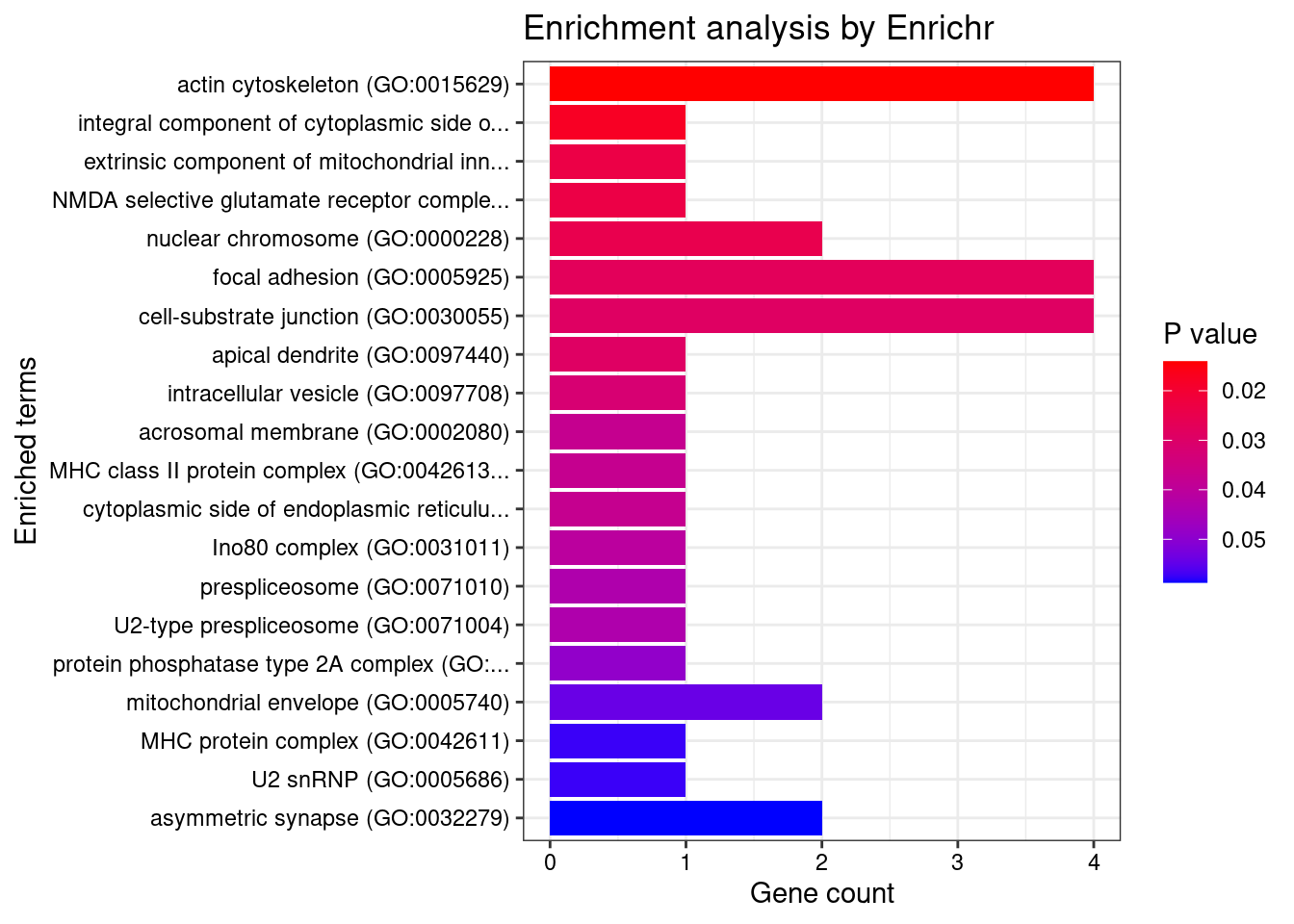
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)
[1] "GO_Molecular_Function_2021"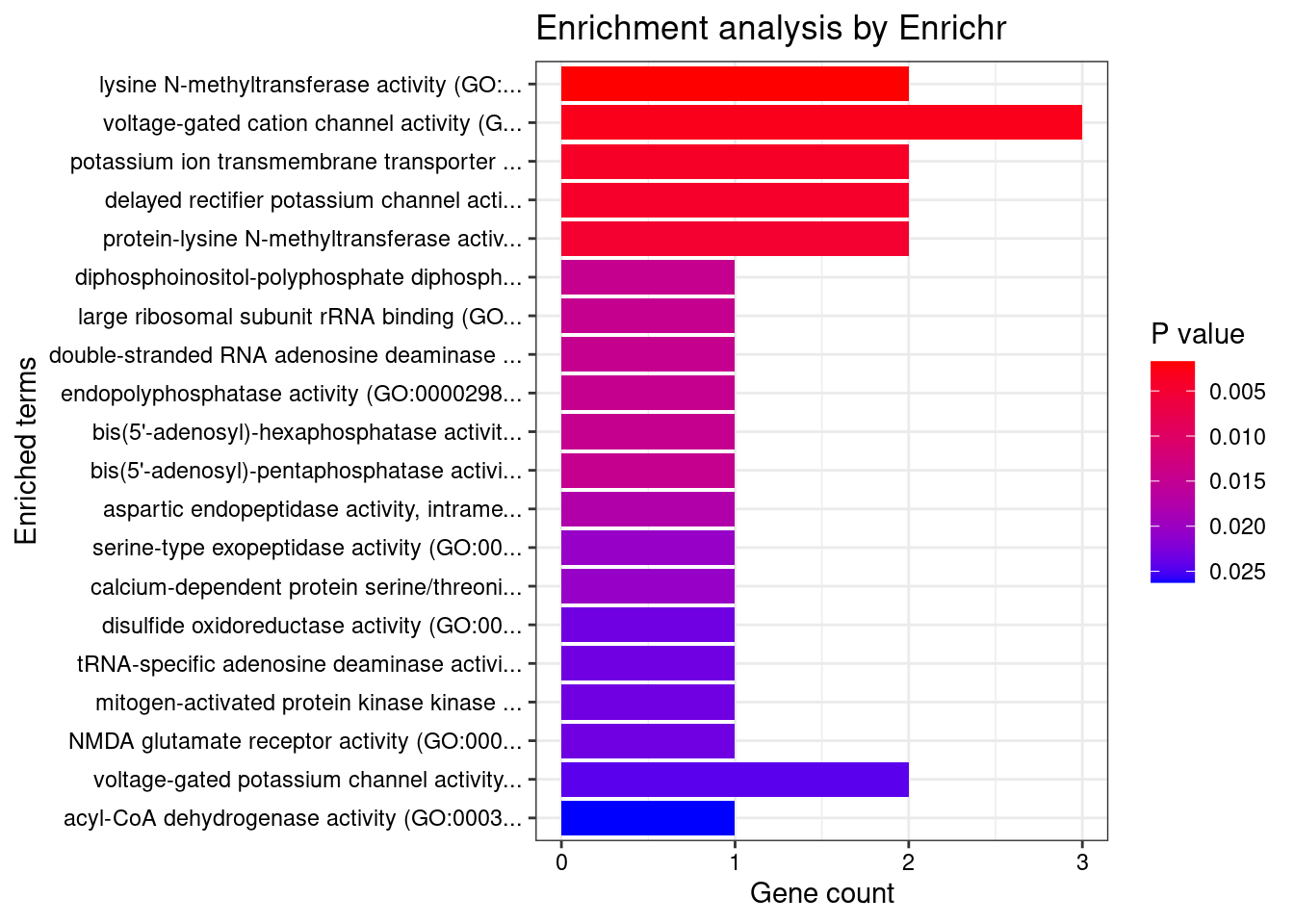
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)DisGeNET enrichment analysis for genes with PIP>0.5
Description FDR Ratio BgRatio
26 Confusion 0.02379 1/27 1/9703
51 Gingival Hypertrophy 0.02379 1/27 1/9703
62 Infant, Premature, Diseases 0.02379 1/27 1/9703
94 Pneumonia, Viral 0.02379 1/27 1/9703
96 Prostatic Neoplasms 0.02379 7/27 616/9703
103 Schizophrenia 0.02379 8/27 883/9703
125 Left Ventricular Hypertrophy 0.02379 2/27 25/9703
142 Speech impairment 0.02379 1/27 1/9703
143 Derealization 0.02379 1/27 1/9703
154 Spondylometaphyseal dysplasia, Kozlowski type 0.02379 1/27 1/9703WebGestalt enrichment analysis for genes with PIP>0.5
Loading the functional categories...
Loading the ID list...
Loading the reference list...
Performing the enrichment analysis...Warning in oraEnrichment(interestGeneList, referenceGeneList, geneSet, minNum =
minNum, : No significant gene set is identified based on FDR 0.05!NULLPIP Manhattan Plot
Warning: 'timedatectl' indicates the non-existent timezone name 'n/a'Warning: Your system is mis-configured: '/etc/localtime' is not a symlinkWarning: It is strongly recommended to set envionment variable TZ to 'America/
Chicago' (or equivalent)Warning: ggrepel: 8 unlabeled data points (too many overlaps). Consider
increasing max.overlaps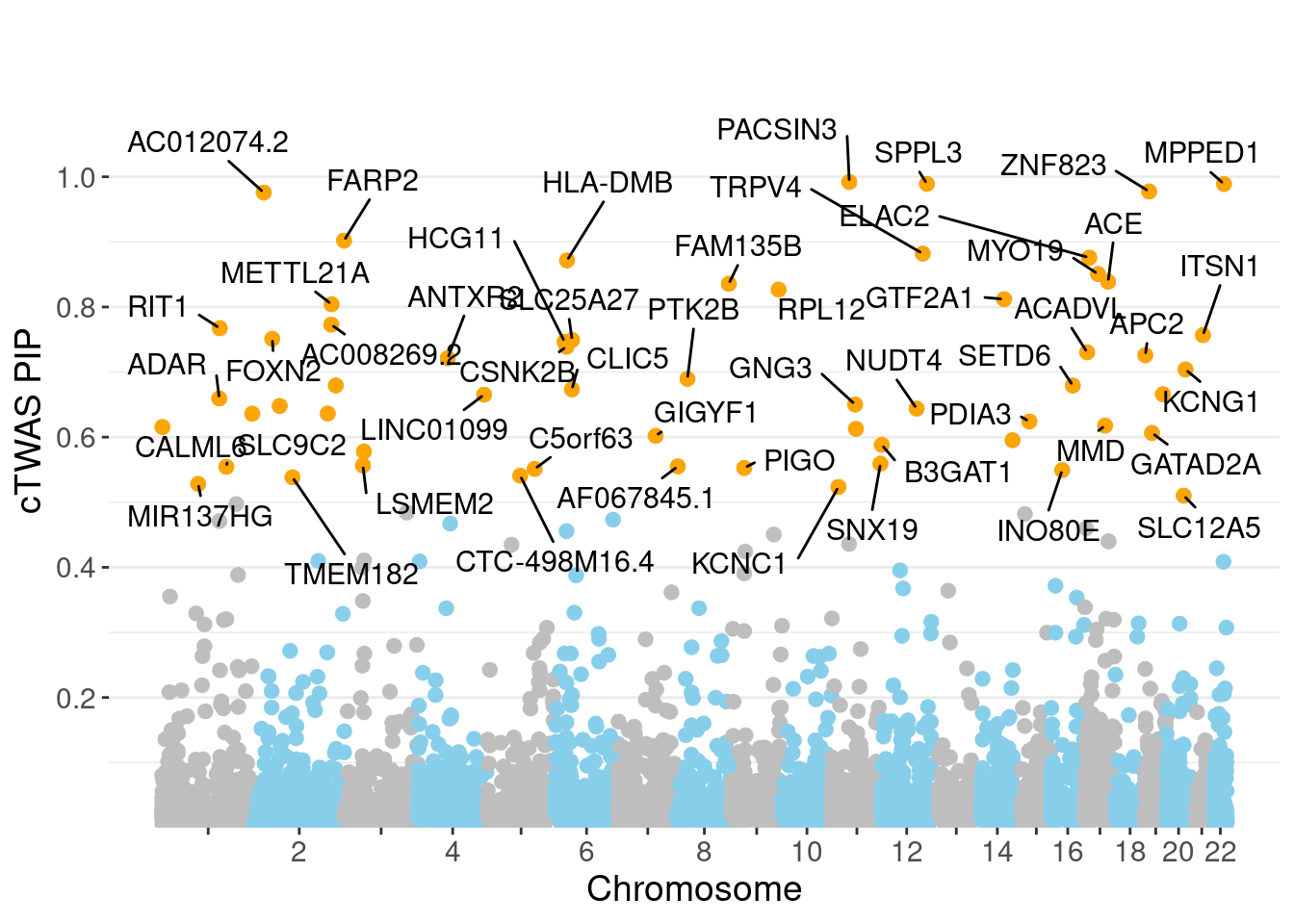
Sensitivity, specificity and precision for silver standard genes
#number of genes in known annotations
print(length(known_annotations))[1] 130#number of genes in known annotations with imputed expression
print(sum(known_annotations %in% ctwas_gene_res$genename))[1] 65#significance threshold for TWAS
print(sig_thresh)[1] 4.595#number of ctwas genes
length(ctwas_genes)[1] 15#number of TWAS genes
length(twas_genes)[1] 168#show novel genes (ctwas genes with not in TWAS genes)
ctwas_gene_res[ctwas_gene_res$genename %in% novel_genes,report_cols] genename region_tag susie_pip mu2 PVE z num_eqtl
5922 METTL21A 2_122 0.8042 25.32 0.0001261 -4.404 1
101 FARP2 2_144 0.9020 22.41 0.0001253 4.355 3
6200 FAM135B 8_91 0.8356 22.25 0.0001152 -3.461 1
2677 TRPV4 12_66 0.8820 24.20 0.0001322 4.416 1#sensitivity / recall
print(sensitivity) ctwas TWAS
0.02308 0.16154 #specificity
print(specificity) ctwas TWAS
0.9990 0.9872 #precision / PPV
print(precision)ctwas TWAS
0.200 0.125 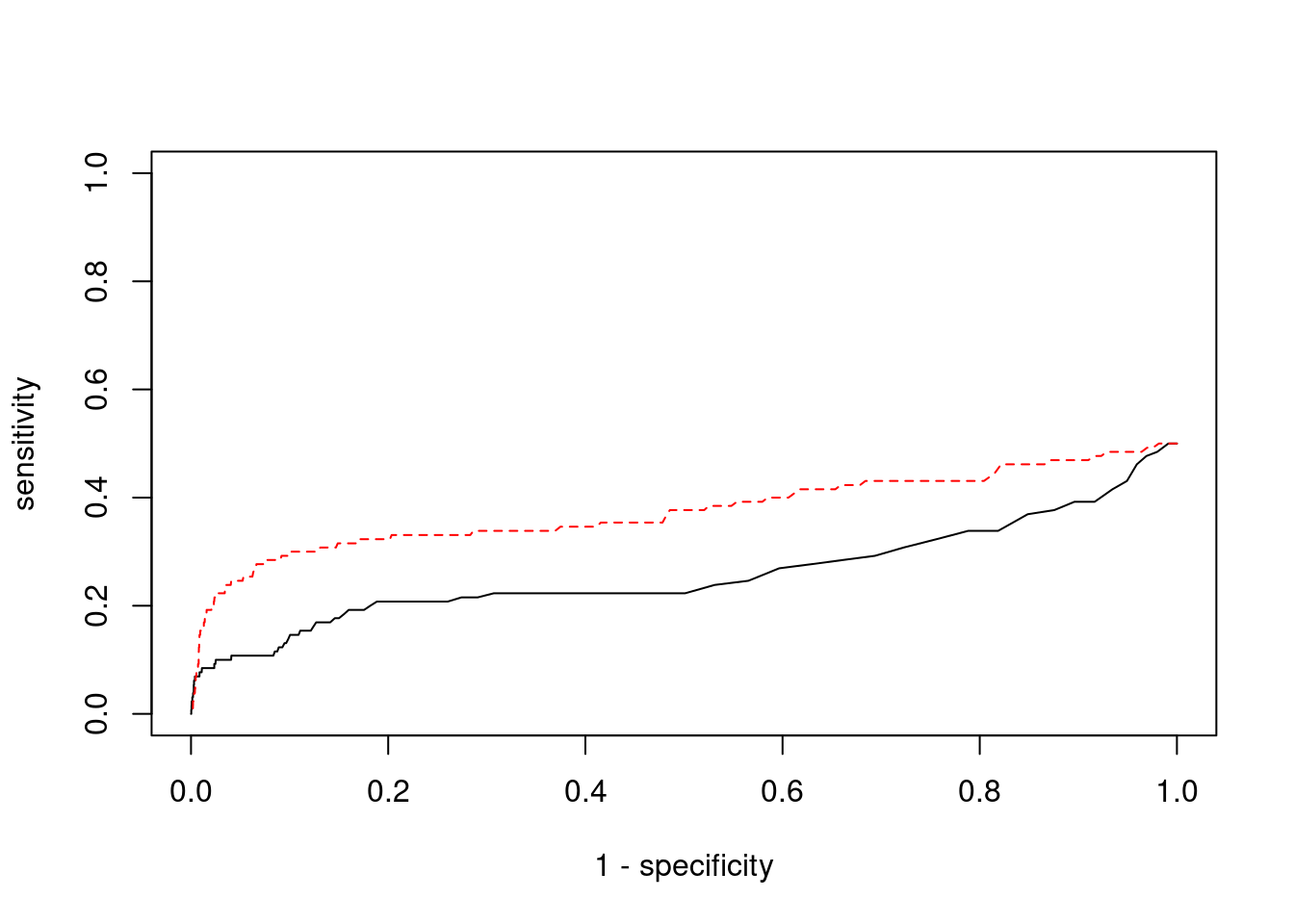
cTWAS is more precise than TWAS in distinguishing silver standard and bystander genes
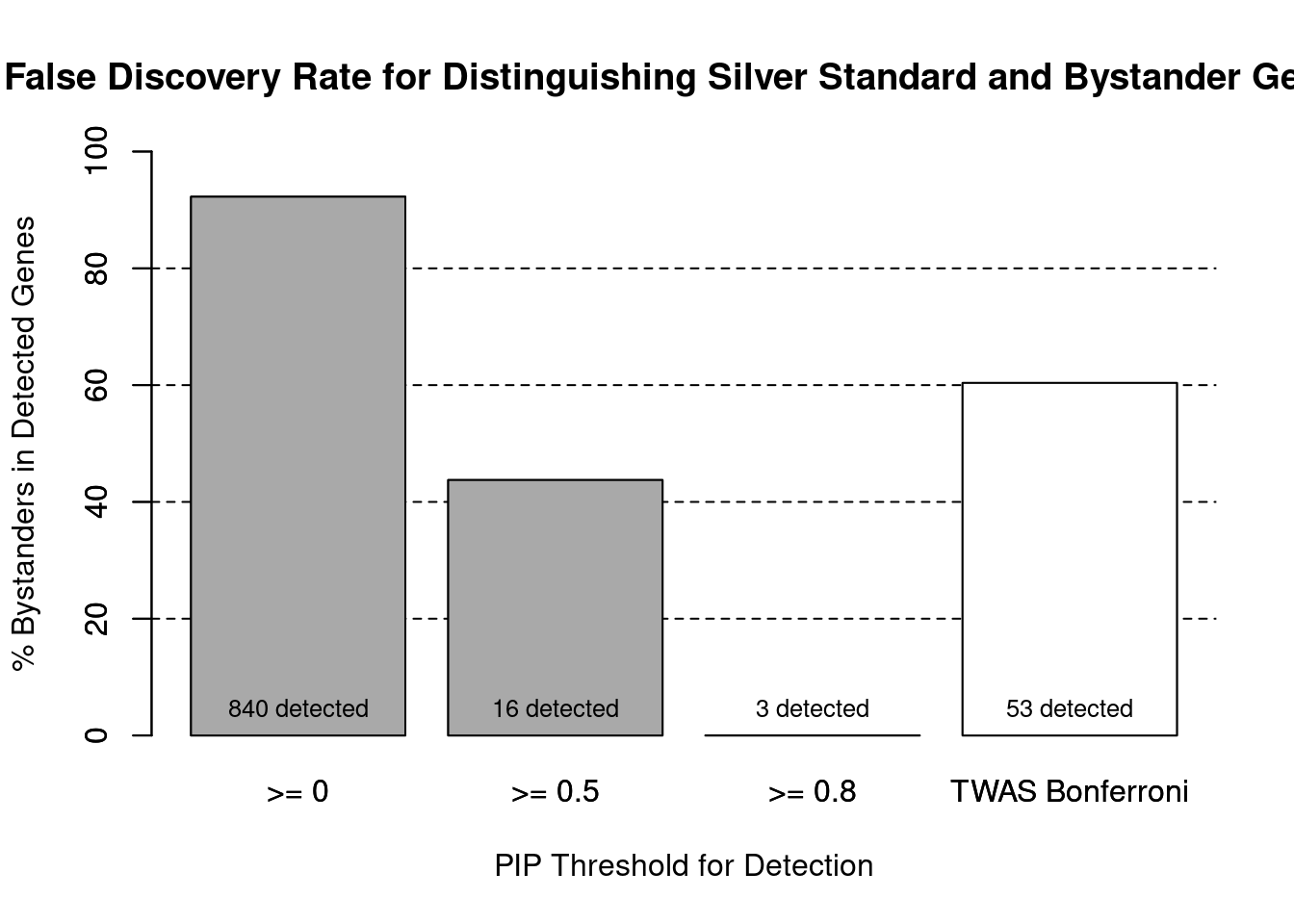
#number of genes in known annotations (with imputed expression)
print(length(known_annotations))[1] 65#number of bystander genes (with imputed expression)
print(length(unrelated_genes))[1] 775#subset results to genes in known annotations or bystanders
ctwas_gene_res_subset <- ctwas_gene_res[ctwas_gene_res$genename %in% c(known_annotations, unrelated_genes),]
#assign ctwas and TWAS genes
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>0.8]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>sig_thresh]
#significance threshold for TWAS
print(sig_thresh)[1] 4.595#number of ctwas genes (in known annotations or bystanders)
length(ctwas_genes)[1] 3#number of TWAS genes (in known annotations or bystanders)
length(twas_genes)[1] 53#sensitivity / recall
sensitivity ctwas TWAS
0.04615 0.32308 #specificity / (1 - False Positive Rate)
specificity ctwas TWAS
1.0000 0.9587 #precision / PPV / (1 - False Discovery Rate)
precision ctwas TWAS
1.0000 0.3962 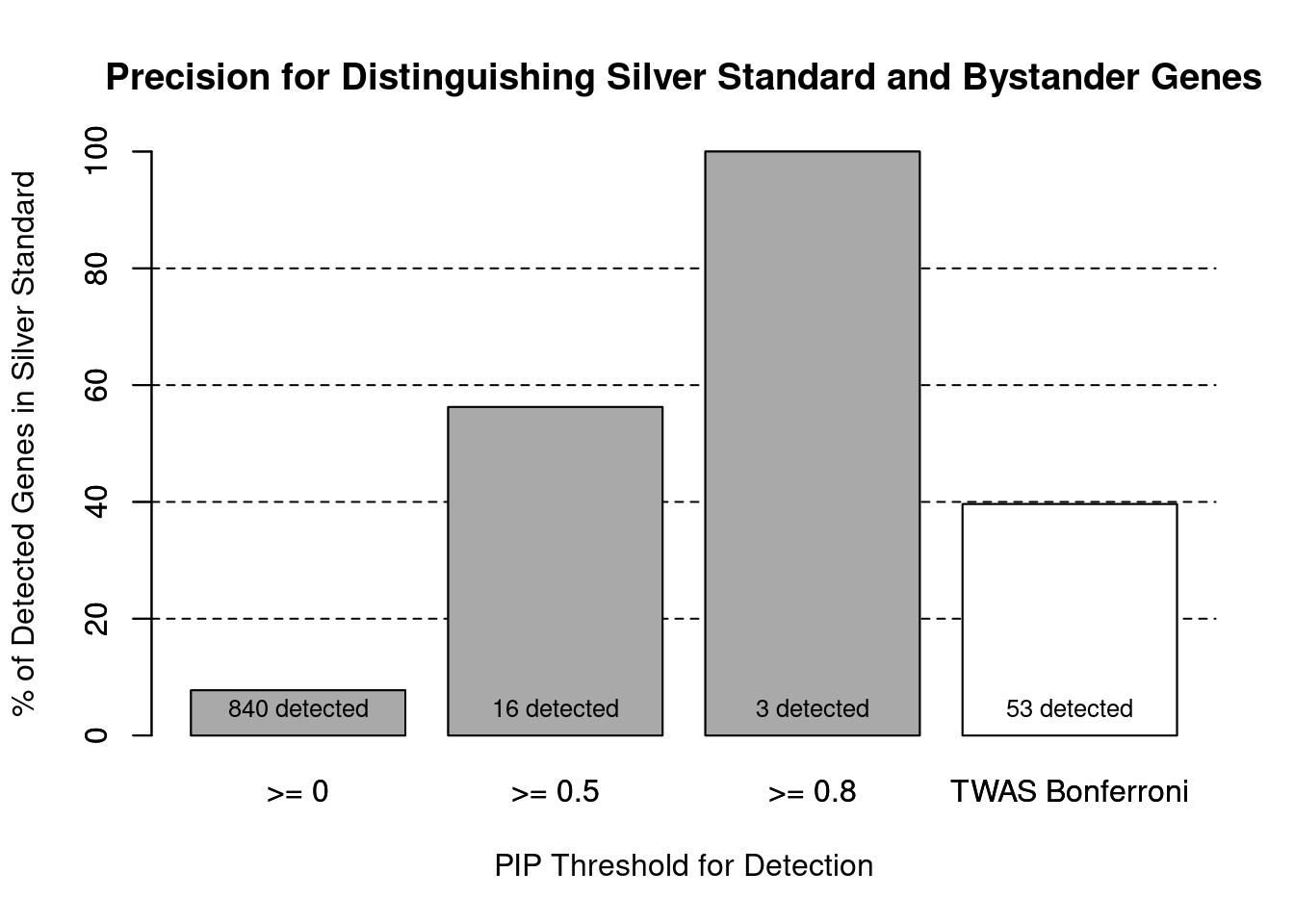
pip_range <- (0:1000)/1000
sensitivity <- rep(NA, length(pip_range))
specificity <- rep(NA, length(pip_range))
for (index in 1:length(pip_range)){
pip <- pip_range[index]
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>=pip]
sensitivity[index] <- sum(ctwas_genes %in% known_annotations)/length(known_annotations)
specificity[index] <- sum(!(unrelated_genes %in% ctwas_genes))/length(unrelated_genes)
}
plot(1-specificity, sensitivity, type="l", xlim=c(0,1), ylim=c(0,1), main="", xlab="1 - Specificity", ylab="Sensitivity")
title(expression("ROC Curve for cTWAS (black) and TWAS (" * phantom("red") * ")"))
title(expression(phantom("ROC Curve for cTWAS (black) and TWAS (") * "red" * phantom(")")), col.main="red")
sig_thresh_range <- seq(from=0, to=max(abs(ctwas_gene_res_subset$z)), length.out=length(pip_range))
for (index in 1:length(sig_thresh_range)){
sig_thresh_plot <- sig_thresh_range[index]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>=sig_thresh_plot]
sensitivity[index] <- sum(twas_genes %in% known_annotations)/length(known_annotations)
specificity[index] <- sum(!(unrelated_genes %in% twas_genes))/length(unrelated_genes)
}
lines(1-specificity, sensitivity, xlim=c(0,1), ylim=c(0,1), col="red", lty=1)
abline(a=0,b=1,lty=3)
#add previously computed points from the analysis
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>0.8]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>sig_thresh]
points(1-specificity_plot["ctwas"], sensitivity_plot["ctwas"], pch=21, bg="black")
points(1-specificity_plot["TWAS"], sensitivity_plot["TWAS"], pch=21, bg="red")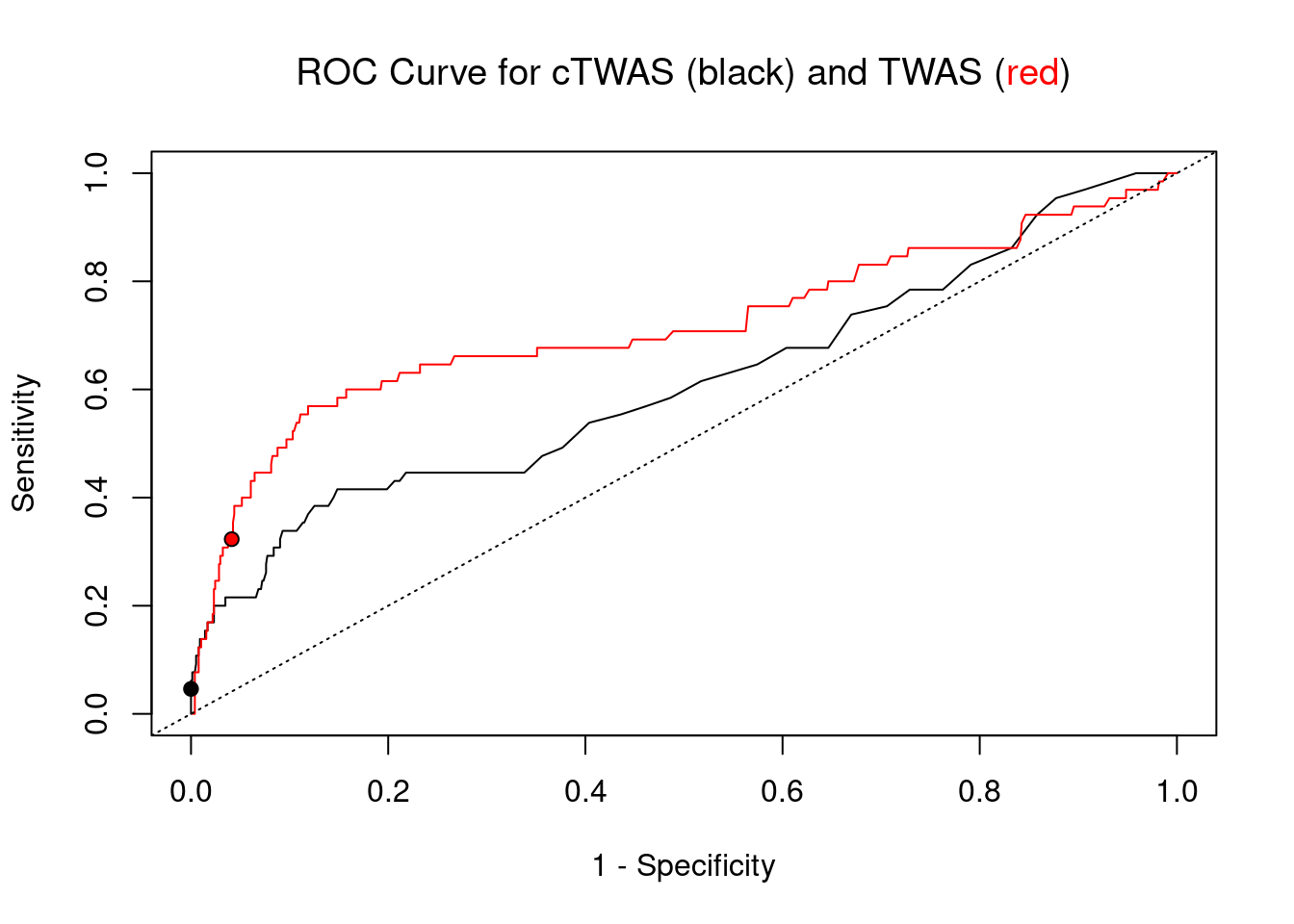
Undetected silver standard genes have low TWAS z-scores or stronger signal from nearby variants
#table of outcomes for silver standard genes
-sort(-table(silver_standard_case))silver_standard_case
Not Imputed Insignificant z-score Nearby SNP(s)
65 44 18
Detected (PIP > 0.8)
3 #show inconclusive genes
silver_standard_case[silver_standard_case=="Inconclusive"]named character(0)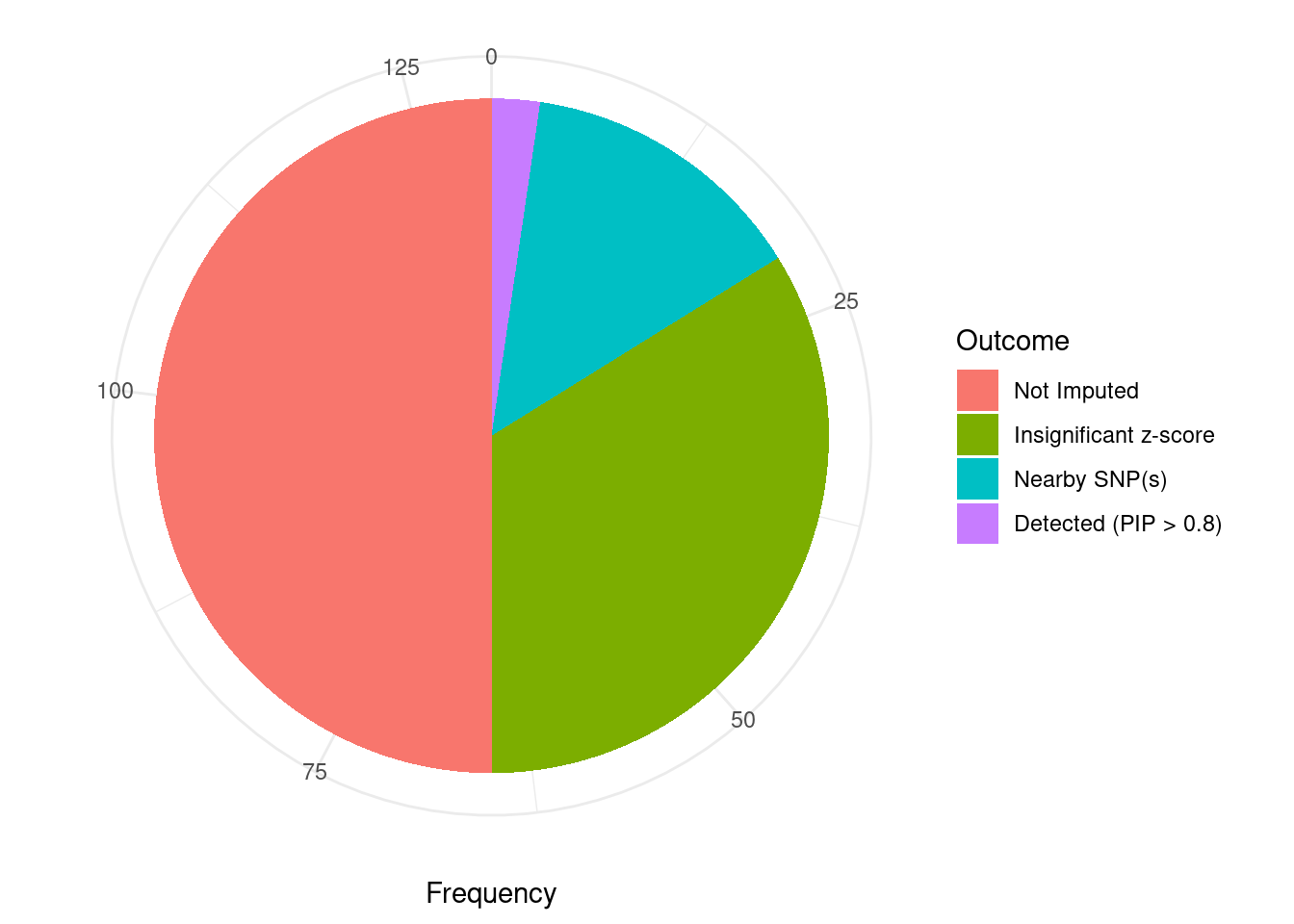
sessionInfo()R version 3.6.1 (2019-07-05)
Platform: x86_64-pc-linux-gnu (64-bit)
Running under: Scientific Linux 7.4 (Nitrogen)
Matrix products: default
BLAS/LAPACK: /software/openblas-0.2.19-el7-x86_64/lib/libopenblas_haswellp-r0.2.19.so
locale:
[1] LC_CTYPE=en_US.UTF-8 LC_NUMERIC=C
[3] LC_TIME=en_US.UTF-8 LC_COLLATE=en_US.UTF-8
[5] LC_MONETARY=en_US.UTF-8 LC_MESSAGES=en_US.UTF-8
[7] LC_PAPER=en_US.UTF-8 LC_NAME=C
[9] LC_ADDRESS=C LC_TELEPHONE=C
[11] LC_MEASUREMENT=en_US.UTF-8 LC_IDENTIFICATION=C
attached base packages:
[1] parallel stats4 stats graphics grDevices utils datasets
[8] methods base
other attached packages:
[1] GenomicRanges_1.36.1 GenomeInfoDb_1.20.0 IRanges_2.18.1
[4] S4Vectors_0.22.1 BiocGenerics_0.30.0 biomaRt_2.40.1
[7] readxl_1.3.1 forcats_0.5.1 stringr_1.4.0
[10] dplyr_1.0.7 purrr_0.3.4 readr_2.1.1
[13] tidyr_1.1.4 tidyverse_1.3.1 tibble_3.1.6
[16] WebGestaltR_0.4.4 disgenet2r_0.99.2 enrichR_3.0
[19] cowplot_1.1.1 ggplot2_3.3.5 workflowr_1.7.0
loaded via a namespace (and not attached):
[1] ggbeeswarm_0.6.0 colorspace_2.0-2 rjson_0.2.20
[4] ellipsis_0.3.2 rprojroot_2.0.2 XVector_0.24.0
[7] fs_1.5.2 rstudioapi_0.13 farver_2.1.0
[10] ggrepel_0.9.1 bit64_4.0.5 AnnotationDbi_1.46.0
[13] fansi_1.0.2 lubridate_1.8.0 xml2_1.3.3
[16] codetools_0.2-16 doParallel_1.0.17 cachem_1.0.6
[19] knitr_1.36 jsonlite_1.7.2 apcluster_1.4.8
[22] Cairo_1.5-12.2 broom_0.7.10 dbplyr_2.1.1
[25] compiler_3.6.1 httr_1.4.2 backports_1.4.1
[28] assertthat_0.2.1 Matrix_1.2-18 fastmap_1.1.0
[31] cli_3.1.0 later_0.8.0 prettyunits_1.1.1
[34] htmltools_0.5.2 tools_3.6.1 igraph_1.2.10
[37] GenomeInfoDbData_1.2.1 gtable_0.3.0 glue_1.6.2
[40] reshape2_1.4.4 doRNG_1.8.2 Rcpp_1.0.8
[43] Biobase_2.44.0 cellranger_1.1.0 jquerylib_0.1.4
[46] vctrs_0.3.8 svglite_1.2.2 iterators_1.0.14
[49] xfun_0.29 ps_1.6.0 rvest_1.0.2
[52] lifecycle_1.0.1 rngtools_1.5.2 XML_3.99-0.3
[55] zlibbioc_1.30.0 getPass_0.2-2 scales_1.1.1
[58] vroom_1.5.7 hms_1.1.1 promises_1.0.1
[61] yaml_2.2.1 curl_4.3.2 memoise_2.0.1
[64] ggrastr_1.0.1 gdtools_0.1.9 stringi_1.7.6
[67] RSQLite_2.2.8 highr_0.9 foreach_1.5.2
[70] rlang_1.0.1 pkgconfig_2.0.3 bitops_1.0-7
[73] evaluate_0.14 lattice_0.20-38 labeling_0.4.2
[76] bit_4.0.4 processx_3.5.2 tidyselect_1.1.1
[79] plyr_1.8.6 magrittr_2.0.2 R6_2.5.1
[82] generics_0.1.1 DBI_1.1.2 pillar_1.6.4
[85] haven_2.4.3 whisker_0.3-2 withr_2.4.3
[88] RCurl_1.98-1.5 modelr_0.1.8 crayon_1.5.0
[91] utf8_1.2.2 tzdb_0.2.0 rmarkdown_2.11
[94] progress_1.2.2 grid_3.6.1 data.table_1.14.2
[97] blob_1.2.2 callr_3.7.0 git2r_0.26.1
[100] reprex_2.0.1 digest_0.6.29 httpuv_1.5.1
[103] munsell_0.5.0 beeswarm_0.2.3 vipor_0.4.5