SCZ - Brain Hypothalamus
sheng Qian
2021-2-6
Last updated: 2022-03-05
Checks: 5 2
Knit directory: cTWAS_analysis/
This reproducible R Markdown analysis was created with workflowr (version 1.7.0). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
The R Markdown file has unstaged changes. To know which version of the R Markdown file created these results, you’ll want to first commit it to the Git repo. If you’re still working on the analysis, you can ignore this warning. When you’re finished, you can run wflow_publish to commit the R Markdown file and build the HTML.
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
The command set.seed(20211220) was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
Nice! There were no cached chunks for this analysis, so you can be confident that you successfully produced the results during this run.
Using absolute paths to the files within your workflowr project makes it difficult for you and others to run your code on a different machine. Change the absolute path(s) below to the suggested relative path(s) to make your code more reproducible.
| absolute | relative |
|---|---|
| /project2/xinhe/shengqian/cTWAS/cTWAS_analysis/data/ | data |
| /project2/xinhe/shengqian/cTWAS/cTWAS_analysis/code/ctwas_config.R | code/ctwas_config.R |
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility.
The results in this page were generated with repository version 4a5db1c. See the Past versions tab to see a history of the changes made to the R Markdown and HTML files.
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can use wflow_publish or wflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Ignored files:
Ignored: .ipynb_checkpoints/
Ignored: data/AF/
Untracked files:
Untracked: Rplot.png
Untracked: analysis/.ipynb_checkpoints/
Untracked: code/.ipynb_checkpoints/
Untracked: code/AF_out/
Untracked: code/Autism_out/
Untracked: code/BMI_S_out/
Untracked: code/BMI_out/
Untracked: code/Glucose_out/
Untracked: code/LDL_S_out/
Untracked: code/SCZ_S_out/
Untracked: code/SCZ_out/
Untracked: code/T2D_out/
Untracked: code/ctwas_config.R
Untracked: code/mapping.R
Untracked: code/out/
Untracked: code/run_AF_analysis.sbatch
Untracked: code/run_AF_analysis.sh
Untracked: code/run_AF_ctwas_rss_LDR.R
Untracked: code/run_Autism_analysis.sbatch
Untracked: code/run_Autism_analysis.sh
Untracked: code/run_Autism_ctwas_rss_LDR.R
Untracked: code/run_BMI_analysis.sbatch
Untracked: code/run_BMI_analysis.sh
Untracked: code/run_BMI_analysis_S.sbatch
Untracked: code/run_BMI_analysis_S.sh
Untracked: code/run_BMI_ctwas_rss_LDR.R
Untracked: code/run_BMI_ctwas_rss_LDR_S.R
Untracked: code/run_Glucose_analysis.sbatch
Untracked: code/run_Glucose_analysis.sh
Untracked: code/run_Glucose_ctwas_rss_LDR.R
Untracked: code/run_LDL_analysis_S.sbatch
Untracked: code/run_LDL_analysis_S.sh
Untracked: code/run_LDL_ctwas_rss_LDR_S.R
Untracked: code/run_SCZ_analysis.sbatch
Untracked: code/run_SCZ_analysis.sh
Untracked: code/run_SCZ_analysis_S.sbatch
Untracked: code/run_SCZ_analysis_S.sh
Untracked: code/run_SCZ_ctwas_rss_LDR.R
Untracked: code/run_SCZ_ctwas_rss_LDR_S.R
Untracked: code/run_T2D_analysis.sbatch
Untracked: code/run_T2D_analysis.sh
Untracked: code/run_T2D_ctwas_rss_LDR.R
Untracked: code/wflow_build.R
Untracked: code/wflow_build.sbatch
Untracked: data/.ipynb_checkpoints/
Untracked: data/BMI/
Untracked: data/SCZ/
Untracked: data/SCZ_S/
Untracked: data/T2D/
Untracked: data/UKBB/
Untracked: data/UKBB_SNPs_Info.text
Untracked: data/gene_OMIM.txt
Untracked: data/gene_pip_0.8.txt
Untracked: data/mashr_Heart_Atrial_Appendage.db
Untracked: data/mashr_sqtl/
Untracked: data/summary_known_genes_annotations.xlsx
Untracked: data/untitled.txt
Unstaged changes:
Modified: analysis/BMI_Brain_Putamen_basal_ganglia.Rmd
Modified: analysis/SCZ_Brain_Amygdala.Rmd
Modified: analysis/SCZ_Brain_Anterior_cingulate_cortex_BA24.Rmd
Modified: analysis/SCZ_Brain_Caudate_basal_ganglia.Rmd
Modified: analysis/SCZ_Brain_Cerebellar_Hemisphere.Rmd
Modified: analysis/SCZ_Brain_Cerebellum.Rmd
Modified: analysis/SCZ_Brain_Cortex.Rmd
Modified: analysis/SCZ_Brain_Frontal_Cortex_BA9.Rmd
Modified: analysis/SCZ_Brain_Hippocampus.Rmd
Modified: analysis/SCZ_Brain_Hypothalamus.Rmd
Modified: analysis/SCZ_Brain_Nucleus_accumbens_basal_ganglia.Rmd
Modified: analysis/SCZ_Brain_Spinal_cord_cervical_c-1.Rmd
Modified: analysis/SCZ_Brain_Substantia_nigra.Rmd
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
These are the previous versions of the repository in which changes were made to the R Markdown (analysis/SCZ_Brain_Hypothalamus.Rmd) and HTML (docs/SCZ_Brain_Hypothalamus.html) files. If you’ve configured a remote Git repository (see ?wflow_git_remote), click on the hyperlinks in the table below to view the files as they were in that past version.
| File | Version | Author | Date | Message |
|---|---|---|---|---|
| Rmd | 4a5db1c | sq-96 | 2022-03-03 | update |
| html | 4a5db1c | sq-96 | 2022-03-03 | update |
| html | addb825 | sq-96 | 2022-02-28 | Build site. |
| Rmd | 3f9e35f | sq-96 | 2022-02-28 | update |
| html | 75a1466 | sq-96 | 2022-02-27 | Build site. |
| Rmd | 1c69dd2 | sq-96 | 2022-02-27 | update |
| html | ff6403a | sq-96 | 2022-02-27 | Build site. |
| Rmd | 3dd5b4c | sq-96 | 2022-02-27 | update |
Weight QC
#number of imputed weights
nrow(qclist_all)[1] 11152#number of imputed weights by chromosome
table(qclist_all$chr)
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
1097 778 665 440 568 568 537 435 425 444 658 638 212 359 377 502
17 18 19 20 21 22
679 169 849 331 133 288 #number of imputed weights without missing variants
sum(qclist_all$nmiss==0)[1] 8929#proportion of imputed weights without missing variants
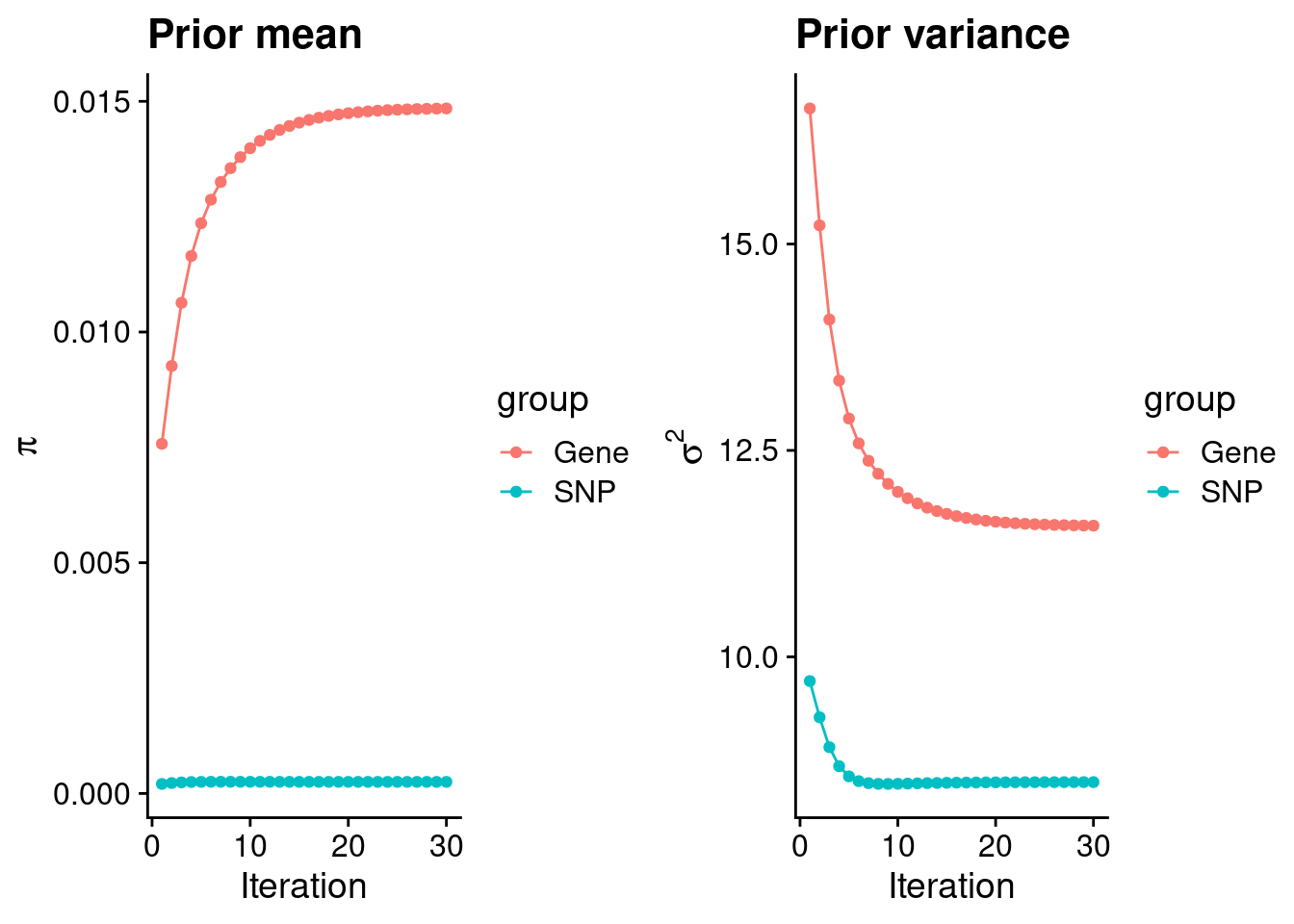
mean(qclist_all$nmiss==0)[1] 0.8007Check convergence of parameters
| Version | Author | Date |
|---|---|---|
| ff6403a | sq-96 | 2022-02-27 |
#estimated group prior
estimated_group_prior <- group_prior_rec[,ncol(group_prior_rec)]
names(estimated_group_prior) <- c("gene", "snp")
estimated_group_prior["snp"] <- estimated_group_prior["snp"]*thin #adjust parameter to account for thin argument
print(estimated_group_prior) gene snp
0.0148454 0.0002505 #estimated group prior variance
estimated_group_prior_var <- group_prior_var_rec[,ncol(group_prior_var_rec)]
names(estimated_group_prior_var) <- c("gene", "snp")
print(estimated_group_prior_var) gene snp
11.589 8.483 #report sample size
print(sample_size)[1] 82315#report group size
group_size <- c(nrow(ctwas_gene_res), n_snps)
print(group_size)[1] 11152 7573890#estimated group PVE
estimated_group_pve <- estimated_group_prior_var*estimated_group_prior*group_size/sample_size #check PVE calculation
names(estimated_group_pve) <- c("gene", "snp")
print(estimated_group_pve) gene snp
0.02331 0.19553 #compare sum(PIP*mu2/sample_size) with above PVE calculation
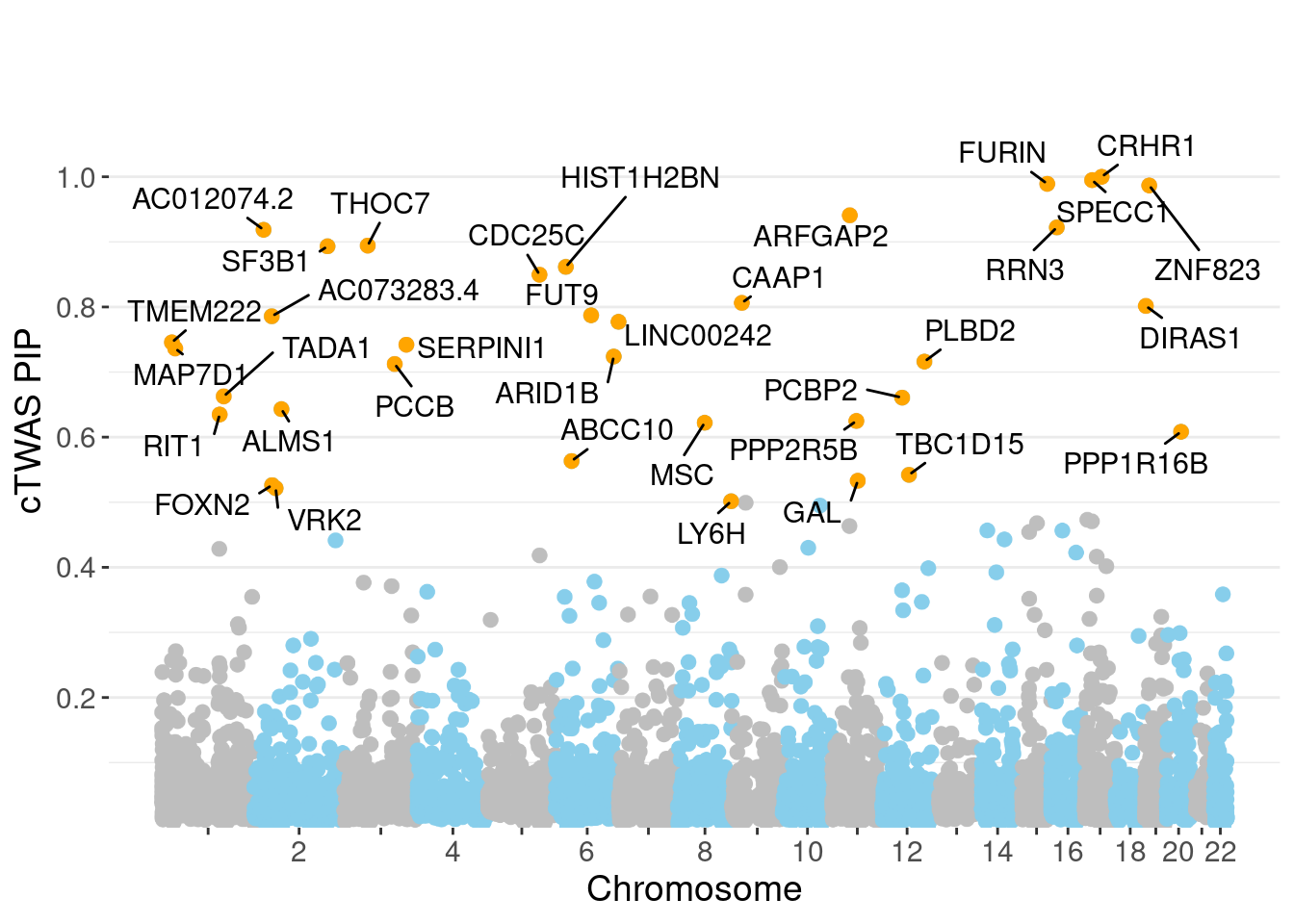
c(sum(ctwas_gene_res$PVE),sum(ctwas_snp_res$PVE))[1] 0.1358 1.4127Genes with highest PIPs
| Version | Author | Date |
|---|---|---|
| ff6403a | sq-96 | 2022-02-27 |
genename region_tag susie_pip mu2 PVE z num_eqtl
3376 CRHR1 17_27 1.0000 3865.12 0.0469552 3.362 1
3993 SPECC1 17_16 0.9949 32.10 0.0003879 5.624 2
5324 FURIN 15_42 0.9892 46.47 0.0005584 -7.000 1
10843 ZNF823 19_10 0.9867 29.93 0.0003588 5.479 2
6111 ARFGAP2 11_29 0.9408 24.98 0.0002856 4.740 1
1089 RRN3 16_15 0.9222 24.23 0.0002714 -4.560 2
11997 AC012074.2 2_15 0.9186 21.83 0.0002436 4.620 2
7382 THOC7 3_43 0.8943 31.42 0.0003414 -5.578 1
2970 SF3B1 2_117 0.8932 44.56 0.0004835 6.725 1
11948 HIST1H2BN 6_21 0.8616 93.98 0.0009837 10.773 1
6802 CDC25C 5_82 0.8496 25.55 0.0002637 -5.591 1
3381 CAAP1 9_20 0.8063 24.10 0.0002361 4.751 2
9203 DIRAS1 19_3 0.8016 24.23 0.0002360 4.765 2
8764 FUT9 6_65 0.7872 29.99 0.0002868 5.427 1
11985 AC073283.4 2_30 0.7857 20.57 0.0001964 -3.881 3
11817 LINC00242 6_112 0.7772 20.55 0.0001940 3.921 2
10218 TMEM222 1_19 0.7456 22.33 0.0002023 3.902 1
7356 SERPINI1 3_103 0.7420 20.10 0.0001812 -4.038 1
3112 MAP7D1 1_22 0.7364 24.37 0.0002180 4.907 1
434 ARID1B 6_102 0.7239 21.68 0.0001906 -3.907 1Genes with largest effect sizes
| Version | Author | Date |
|---|---|---|
| ff6403a | sq-96 | 2022-02-27 |
genename region_tag susie_pip mu2 PVE z num_eqtl
3376 CRHR1 17_27 1.000e+00 3865.12 4.696e-02 3.3623 1
10942 HLA-DRB5 6_26 0.000e+00 921.30 0.000e+00 2.9680 1
11166 MSH5 6_26 5.429e-09 839.87 5.539e-11 8.8255 2
10534 HLA-DRB1 6_26 0.000e+00 677.09 0.000e+00 5.1518 1
10645 HLA-DQA1 6_26 0.000e+00 376.40 0.000e+00 -1.6175 1
11162 HSPA1A 6_26 0.000e+00 359.84 0.000e+00 7.1259 1
11158 SLC44A4 6_26 0.000e+00 313.06 0.000e+00 6.1896 1
12073 HLA-DQA2 6_26 0.000e+00 306.49 0.000e+00 0.2164 1
11418 CLIC1 6_26 0.000e+00 262.34 0.000e+00 -0.4634 1
9485 HLA-DQB1 6_26 0.000e+00 217.87 0.000e+00 -1.4137 1
9661 ACBD4 17_27 0.000e+00 196.79 0.000e+00 1.6219 2
11620 C4B 6_26 0.000e+00 158.66 0.000e+00 -4.9282 1
11888 CYP21A2 6_26 0.000e+00 157.05 0.000e+00 3.7459 1
10008 FMNL1 17_27 0.000e+00 152.89 0.000e+00 0.6638 1
11948 HIST1H2BN 6_21 8.616e-01 93.98 9.837e-04 10.7729 1
11799 SAPCD1 6_26 0.000e+00 87.82 0.000e+00 -2.7814 1
2335 GOSR2 17_27 0.000e+00 76.11 0.000e+00 -2.5096 1
2736 PRSS16 6_21 1.002e-01 64.78 7.883e-05 -8.5674 1
11150 STK19 6_26 0.000e+00 56.67 0.000e+00 -2.0635 1
9592 HIST1H2BC 6_20 2.545e-02 54.50 1.685e-05 -8.0277 1Genes with highest PVE
genename region_tag susie_pip mu2 PVE z num_eqtl
3376 CRHR1 17_27 1.0000 3865.12 0.0469552 3.362 1
11948 HIST1H2BN 6_21 0.8616 93.98 0.0009837 10.773 1
5324 FURIN 15_42 0.9892 46.47 0.0005584 -7.000 1
2970 SF3B1 2_117 0.8932 44.56 0.0004835 6.725 1
3993 SPECC1 17_16 0.9949 32.10 0.0003879 5.624 2
10843 ZNF823 19_10 0.9867 29.93 0.0003588 5.479 2
7382 THOC7 3_43 0.8943 31.42 0.0003414 -5.578 1
2829 PCCB 3_84 0.7124 35.08 0.0003036 -6.358 1
8764 FUT9 6_65 0.7872 29.99 0.0002868 5.427 1
6111 ARFGAP2 11_29 0.9408 24.98 0.0002856 4.740 1
1089 RRN3 16_15 0.9222 24.23 0.0002714 -4.560 2
6802 CDC25C 5_82 0.8496 25.55 0.0002637 -5.591 1
1685 PPP1R16B 20_23 0.6083 35.39 0.0002615 6.091 1
11997 AC012074.2 2_15 0.9186 21.83 0.0002436 4.620 2
3381 CAAP1 9_20 0.8063 24.10 0.0002361 4.751 2
9203 DIRAS1 19_3 0.8016 24.23 0.0002360 4.765 2
2505 MDK 11_28 0.4635 38.86 0.0002188 -6.357 1
3112 MAP7D1 1_22 0.7364 24.37 0.0002180 4.907 1
6178 TAOK2 16_24 0.4564 37.80 0.0002096 6.189 1
3041 ALMS1 2_48 0.6430 26.52 0.0002072 -5.154 1Genes with largest z scores
genename region_tag susie_pip mu2 PVE z num_eqtl
11948 HIST1H2BN 6_21 8.616e-01 93.98 9.837e-04 10.773 1
11166 MSH5 6_26 5.429e-09 839.87 5.539e-11 8.825 2
2736 PRSS16 6_21 1.002e-01 64.78 7.883e-05 -8.567 1
10214 BTN3A2 6_20 1.908e-02 53.75 1.246e-05 8.047 3
9592 HIST1H2BC 6_20 2.545e-02 54.50 1.685e-05 -8.028 1
2696 TRIM38 6_20 1.994e-02 49.73 1.205e-05 -7.769 2
11162 HSPA1A 6_26 0.000e+00 359.84 0.000e+00 7.126 1
5324 FURIN 15_42 9.892e-01 46.47 5.584e-04 -7.000 1
2970 SF3B1 2_117 8.932e-01 44.56 4.835e-04 6.725 1
10360 ZSCAN23 6_22 1.101e-01 46.18 6.177e-05 -6.717 2
1571 ZFYVE21 14_54 1.435e-01 40.59 7.079e-05 -6.708 1
3855 XRCC3 14_54 1.179e-01 42.60 6.100e-05 6.690 1
9715 ARL6IP4 12_75 1.257e-02 40.62 6.201e-06 6.491 1
10694 ZSCAN26 6_22 2.479e-02 28.09 8.460e-06 6.389 2
2829 PCCB 3_84 7.124e-01 35.08 3.036e-04 -6.358 1
2505 MDK 11_28 4.635e-01 38.86 2.188e-04 -6.357 1
11158 SLC44A4 6_26 0.000e+00 313.06 0.000e+00 6.190 1
6178 TAOK2 16_24 4.564e-01 37.80 2.096e-04 6.189 1
12000 ZSCAN31 6_22 1.972e-02 33.00 7.908e-06 -6.182 3
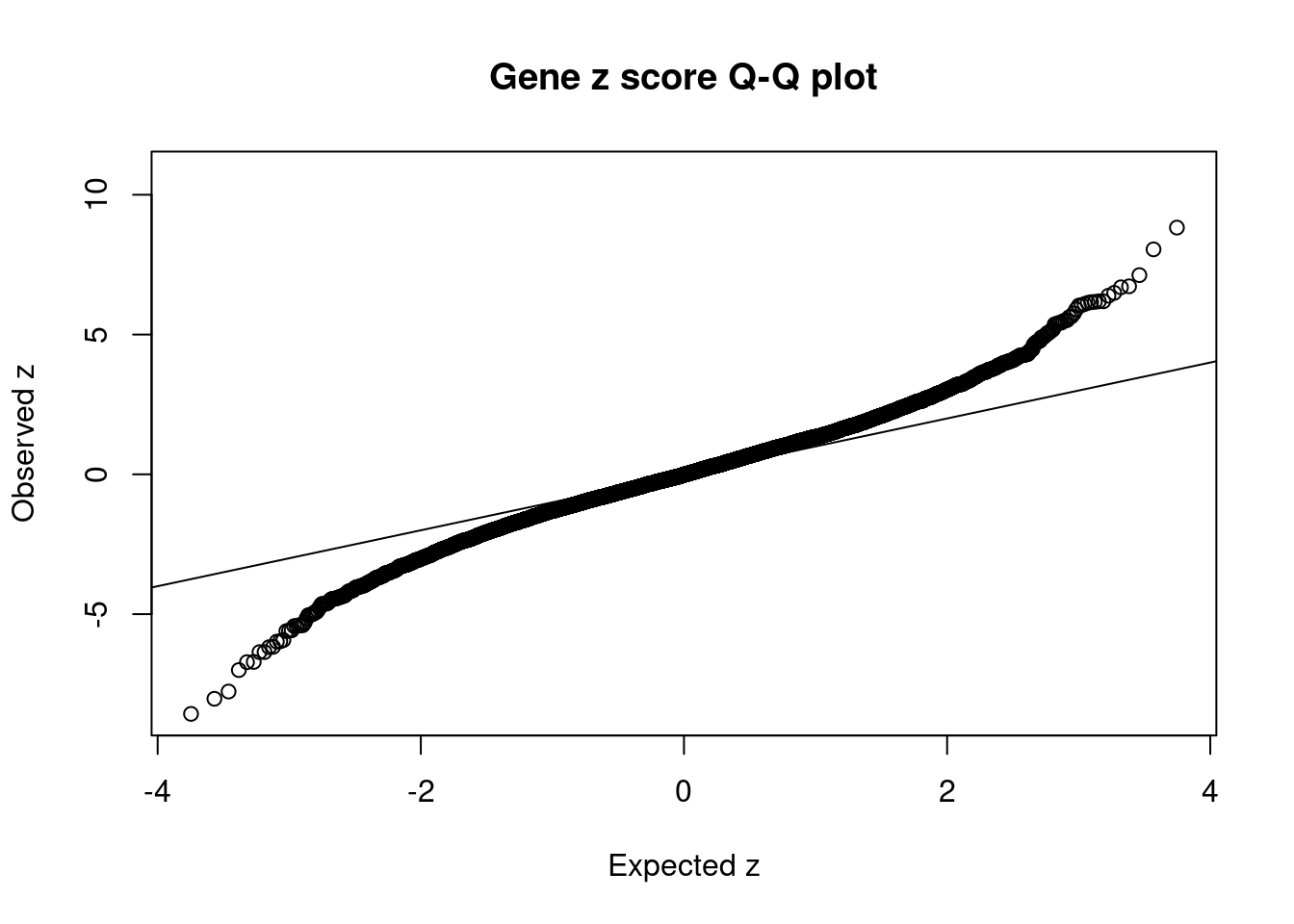
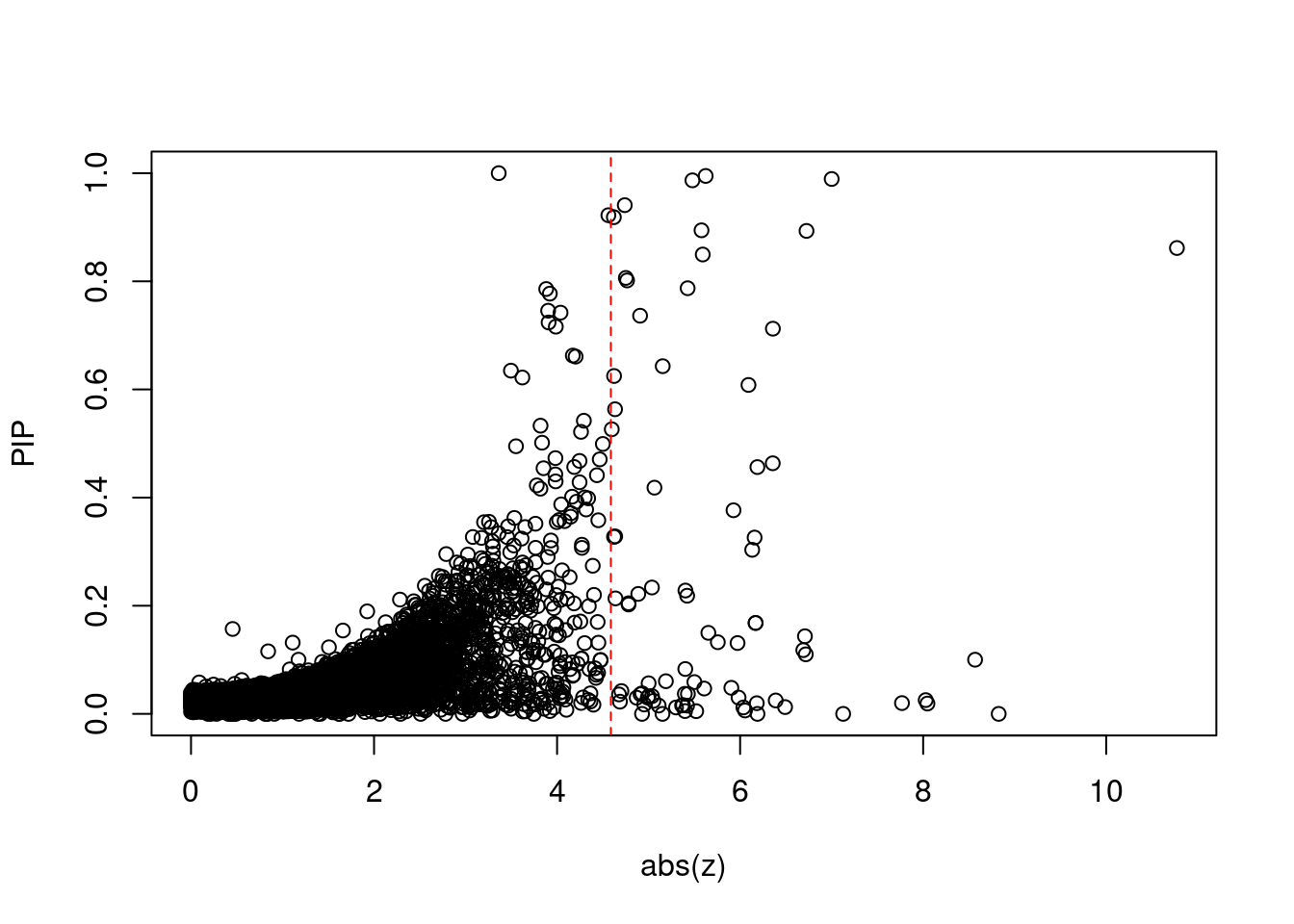
9577 HARBI1 11_28 1.681e-01 36.17 7.388e-05 6.169 1Comparing z scores and PIPs
| Version | Author | Date |
|---|---|---|
| ff6403a | sq-96 | 2022-02-27 |
| Version | Author | Date |
|---|---|---|
| ff6403a | sq-96 | 2022-02-27 |
[1] 0.007443GO enrichment analysis for genes with PIP>0.5
#number of genes for gene set enrichment
length(genes)[1] 35Uploading data to Enrichr... Done.
Querying GO_Biological_Process_2021... Done.
Querying GO_Cellular_Component_2021... Done.
Querying GO_Molecular_Function_2021... Done.
Parsing results... Done.
[1] "GO_Biological_Process_2021"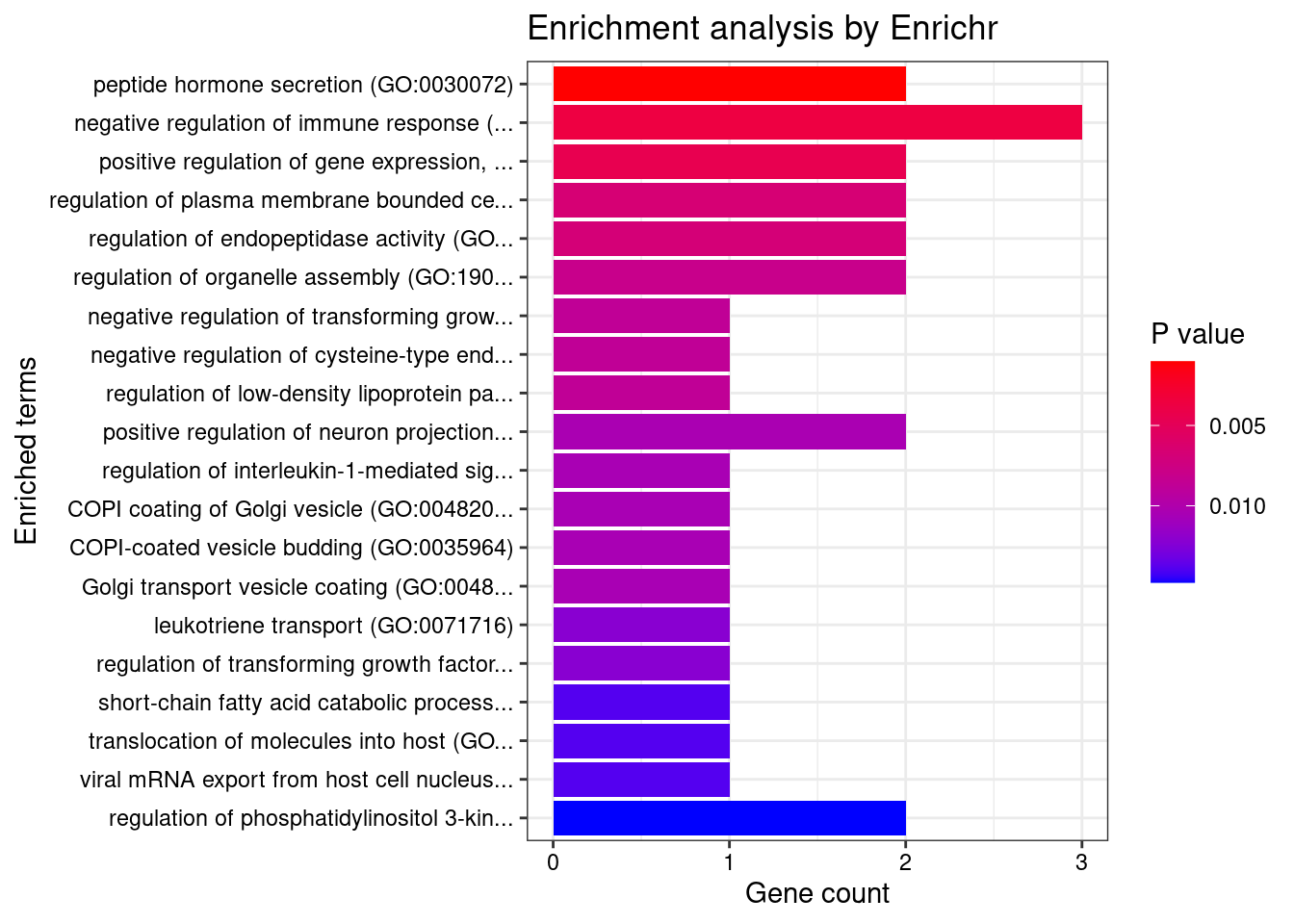
| Version | Author | Date |
|---|---|---|
| ff6403a | sq-96 | 2022-02-27 |
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)
[1] "GO_Cellular_Component_2021"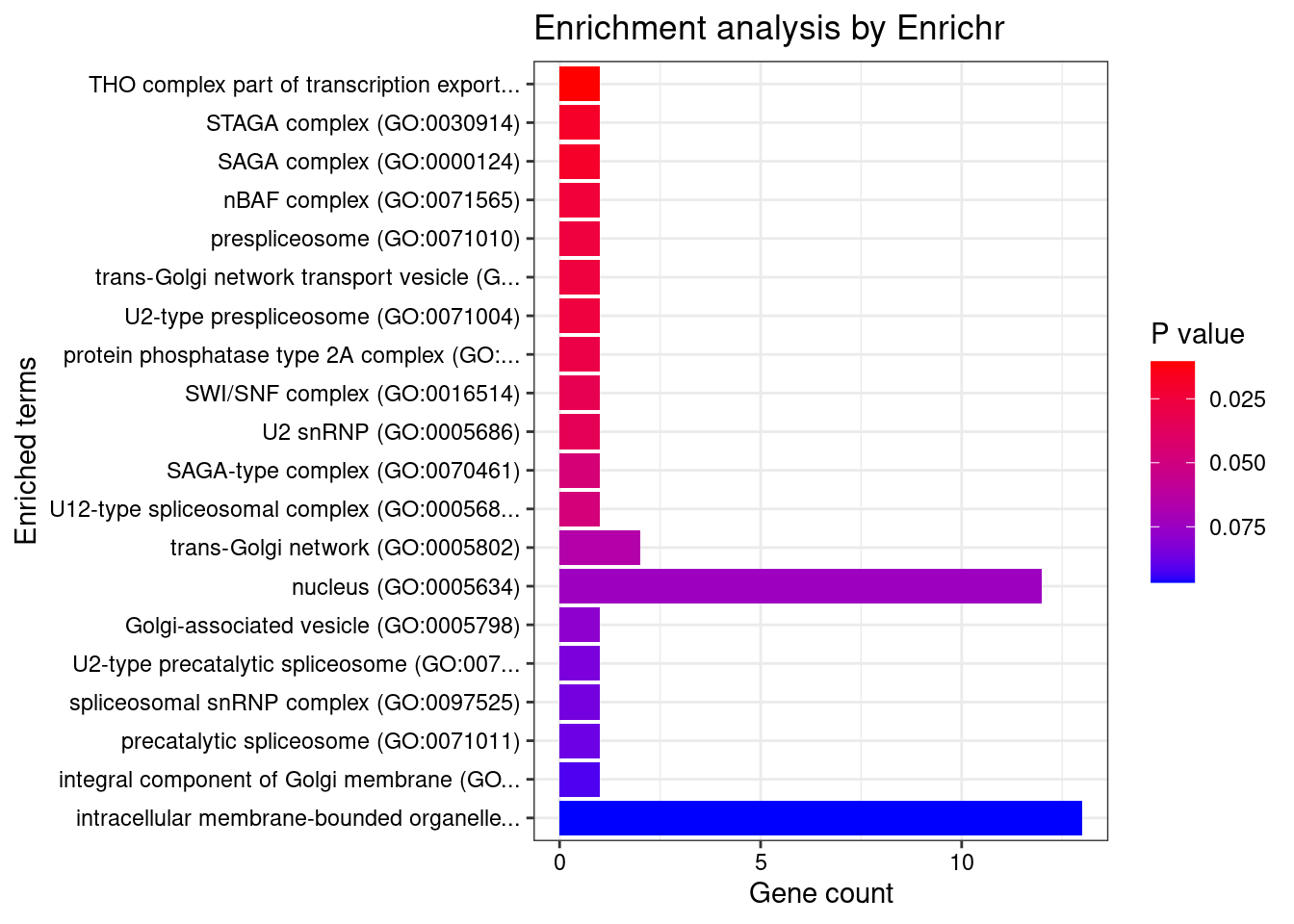
| Version | Author | Date |
|---|---|---|
| ff6403a | sq-96 | 2022-02-27 |
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)
[1] "GO_Molecular_Function_2021"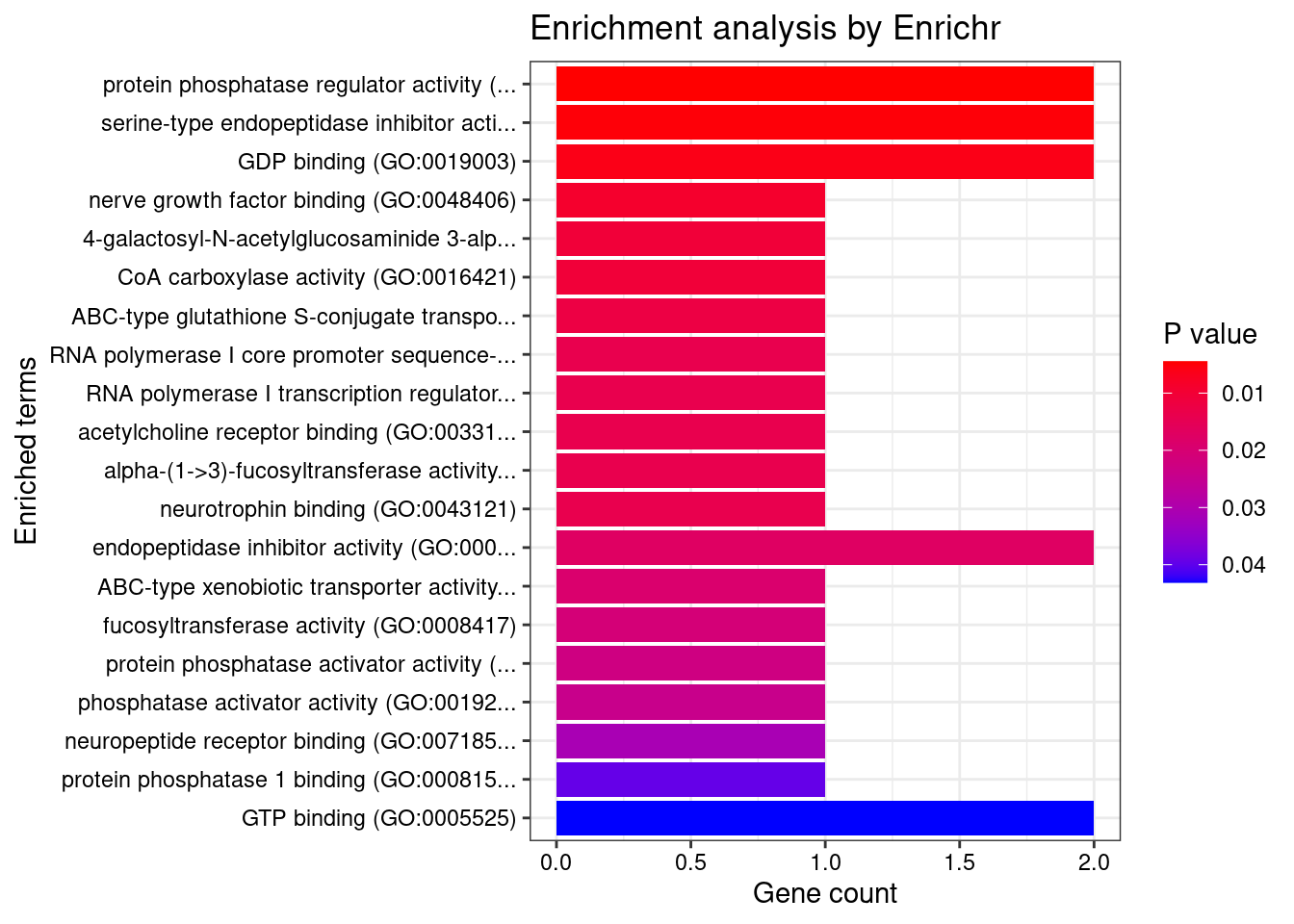
| Version | Author | Date |
|---|---|---|
| ff6403a | sq-96 | 2022-02-27 |
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)DisGeNET enrichment analysis for genes with PIP>0.5
Description FDR Ratio
11 Involutional Depression 0.01732 2/12
17 Hirschsprung Disease 0.01732 2/12
63 Alstrom Syndrome 0.01732 1/12
109 Involutional paraphrenia 0.01732 2/12
110 Psychosis, Involutional 0.01732 2/12
113 Familial encephalopathy with neuroserpin inclusion bodies 0.01732 1/12
114 Childhood-onset truncal obesity 0.01732 1/12
123 NOONAN SYNDROME 8 0.01732 1/12
125 EPILEPSY, FAMILIAL TEMPORAL LOBE, 8 0.01732 1/12
106 Cardiomyopathy, Familial Idiopathic 0.02095 2/12
BgRatio
11 25/9703
17 31/9703
63 1/9703
109 25/9703
110 25/9703
113 1/9703
114 1/9703
123 1/9703
125 1/9703
106 50/9703WebGestalt enrichment analysis for genes with PIP>0.5
Loading the functional categories...
Loading the ID list...
Loading the reference list...
Performing the enrichment analysis...Warning in oraEnrichment(interestGeneList, referenceGeneList, geneSet, minNum =
minNum, : No significant gene set is identified based on FDR 0.05!NULLSensitivity, specificity and precision for silver standard genes
#number of genes in known annotations
print(length(known_annotations))[1] 130#number of genes in known annotations with imputed expression
print(sum(known_annotations %in% ctwas_gene_res$genename))[1] 60#significance threshold for TWAS
print(sig_thresh)[1] 4.588#number of ctwas genes
length(ctwas_genes)[1] 13#number of TWAS genes
length(twas_genes)[1] 83#show novel genes (ctwas genes with not in TWAS genes)
ctwas_gene_res[ctwas_gene_res$genename %in% novel_genes,report_cols] genename region_tag susie_pip mu2 PVE z num_eqtl
1089 RRN3 16_15 0.9222 24.23 0.0002714 -4.560 2
3376 CRHR1 17_27 1.0000 3865.12 0.0469552 3.362 1#sensitivity / recall
print(sensitivity) ctwas TWAS
0.03846 0.07692 #specificity
print(specificity) ctwas TWAS
0.9993 0.9934 #precision / PPV
print(precision) ctwas TWAS
0.3846 0.1205 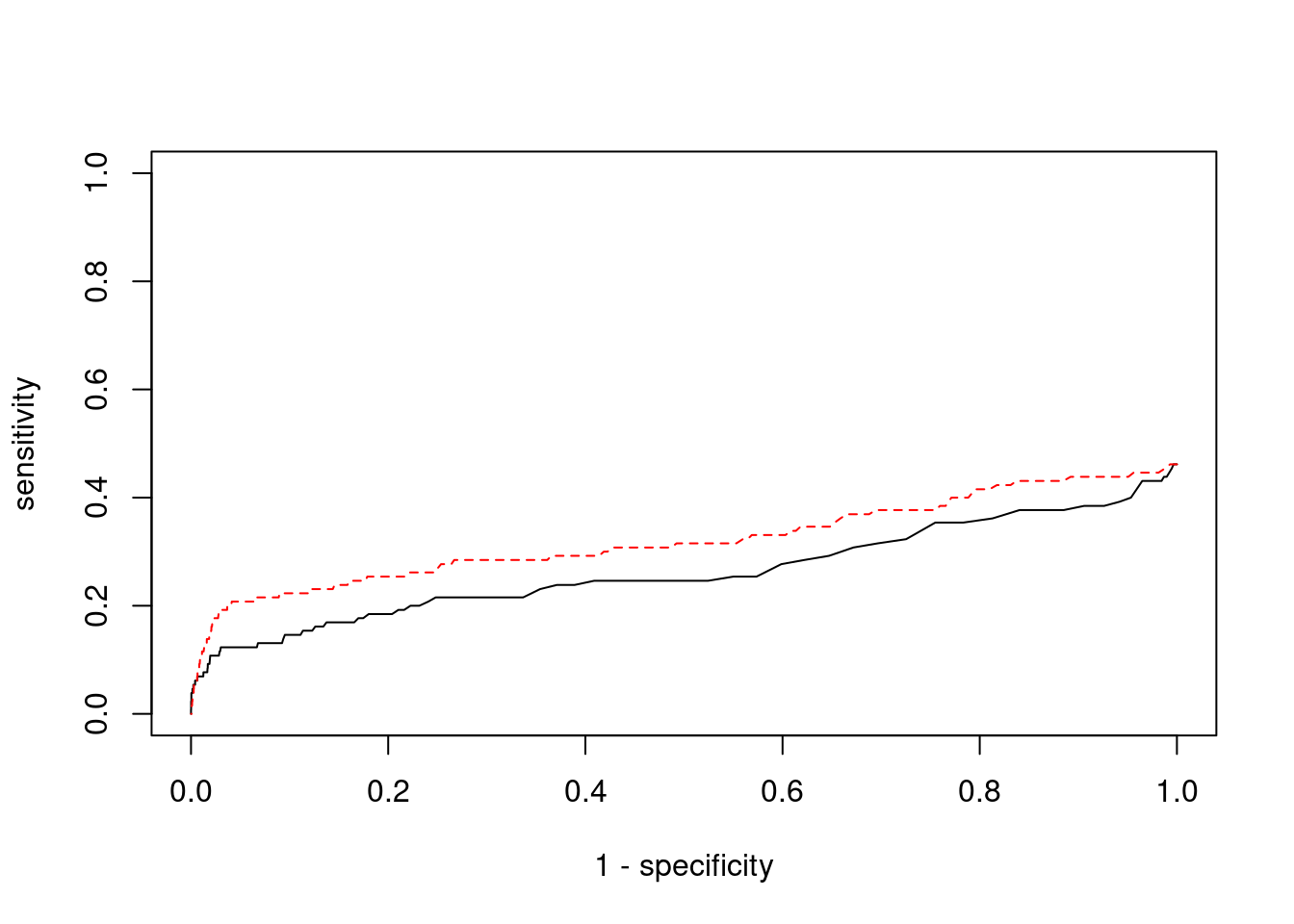
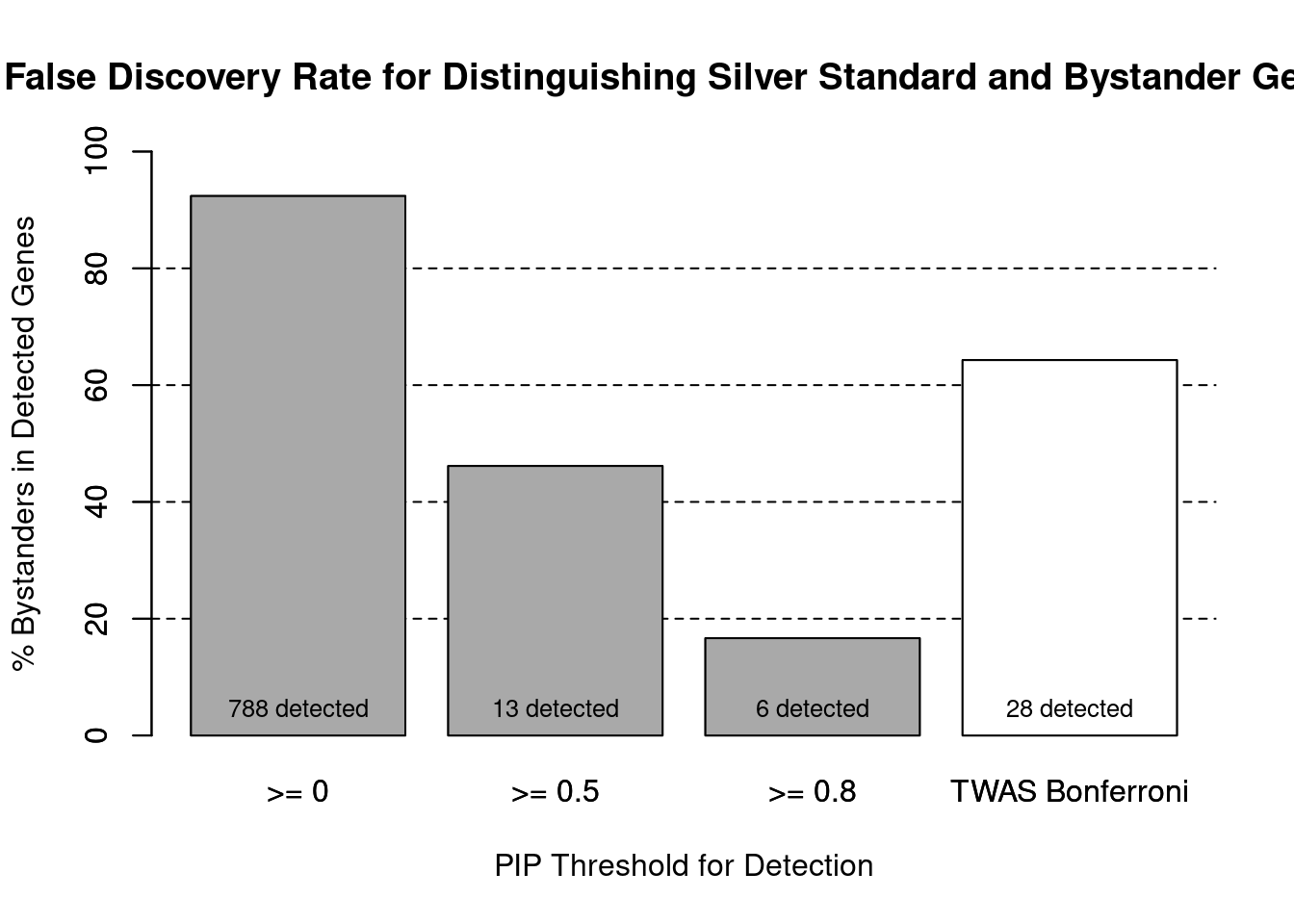
cTWAS is more precise than TWAS in distinguishing silver standard and bystander genes
#number of genes in known annotations (with imputed expression)
print(length(known_annotations))[1] 60#number of bystander genes (with imputed expression)
print(length(unrelated_genes))[1] 727#subset results to genes in known annotations or bystanders
ctwas_gene_res_subset <- ctwas_gene_res[ctwas_gene_res$genename %in% c(known_annotations, unrelated_genes),]
#assign ctwas and TWAS genes
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>0.8]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>sig_thresh]
#significance threshold for TWAS
print(sig_thresh)[1] 4.588#number of ctwas genes (in known annotations or bystanders)
length(ctwas_genes)[1] 6#number of TWAS genes (in known annotations or bystanders)
length(twas_genes)[1] 28#sensitivity / recall
sensitivity ctwas TWAS
0.08333 0.16667 #specificity / (1 - False Positive Rate)
specificity ctwas TWAS
0.9986 0.9752 #precision / PPV / (1 - False Discovery Rate)
precision ctwas TWAS
0.8333 0.3571 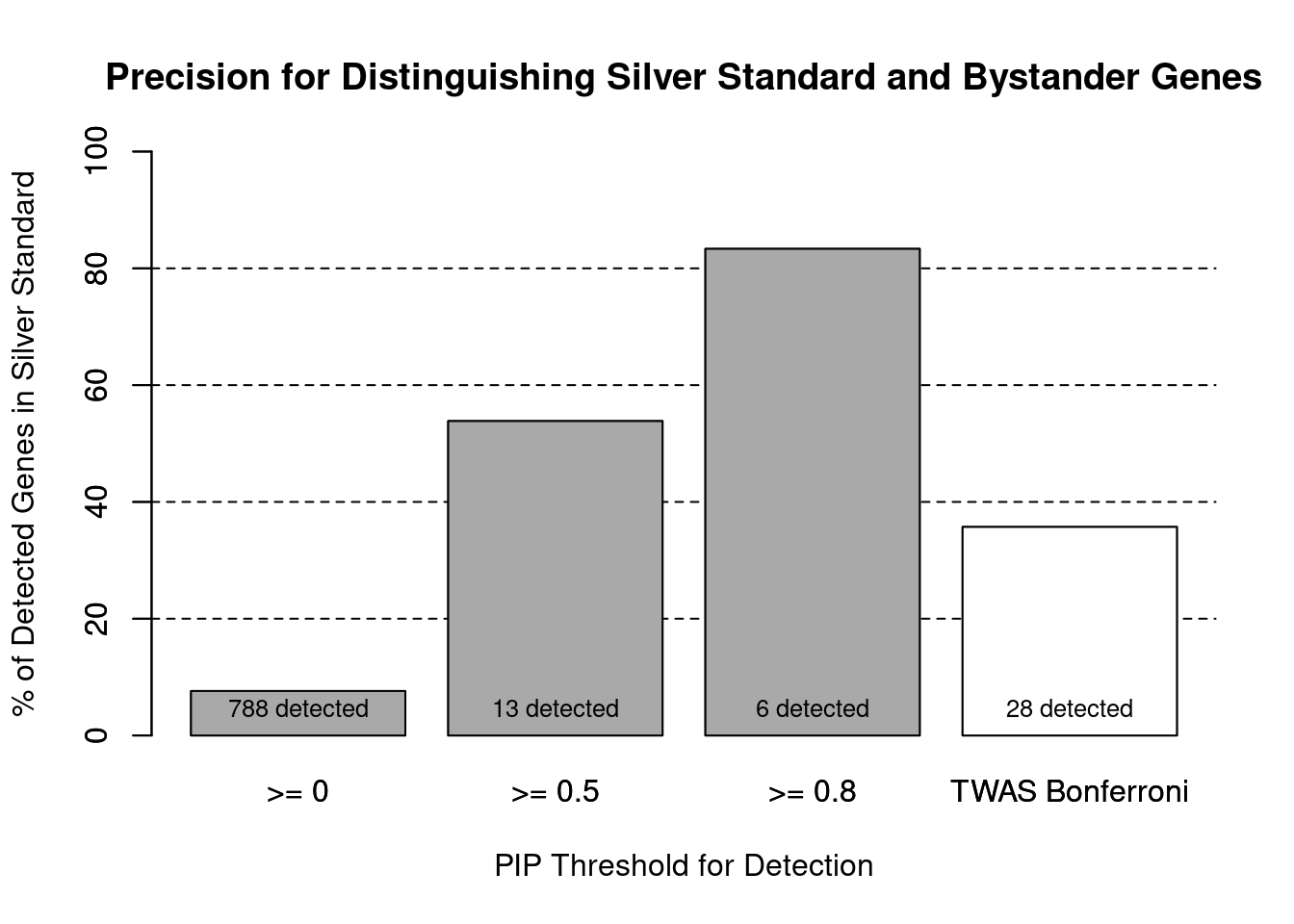
| Version | Author | Date |
|---|---|---|
| addb825 | sq-96 | 2022-02-28 |
| Version | Author | Date |
|---|---|---|
| addb825 | sq-96 | 2022-02-28 |
pip_range <- (0:1000)/1000
sensitivity <- rep(NA, length(pip_range))
specificity <- rep(NA, length(pip_range))
for (index in 1:length(pip_range)){
pip <- pip_range[index]
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>=pip]
sensitivity[index] <- sum(ctwas_genes %in% known_annotations)/length(known_annotations)
specificity[index] <- sum(!(unrelated_genes %in% ctwas_genes))/length(unrelated_genes)
}
plot(1-specificity, sensitivity, type="l", xlim=c(0,1), ylim=c(0,1), main="", xlab="1 - Specificity", ylab="Sensitivity")
title(expression("ROC Curve for cTWAS (black) and TWAS (" * phantom("red") * ")"))
title(expression(phantom("ROC Curve for cTWAS (black) and TWAS (") * "red" * phantom(")")), col.main="red")
sig_thresh_range <- seq(from=0, to=max(abs(ctwas_gene_res_subset$z)), length.out=length(pip_range))
for (index in 1:length(sig_thresh_range)){
sig_thresh_plot <- sig_thresh_range[index]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>=sig_thresh_plot]
sensitivity[index] <- sum(twas_genes %in% known_annotations)/length(known_annotations)
specificity[index] <- sum(!(unrelated_genes %in% twas_genes))/length(unrelated_genes)
}
lines(1-specificity, sensitivity, xlim=c(0,1), ylim=c(0,1), col="red", lty=1)
abline(a=0,b=1,lty=3)
#add previously computed points from the analysis
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>0.8]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>sig_thresh]
points(1-specificity_plot["ctwas"], sensitivity_plot["ctwas"], pch=21, bg="black")
points(1-specificity_plot["TWAS"], sensitivity_plot["TWAS"], pch=21, bg="red")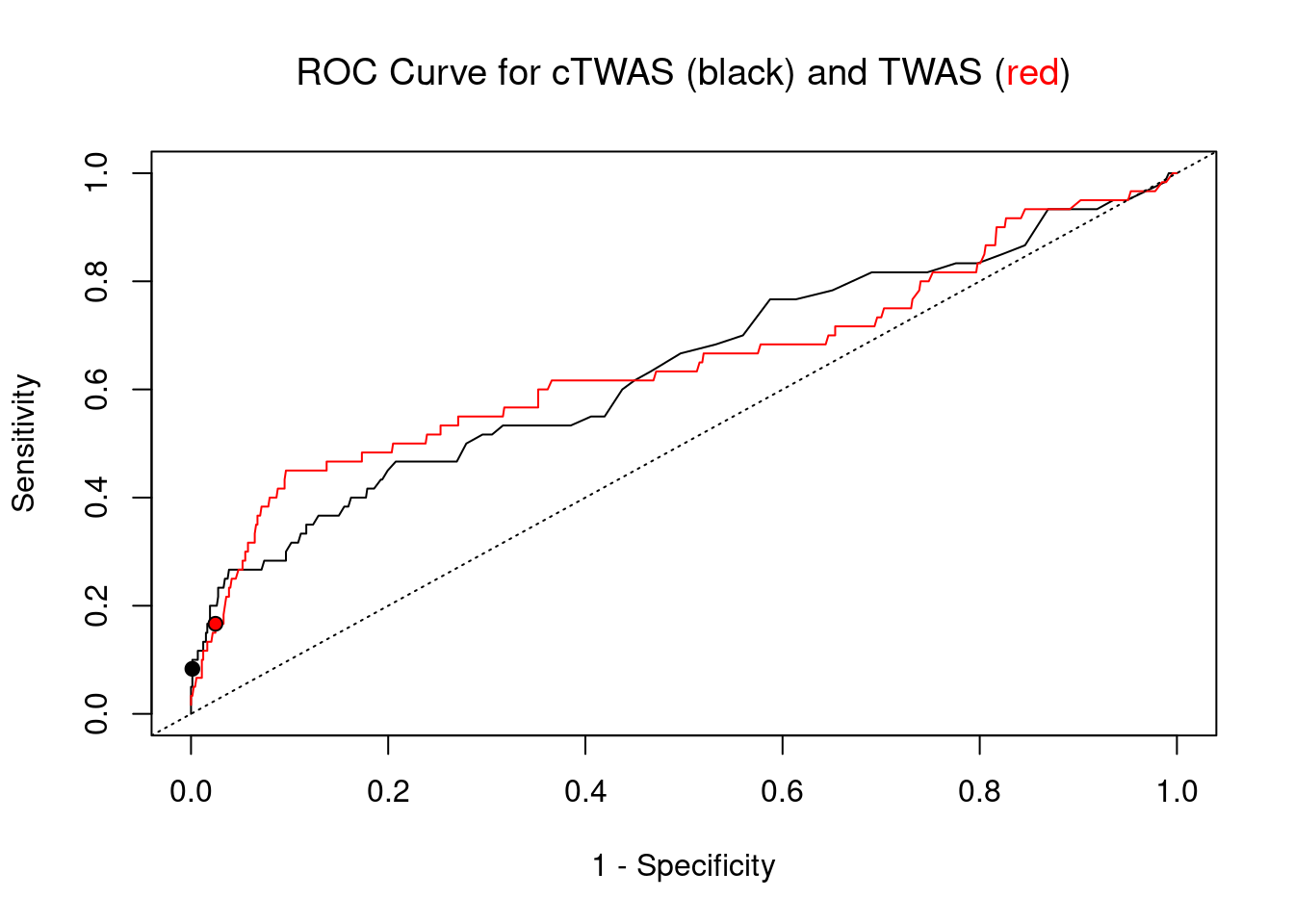
| Version | Author | Date |
|---|---|---|
| 4a5db1c | sq-96 | 2022-03-03 |
Undetected silver standard genes have low TWAS z-scores or stronger signal from nearby variants
#table of outcomes for silver standard genes
-sort(-table(silver_standard_case))silver_standard_case
Not Imputed Insignificant z-score Nearby SNP(s)
70 49 6
Detected (PIP > 0.8)
5 #show inconclusive genes
silver_standard_case[silver_standard_case=="Inconclusive"]named character(0)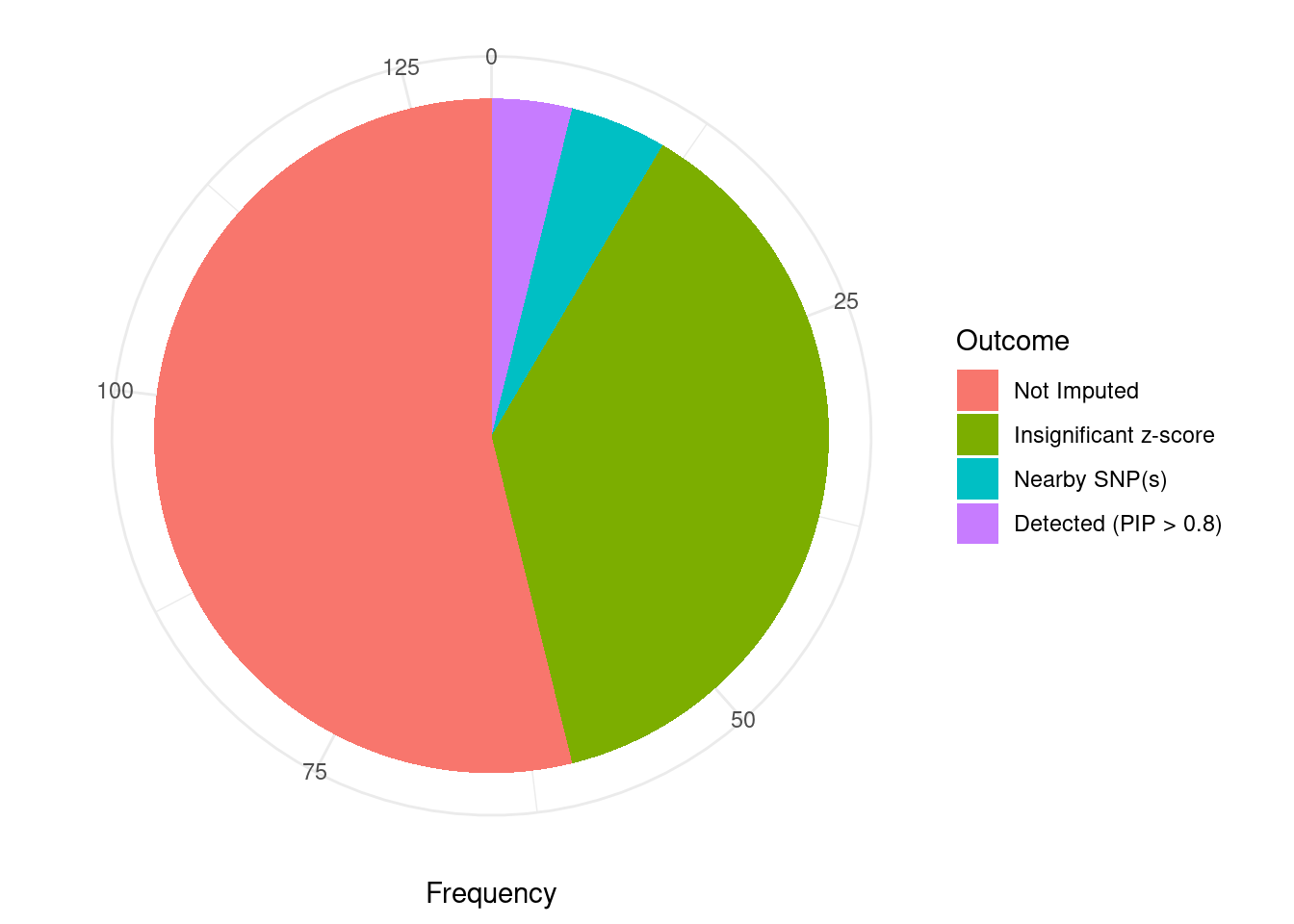
| Version | Author | Date |
|---|---|---|
| 4a5db1c | sq-96 | 2022-03-03 |
Locus plot for three silver standard genes that cTWAS identifies
Locus 17_27: In TWAS, no SNP or gene passes the threshold. But cTWAS is able to identify gene CRHR1 with high PIP.
locus_plot("17_27", label="TWAS")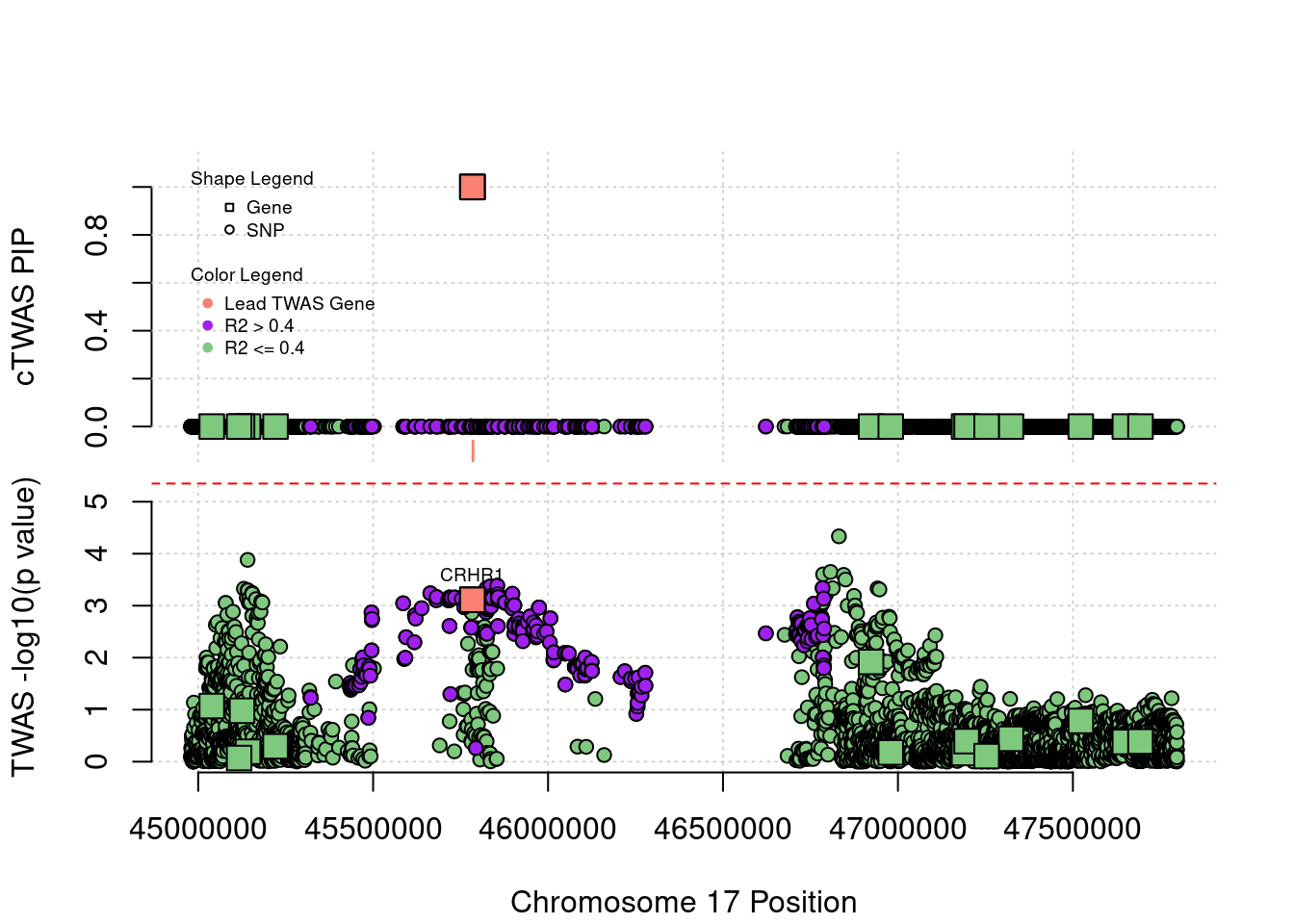
Locus 15_42: Known risk gene FURIN has very high PIP in this tissue
locus_plot("15_42", label="TWAS")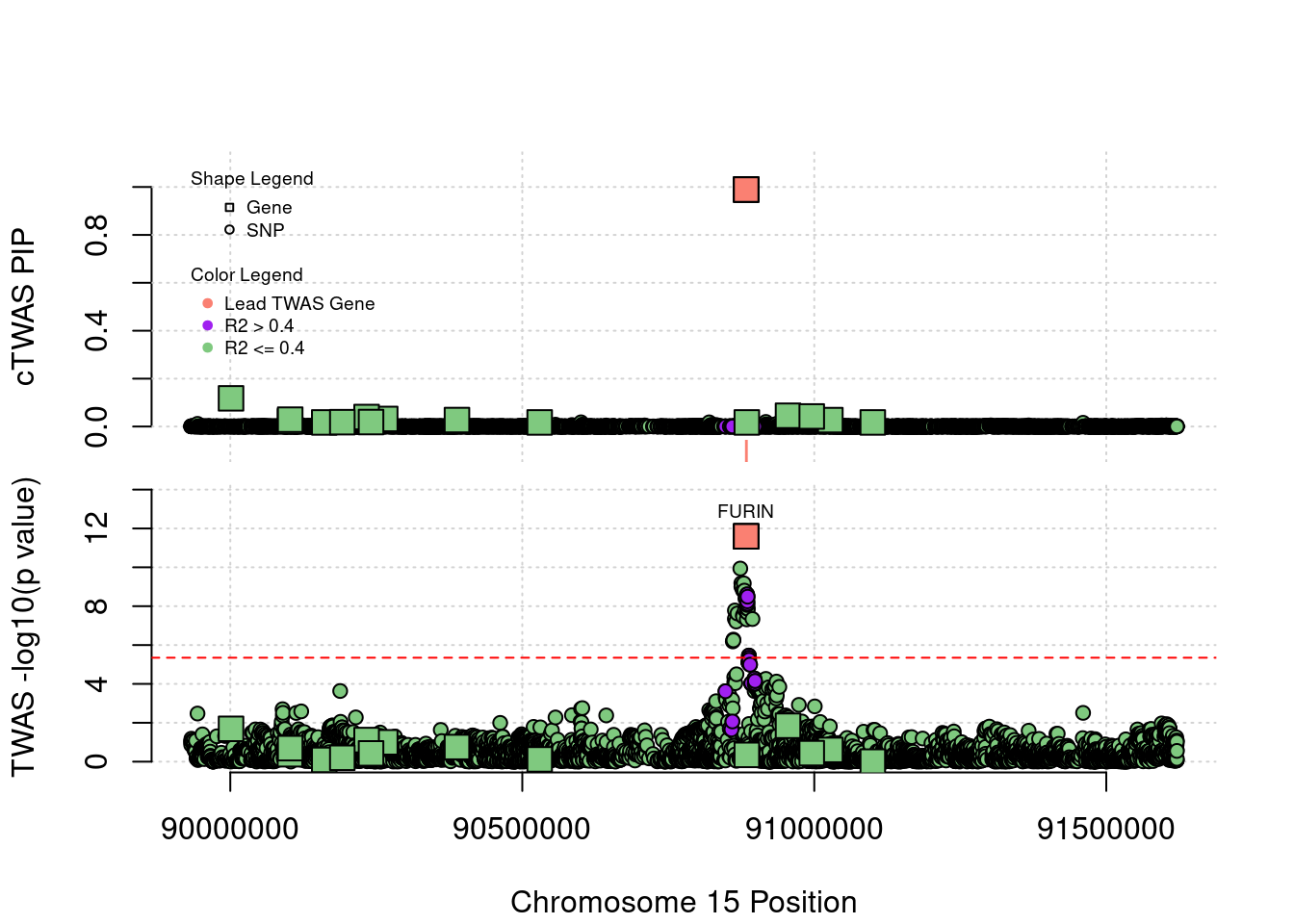
Locus 19_10: Both TWAS and cTWAS work well in this locus.
locus_plot("19_10", label="TWAS")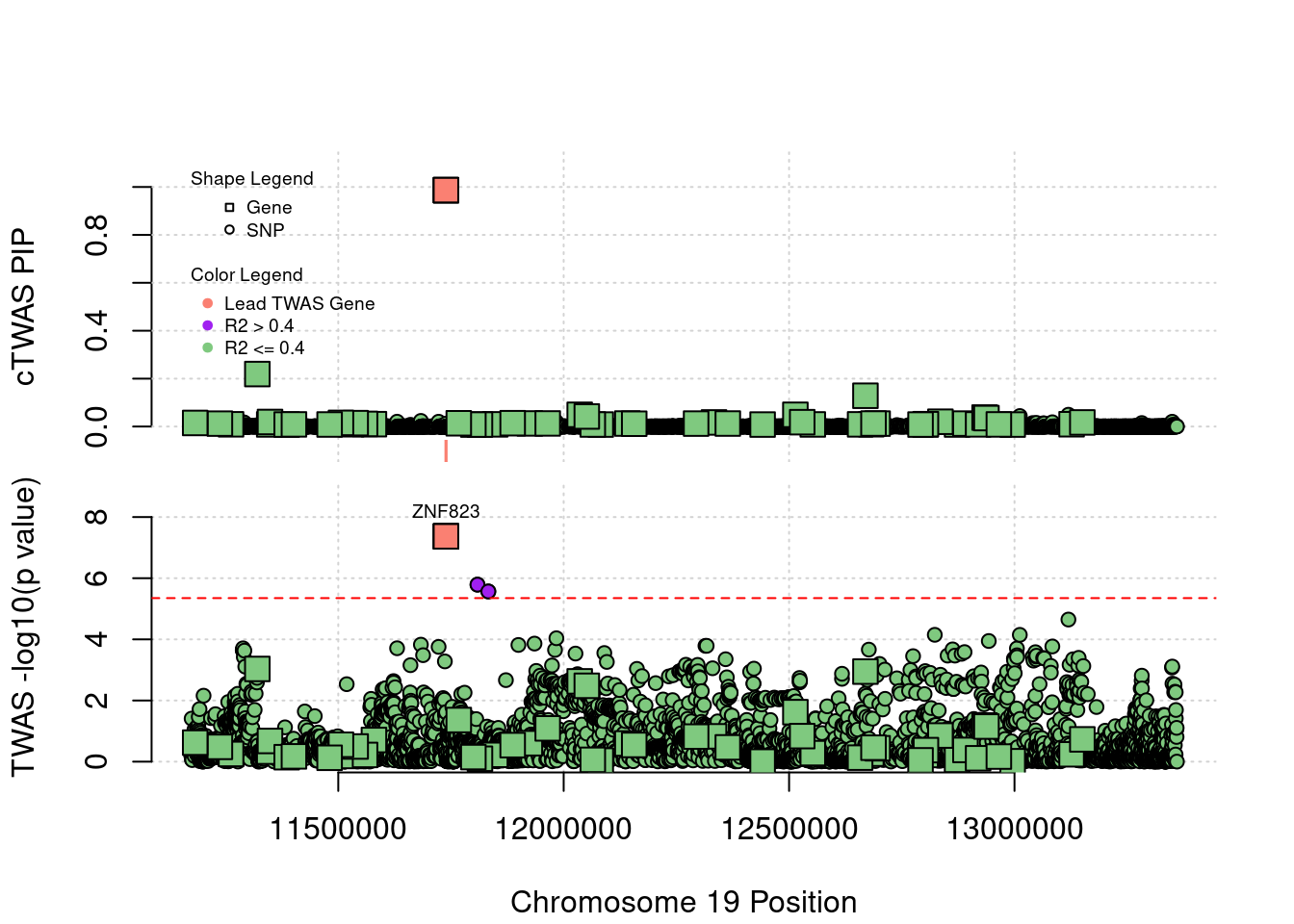
Locus 3_43: The output of cTWAS is very clean in this locus.
locus_plot("3_43", label="TWAS")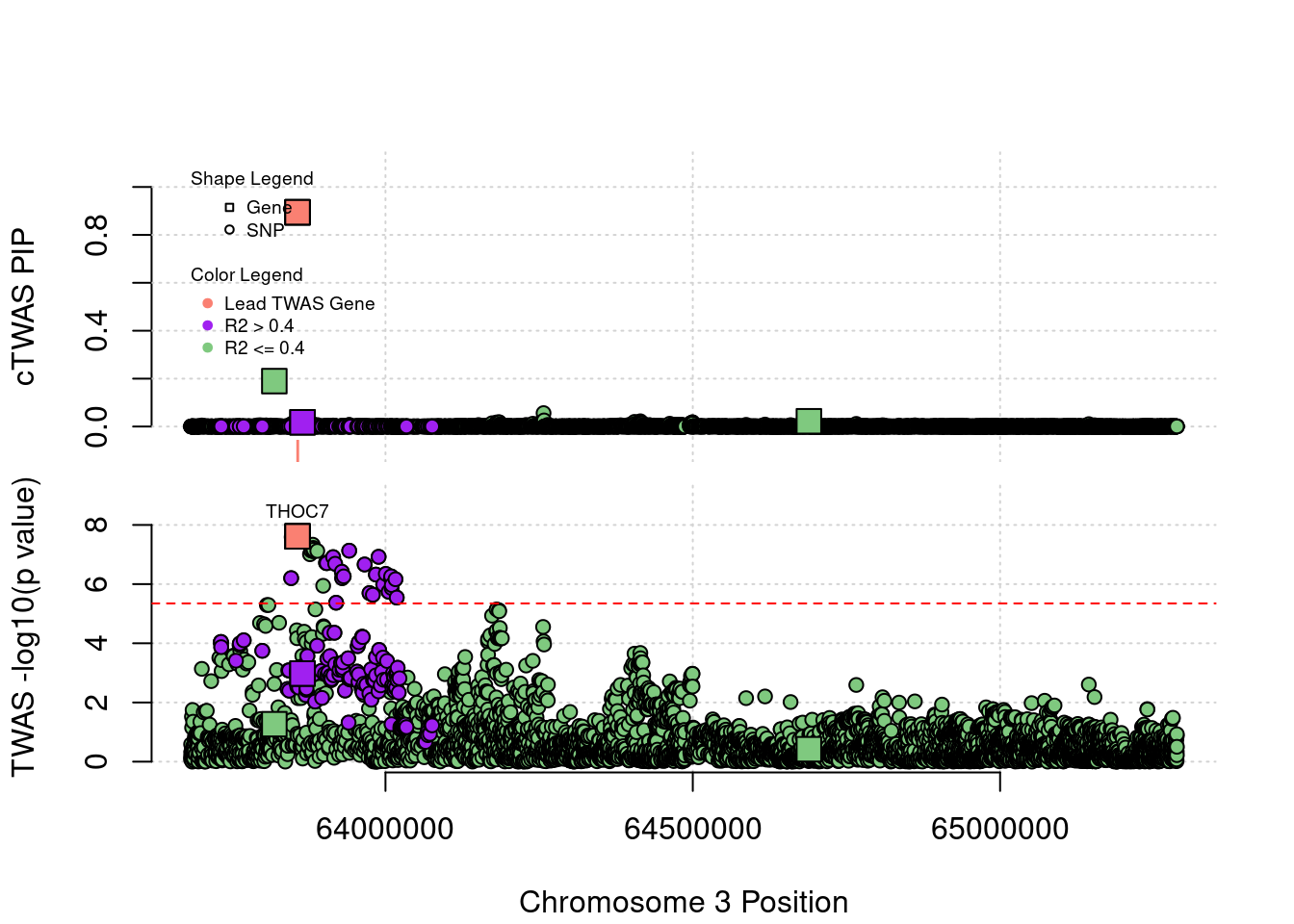
Locus 2_117: The output of cTWAS is very clean in this locus.
locus_plot("2_117", label="TWAS")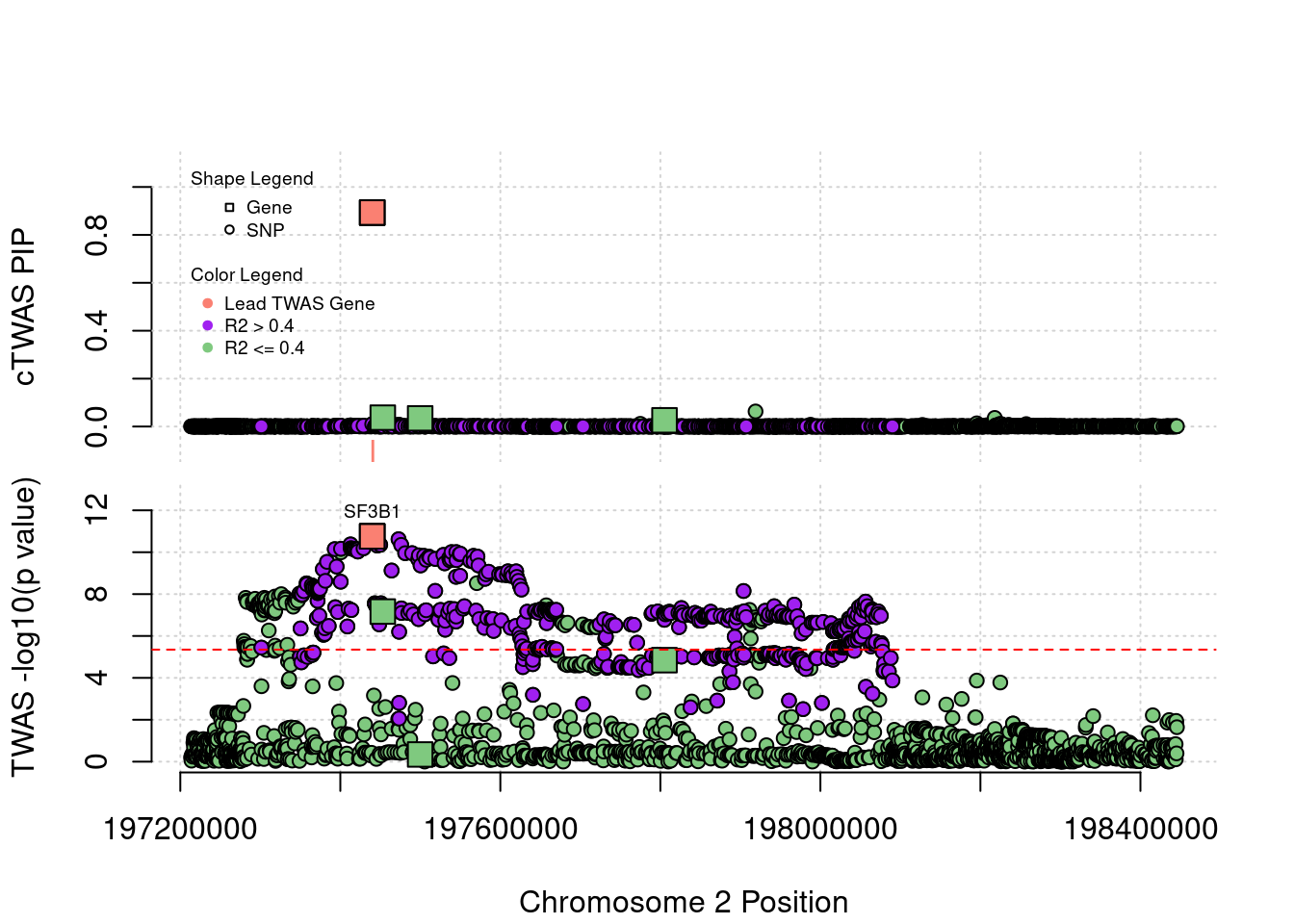
Some know SCZ risk genes that cTWAS does not find
Locus 19_35: Gene IRF3 is around the threshold in TWAS. There is another gene in the right hand side getting higher PIP.
locus_plot5("19_35", focus="IRF3")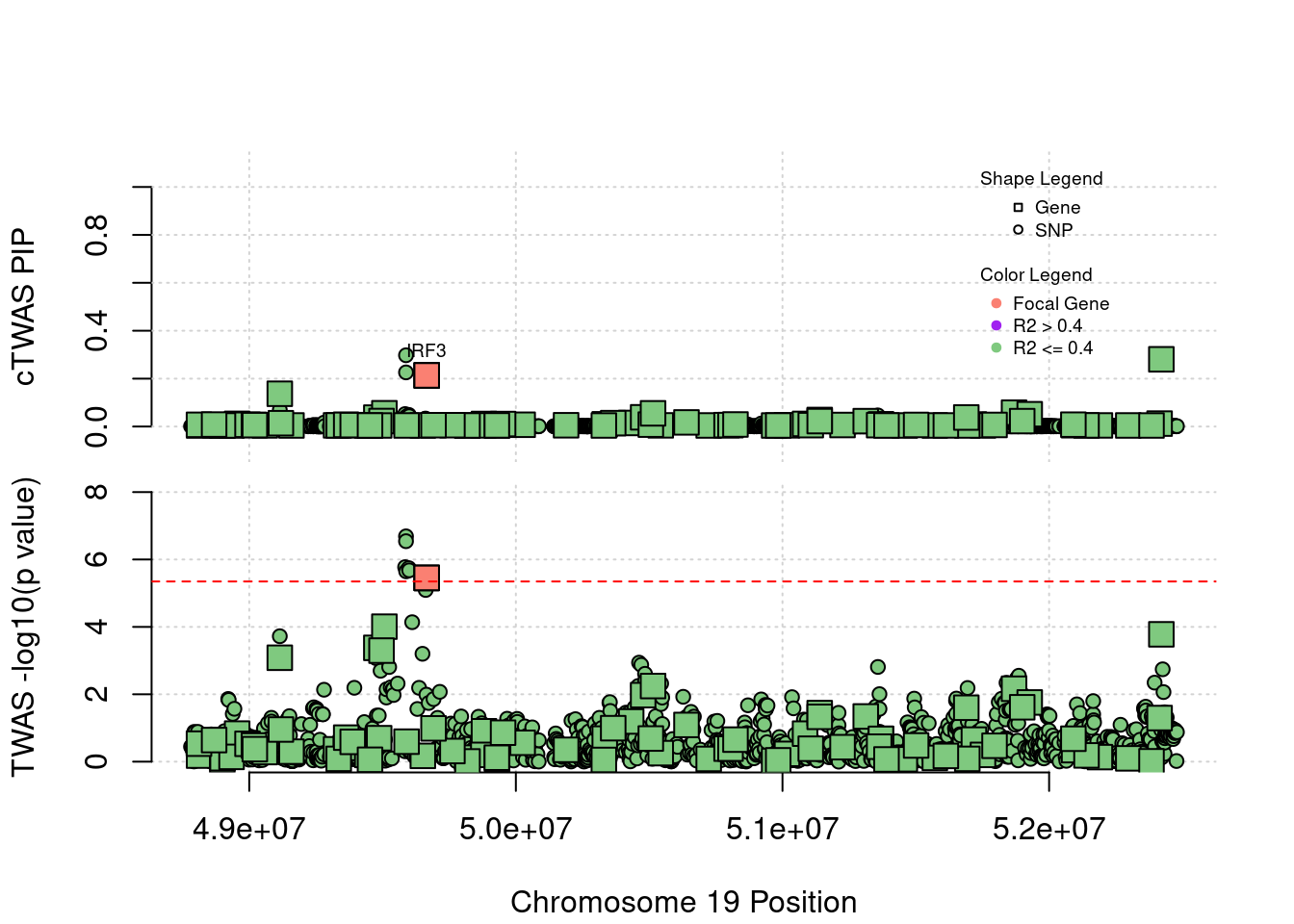
Locus 1_6: The neurodevelopmental disorder gene RERE is not imputed in this tissue.
Locus 17_38: ACE encoding angiotensin converting enzyme, the target of a major class of anti-hypertensive drugs (schizophrenia under expression), has insignificant z-score
locus_plot5("17_38", focus="ACE")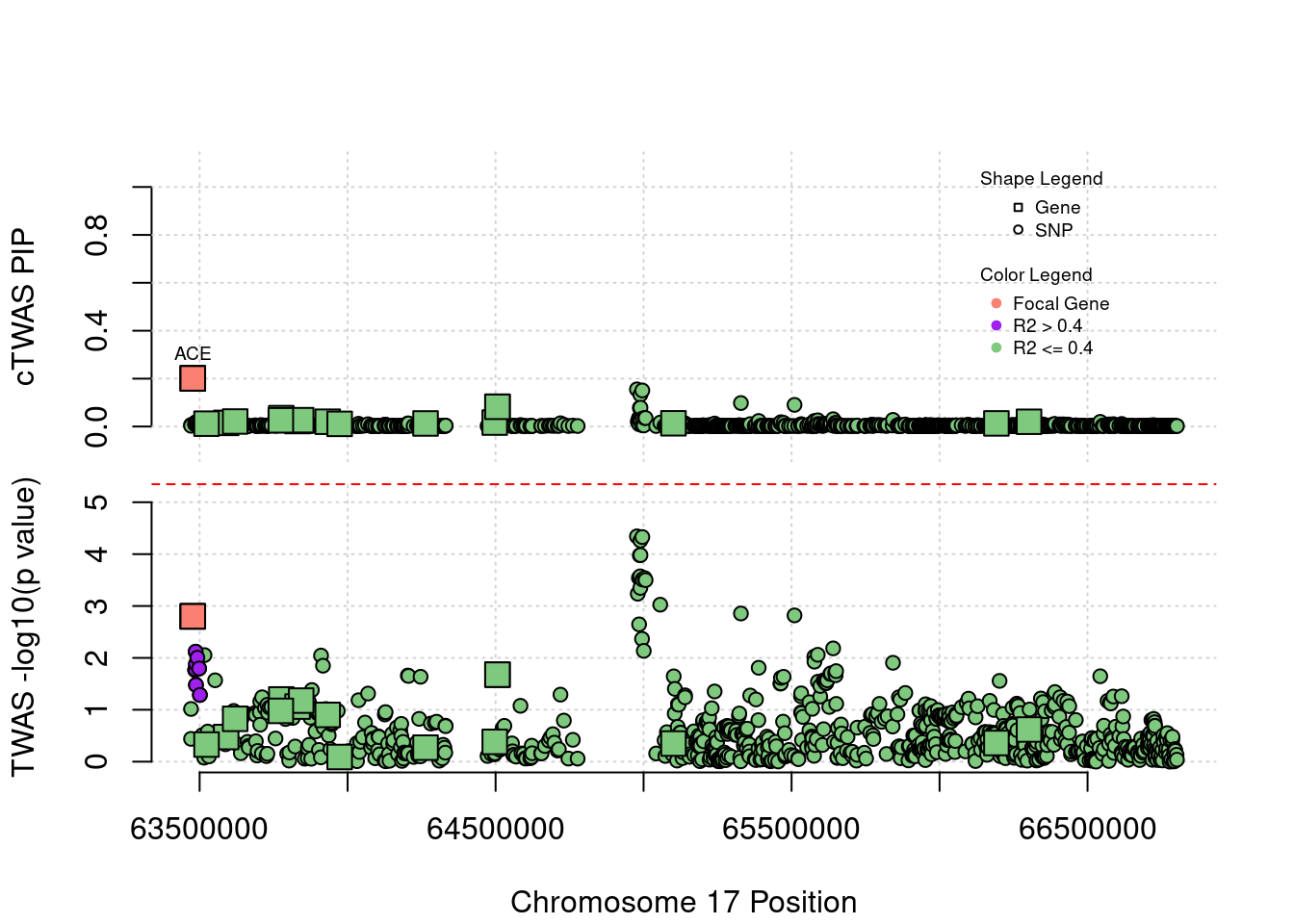
Locus 3_27: DCLK3 encoding a neuroprotective kinase (schizophrenia under-expression), has insignificant z-score. No gene in this locus has high PIP or z-score
locus_plot("3_27", label="TWAS")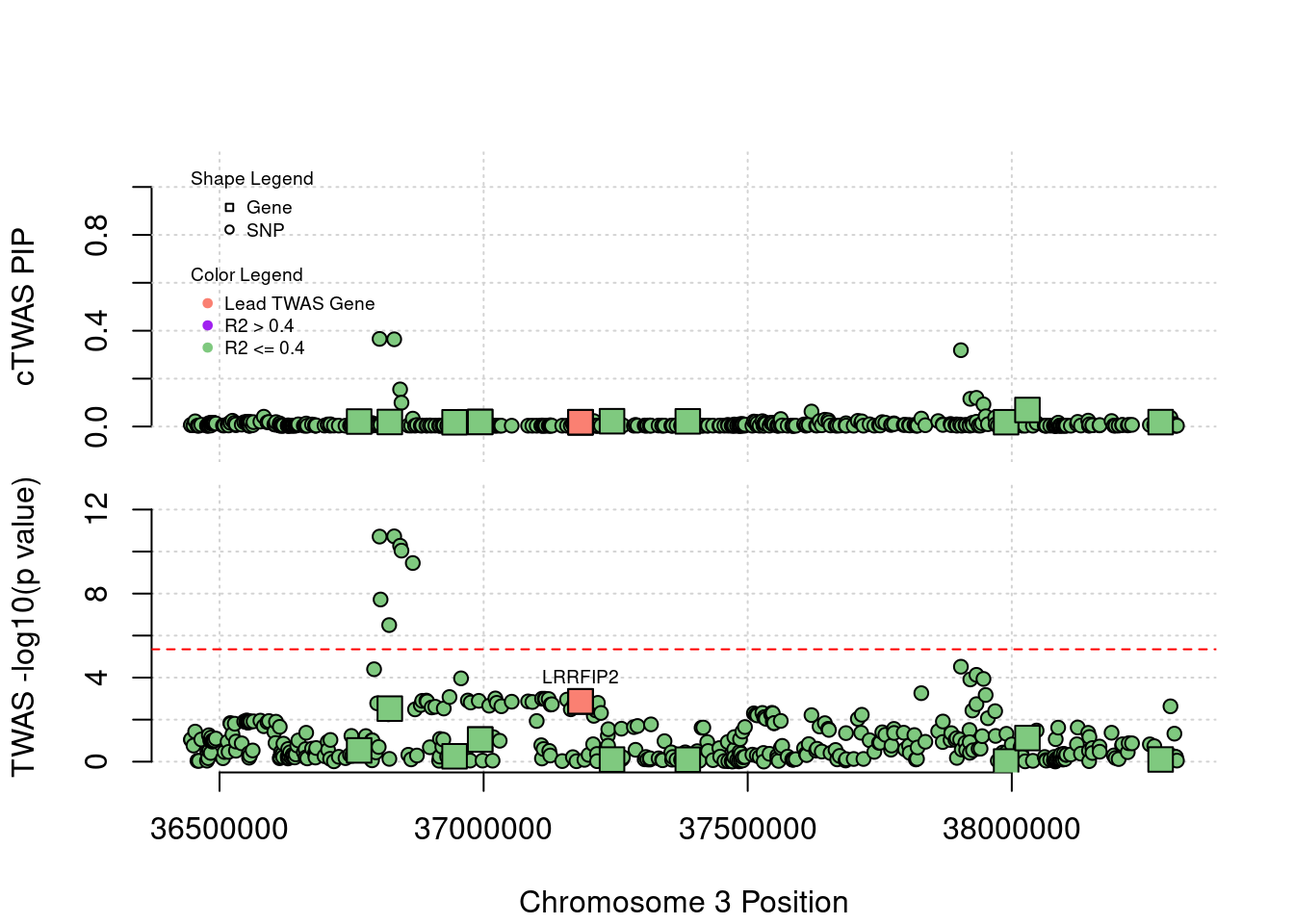
sessionInfo()R version 3.6.1 (2019-07-05)
Platform: x86_64-pc-linux-gnu (64-bit)
Running under: Scientific Linux 7.4 (Nitrogen)
Matrix products: default
BLAS/LAPACK: /software/openblas-0.2.19-el7-x86_64/lib/libopenblas_haswellp-r0.2.19.so
locale:
[1] LC_CTYPE=en_US.UTF-8 LC_NUMERIC=C
[3] LC_TIME=en_US.UTF-8 LC_COLLATE=en_US.UTF-8
[5] LC_MONETARY=en_US.UTF-8 LC_MESSAGES=en_US.UTF-8
[7] LC_PAPER=en_US.UTF-8 LC_NAME=C
[9] LC_ADDRESS=C LC_TELEPHONE=C
[11] LC_MEASUREMENT=en_US.UTF-8 LC_IDENTIFICATION=C
attached base packages:
[1] stats graphics grDevices utils datasets methods base
other attached packages:
[1] readxl_1.3.1 forcats_0.5.1 stringr_1.4.0 dplyr_1.0.7
[5] purrr_0.3.4 readr_2.1.1 tidyr_1.1.4 tidyverse_1.3.1
[9] tibble_3.1.6 WebGestaltR_0.4.4 disgenet2r_0.99.2 enrichR_3.0
[13] cowplot_1.0.0 ggplot2_3.3.5 workflowr_1.7.0
loaded via a namespace (and not attached):
[1] fs_1.5.2 lubridate_1.8.0 bit64_4.0.5 doParallel_1.0.17
[5] httr_1.4.2 rprojroot_2.0.2 tools_3.6.1 backports_1.4.1
[9] doRNG_1.8.2 utf8_1.2.2 R6_2.5.1 vipor_0.4.5
[13] DBI_1.1.2 colorspace_2.0-2 withr_2.4.3 ggrastr_1.0.1
[17] tidyselect_1.1.1 processx_3.5.2 bit_4.0.4 curl_4.3.2
[21] compiler_3.6.1 git2r_0.26.1 rvest_1.0.2 cli_3.1.0
[25] Cairo_1.5-12.2 xml2_1.3.3 labeling_0.4.2 scales_1.1.1
[29] callr_3.7.0 apcluster_1.4.8 digest_0.6.29 rmarkdown_2.11
[33] svglite_1.2.2 pkgconfig_2.0.3 htmltools_0.5.2 dbplyr_2.1.1
[37] fastmap_1.1.0 highr_0.9 rlang_1.0.1 rstudioapi_0.13
[41] RSQLite_2.2.8 jquerylib_0.1.4 farver_2.1.0 generics_0.1.1
[45] jsonlite_1.7.2 vroom_1.5.7 magrittr_2.0.2 Matrix_1.2-18
[49] ggbeeswarm_0.6.0 Rcpp_1.0.8 munsell_0.5.0 fansi_1.0.2
[53] gdtools_0.1.9 lifecycle_1.0.1 stringi_1.7.6 whisker_0.3-2
[57] yaml_2.2.1 plyr_1.8.6 grid_3.6.1 blob_1.2.2
[61] ggrepel_0.9.1 parallel_3.6.1 promises_1.0.1 crayon_1.5.0
[65] lattice_0.20-38 haven_2.4.3 hms_1.1.1 knitr_1.36
[69] ps_1.6.0 pillar_1.6.4 igraph_1.2.10 rjson_0.2.20
[73] rngtools_1.5.2 reshape2_1.4.4 codetools_0.2-16 reprex_2.0.1
[77] glue_1.6.2 evaluate_0.14 getPass_0.2-2 modelr_0.1.8
[81] data.table_1.14.2 vctrs_0.3.8 tzdb_0.2.0 httpuv_1.5.1
[85] foreach_1.5.2 cellranger_1.1.0 gtable_0.3.0 assertthat_0.2.1
[89] cachem_1.0.6 xfun_0.29 broom_0.7.10 later_0.8.0
[93] iterators_1.0.14 beeswarm_0.2.3 memoise_2.0.1 ellipsis_0.3.2