SCZ - Brain Hippocampus
sheng Qian
2021-2-6
Last updated: 2022-03-14
Checks: 5 2
Knit directory: cTWAS_analysis/
This reproducible R Markdown analysis was created with workflowr (version 1.7.0). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
The R Markdown is untracked by Git. To know which version of the R Markdown file created these results, you’ll want to first commit it to the Git repo. If you’re still working on the analysis, you can ignore this warning. When you’re finished, you can run wflow_publish to commit the R Markdown file and build the HTML.
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
The command set.seed(20211220) was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
Nice! There were no cached chunks for this analysis, so you can be confident that you successfully produced the results during this run.
Using absolute paths to the files within your workflowr project makes it difficult for you and others to run your code on a different machine. Change the absolute path(s) below to the suggested relative path(s) to make your code more reproducible.
| absolute | relative |
|---|---|
| /project2/xinhe/shengqian/cTWAS/cTWAS_analysis/data/ | data |
| /project2/xinhe/shengqian/cTWAS/cTWAS_analysis/code/ctwas_config.R | code/ctwas_config.R |
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility.
The results in this page were generated with repository version 4c71b11. See the Past versions tab to see a history of the changes made to the R Markdown and HTML files.
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can use wflow_publish or wflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Ignored files:
Ignored: .ipynb_checkpoints/
Ignored: data/AF/
Untracked files:
Untracked: Rplot.png
Untracked: analysis/.ipynb_checkpoints/
Untracked: analysis/SCZ_2014_EUR_Brain_Amygdala.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Anterior_cingulate_cortex_BA24.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Caudate_basal_ganglia.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Cerebellar_Hemisphere.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Cerebellum.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Cortex.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Frontal_Cortex_BA9.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Hippocampus.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Hypothalamus.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Nucleus_accumbens_basal_ganglia.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Putamen_basal_ganglia.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Spinal_cord_cervical_c-1.Rmd
Untracked: analysis/SCZ_2014_EUR_Brain_Substantia_nigra.Rmd
Untracked: analysis/SCZ_2020_Brain_Cortex.Rmd
Untracked: analysis/SCZ_2020_Brain_Frontal_Cortex_BA9.Rmd
Untracked: analysis/SCZ_2020_Brain_Hypothalamus.Rmd
Untracked: analysis/SCZ_2020_Brain_Putamen_basal_ganglia.Rmd
Untracked: analysis/SCZ_Cross_Tissue_Analysis.Rmd
Untracked: code/.ipynb_checkpoints/
Untracked: code/AF_out/
Untracked: code/Autism_out/
Untracked: code/BMI_S_out/
Untracked: code/BMI_out/
Untracked: code/Glucose_out/
Untracked: code/LDL_S_out/
Untracked: code/SCZ_2014_EUR_out/
Untracked: code/SCZ_2020_out/
Untracked: code/SCZ_S_out/
Untracked: code/SCZ_out/
Untracked: code/T2D_out/
Untracked: code/ctwas_config.R
Untracked: code/mapping.R
Untracked: code/out/
Untracked: code/run_AF_analysis.sbatch
Untracked: code/run_AF_analysis.sh
Untracked: code/run_AF_ctwas_rss_LDR.R
Untracked: code/run_Autism_analysis.sbatch
Untracked: code/run_Autism_analysis.sh
Untracked: code/run_Autism_ctwas_rss_LDR.R
Untracked: code/run_BMI_analysis.sbatch
Untracked: code/run_BMI_analysis.sh
Untracked: code/run_BMI_analysis_S.sbatch
Untracked: code/run_BMI_analysis_S.sh
Untracked: code/run_BMI_ctwas_rss_LDR.R
Untracked: code/run_BMI_ctwas_rss_LDR_S.R
Untracked: code/run_Glucose_analysis.sbatch
Untracked: code/run_Glucose_analysis.sh
Untracked: code/run_Glucose_ctwas_rss_LDR.R
Untracked: code/run_LDL_analysis_S.sbatch
Untracked: code/run_LDL_analysis_S.sh
Untracked: code/run_LDL_ctwas_rss_LDR_S.R
Untracked: code/run_SCZ_2014_EUR_analysis.sbatch
Untracked: code/run_SCZ_2014_EUR_analysis.sh
Untracked: code/run_SCZ_2014_EUR_ctwas_rss_LDR.R
Untracked: code/run_SCZ_2020_analysis.sbatch
Untracked: code/run_SCZ_2020_analysis.sh
Untracked: code/run_SCZ_2020_ctwas_rss_LDR.R
Untracked: code/run_SCZ_analysis.sbatch
Untracked: code/run_SCZ_analysis.sh
Untracked: code/run_SCZ_analysis_S.sbatch
Untracked: code/run_SCZ_analysis_S.sh
Untracked: code/run_SCZ_ctwas_rss_LDR.R
Untracked: code/run_SCZ_ctwas_rss_LDR_S.R
Untracked: code/run_T2D_analysis.sbatch
Untracked: code/run_T2D_analysis.sh
Untracked: code/run_T2D_ctwas_rss_LDR.R
Untracked: code/wflow_build.R
Untracked: code/wflow_build.sbatch
Untracked: data/.ipynb_checkpoints/
Untracked: data/BMI/
Untracked: data/PGC3_SCZ_wave3_public.v2.tsv
Untracked: data/SCZ/
Untracked: data/SCZ_2014_EUR/
Untracked: data/SCZ_2020/
Untracked: data/SCZ_S/
Untracked: data/T2D/
Untracked: data/UKBB/
Untracked: data/UKBB_SNPs_Info.text
Untracked: data/gene_OMIM.txt
Untracked: data/gene_pip_0.8.txt
Untracked: data/mashr_Heart_Atrial_Appendage.db
Untracked: data/mashr_sqtl/
Untracked: data/summary_known_genes_annotations.xlsx
Untracked: data/untitled.txt
Unstaged changes:
Modified: analysis/SCZ_Brain_Amygdala.Rmd
Modified: analysis/SCZ_Brain_Anterior_cingulate_cortex_BA24.Rmd
Modified: analysis/SCZ_Brain_Caudate_basal_ganglia.Rmd
Modified: analysis/SCZ_Brain_Cerebellar_Hemisphere.Rmd
Modified: analysis/SCZ_Brain_Cerebellum.Rmd
Modified: analysis/SCZ_Brain_Cortex.Rmd
Modified: analysis/SCZ_Brain_Frontal_Cortex_BA9.Rmd
Modified: analysis/SCZ_Brain_Hippocampus.Rmd
Modified: analysis/SCZ_Brain_Hypothalamus.Rmd
Modified: analysis/SCZ_Brain_Nucleus_accumbens_basal_ganglia.Rmd
Modified: analysis/SCZ_Brain_Putamen_basal_ganglia.Rmd
Modified: analysis/SCZ_Brain_Spinal_cord_cervical_c-1.Rmd
Modified: analysis/SCZ_Brain_Substantia_nigra.Rmd
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
There are no past versions. Publish this analysis with wflow_publish() to start tracking its development.
Weight QC
#number of imputed weights
nrow(qclist_all)[1] 10654#number of imputed weights by chromosome
table(qclist_all$chr)
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
1035 753 624 411 532 604 502 391 402 420 627 592 214 352 363 495
17 18 19 20 21 22
649 161 821 318 114 274 #number of imputed weights without missing variants
sum(qclist_all$nmiss==0)[1] 8204#proportion of imputed weights without missing variants
mean(qclist_all$nmiss==0)[1] 0.77Check convergence of parameters
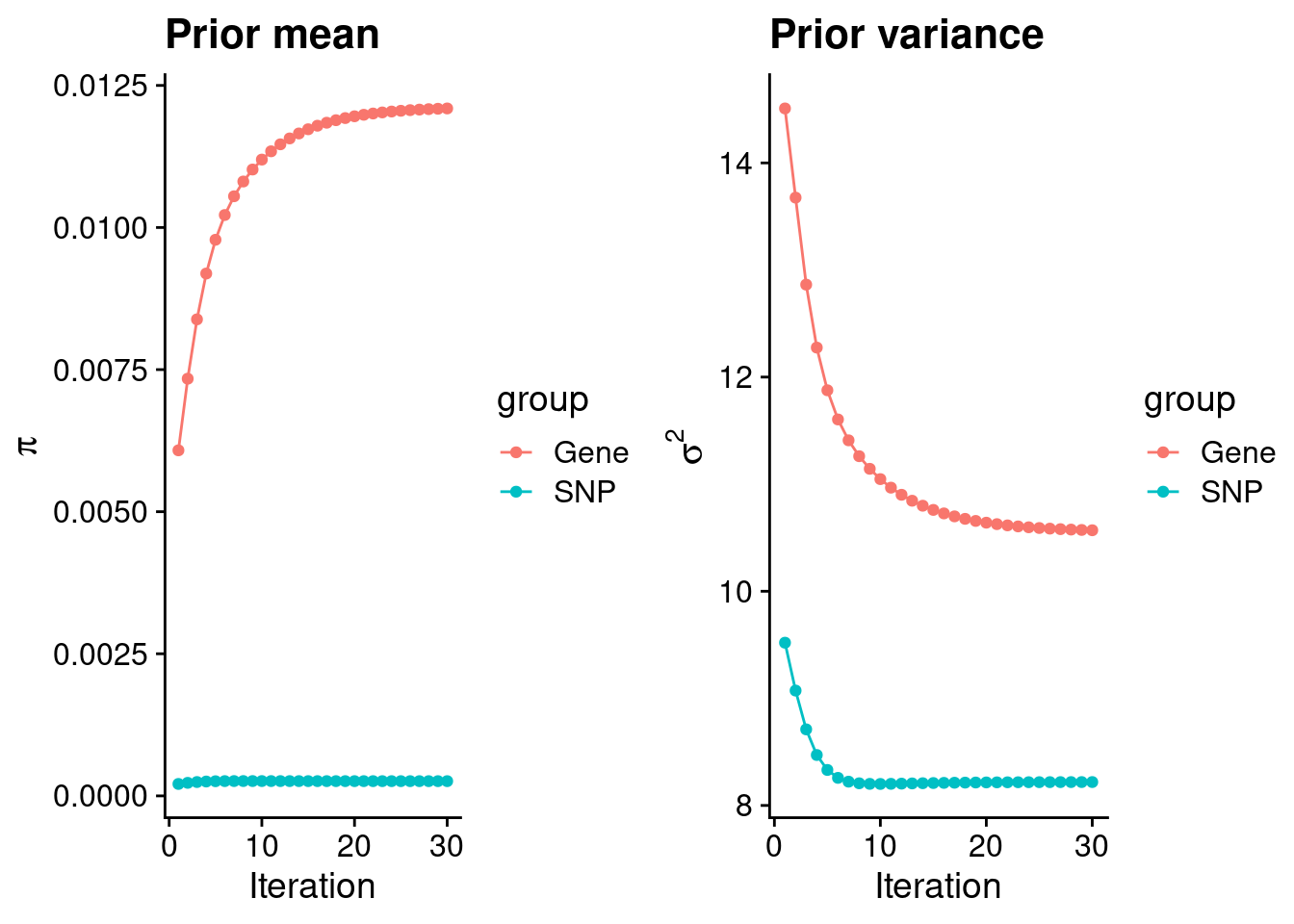
#estimated group prior
estimated_group_prior <- group_prior_rec[,ncol(group_prior_rec)]
names(estimated_group_prior) <- c("gene", "snp")
estimated_group_prior["snp"] <- estimated_group_prior["snp"]*thin #adjust parameter to account for thin argument
print(estimated_group_prior) gene snp
0.012095 0.000259 #estimated group prior variance
estimated_group_prior_var <- group_prior_var_rec[,ncol(group_prior_var_rec)]
names(estimated_group_prior_var) <- c("gene", "snp")
print(estimated_group_prior_var) gene snp
10.570 8.219 #report sample size
print(sample_size)[1] 77096#report group size
group_size <- c(nrow(ctwas_gene_res), n_snps)
print(group_size)[1] 10654 7352670#estimated group PVE
estimated_group_pve <- estimated_group_prior_var*estimated_group_prior*group_size/sample_size #check PVE calculation
names(estimated_group_pve) <- c("gene", "snp")
print(estimated_group_pve) gene snp
0.01767 0.20297 #compare sum(PIP*mu2/sample_size) with above PVE calculation
c(sum(ctwas_gene_res$PVE),sum(ctwas_snp_res$PVE))[1] 0.07701 1.77546Genes with highest PIPs
genename region_tag susie_pip mu2 PVE z num_eqtl
10687 ZNF823 19_10 0.9809 29.59 0.0003764 5.485 1
6084 ARFGAP2 11_29 0.9434 25.73 0.0003148 4.839 1
8759 MAP3K11 11_36 0.8669 22.56 0.0002537 -4.409 1
7929 ENDOG 9_66 0.8524 23.46 0.0002593 4.814 2
104 ELAC2 17_11 0.7884 22.09 0.0002259 4.372 1
4009 SPECC1 17_16 0.7772 22.05 0.0002223 4.167 1
12742 TBC1D29 17_18 0.7667 22.68 0.0002255 4.504 2
10621 SLC28A3 9_42 0.7388 20.69 0.0001982 3.743 1
10075 TMEM222 1_19 0.7030 23.44 0.0002137 3.902 1
2469 TBC1D19 4_22 0.7018 21.22 0.0001932 4.178 1
1516 TTLL1 22_18 0.6929 23.38 0.0002101 -4.510 2
3506 TBC1D15 12_44 0.6802 44.94 0.0003965 4.584 2
424 FAM120A 9_47 0.6753 23.25 0.0002037 4.571 1
1736 PPP1R16B 20_23 0.6717 34.97 0.0003047 6.009 1
2981 KCNJ13 2_137 0.6658 37.90 0.0003273 6.658 1
6538 MOV10 1_69 0.6539 22.16 0.0001879 -4.165 2
8275 INO80E 16_24 0.6509 38.24 0.0003229 6.230 1
3607 BHLHE41 12_18 0.6378 24.96 0.0002065 -3.860 1
2916 LMAN2L 2_57 0.6234 26.42 0.0002136 -4.473 2
9119 LY6H 8_94 0.5960 21.15 0.0001635 4.118 1Genes with largest effect sizes
genename region_tag susie_pip mu2 PVE z num_eqtl
11759 HIST1H2BN 6_21 9.808e-07 988.31 1.257e-08 10.7729 1
12967 RP1-153G14.4 6_21 0.000e+00 545.34 0.000e+00 1.8537 1
13065 RP1-86C11.7 6_21 2.000e-12 428.96 1.113e-14 -9.0332 1
10999 VWA7 6_27 6.840e-05 157.52 1.398e-07 8.9114 1
3217 STAG1 3_84 0.000e+00 154.32 0.000e+00 3.7399 2
10508 HIST1H2AG 6_21 0.000e+00 151.61 0.000e+00 1.6735 1
12063 C4A 6_27 9.696e-09 130.50 1.641e-11 8.3522 2
11006 APOM 6_27 1.374e-08 125.29 2.232e-11 7.8900 2
6779 ZSCAN12 6_22 4.256e-02 125.06 6.903e-05 10.9401 1
2147 MPP6 7_21 2.839e-03 111.44 4.103e-06 -3.3024 1
10995 C6orf48 6_27 5.034e-10 92.92 6.067e-13 3.4168 3
10782 HLA-DRB5 6_27 5.340e-14 81.74 5.662e-17 2.8311 1
10979 RNF5 6_27 6.906e-14 80.93 7.249e-17 7.9213 1
10981 PRRT1 6_27 6.839e-14 80.56 7.146e-17 7.9069 1
4942 IER3 6_24 5.995e-15 76.18 5.924e-18 2.1673 1
10546 ZSCAN26 6_22 1.016e-02 70.00 9.229e-06 8.5444 2
12006 HLA-DMB 6_27 4.488e-01 67.97 3.957e-04 -8.0771 1
11769 TRIM26 6_24 2.298e-14 67.96 2.026e-17 -4.5226 2
10071 BTN3A2 6_20 1.990e-02 66.16 1.708e-05 9.1074 2
9707 GRIN2A 16_10 3.721e-07 65.62 3.167e-10 0.3187 2Genes with highest PVE
genename region_tag susie_pip mu2 PVE z num_eqtl
3506 TBC1D15 12_44 0.6802 44.94 0.0003965 4.584 2
12006 HLA-DMB 6_27 0.4488 67.97 0.0003957 -8.077 1
10687 ZNF823 19_10 0.9809 29.59 0.0003764 5.485 1
2981 KCNJ13 2_137 0.6658 37.90 0.0003273 6.658 1
8275 INO80E 16_24 0.6509 38.24 0.0003229 6.230 1
6084 ARFGAP2 11_29 0.9434 25.73 0.0003148 4.839 1
1736 PPP1R16B 20_23 0.6717 34.97 0.0003047 6.009 1
2535 MDK 11_28 0.5855 38.49 0.0002923 -6.344 1
7929 ENDOG 9_66 0.8524 23.46 0.0002593 4.814 2
8759 MAP3K11 11_36 0.8669 22.56 0.0002537 -4.409 1
104 ELAC2 17_11 0.7884 22.09 0.0002259 4.372 1
12742 TBC1D29 17_18 0.7667 22.68 0.0002255 4.504 2
4009 SPECC1 17_16 0.7772 22.05 0.0002223 4.167 1
10075 TMEM222 1_19 0.7030 23.44 0.0002137 3.902 1
2916 LMAN2L 2_57 0.6234 26.42 0.0002136 -4.473 2
1516 TTLL1 22_18 0.6929 23.38 0.0002101 -4.510 2
3607 BHLHE41 12_18 0.6378 24.96 0.0002065 -3.860 1
424 FAM120A 9_47 0.6753 23.25 0.0002037 4.571 1
8075 BATF2 11_36 0.5647 27.27 0.0001998 -4.859 2
2590 VPS29 12_67 0.5781 26.61 0.0001996 -4.982 1Genes with largest z scores
genename region_tag susie_pip mu2 PVE z num_eqtl
6779 ZSCAN12 6_22 4.256e-02 125.06 6.903e-05 10.940 1
11759 HIST1H2BN 6_21 9.808e-07 988.31 1.257e-08 10.773 1
10071 BTN3A2 6_20 1.990e-02 66.16 1.708e-05 9.107 2
13065 RP1-86C11.7 6_21 2.000e-12 428.96 1.113e-14 -9.033 1
12628 CTA-14H9.5 6_20 1.914e-02 64.08 1.591e-05 8.937 1
10999 VWA7 6_27 6.840e-05 157.52 1.398e-07 8.911 1
10546 ZSCAN26 6_22 1.016e-02 70.00 9.229e-06 8.544 2
12063 C4A 6_27 9.696e-09 130.50 1.641e-11 8.352 2
12006 HLA-DMB 6_27 4.488e-01 67.97 3.957e-04 -8.077 1
9448 HIST1H2BC 6_20 2.014e-02 50.53 1.320e-05 -7.978 1
10979 RNF5 6_27 6.906e-14 80.93 7.249e-17 7.921 1
10981 PRRT1 6_27 6.839e-14 80.56 7.146e-17 7.907 1
11006 APOM 6_27 1.374e-08 125.29 2.232e-11 7.890 2
6064 CNNM2 10_66 1.510e-01 40.14 7.862e-05 -7.876 1
910 NT5C2 10_66 3.130e-01 40.94 1.662e-04 -7.507 2
10219 ZSCAN23 6_22 2.302e-02 47.21 1.409e-05 -7.124 1
10362 ZKSCAN3 6_22 1.140e-02 39.61 5.855e-06 6.866 1
2981 KCNJ13 2_137 6.658e-01 37.90 3.273e-04 6.658 1
10290 DPYD 1_60 1.525e-02 38.86 7.688e-06 -6.455 1
2535 MDK 11_28 5.855e-01 38.49 2.923e-04 -6.344 1Comparing z scores and PIPs
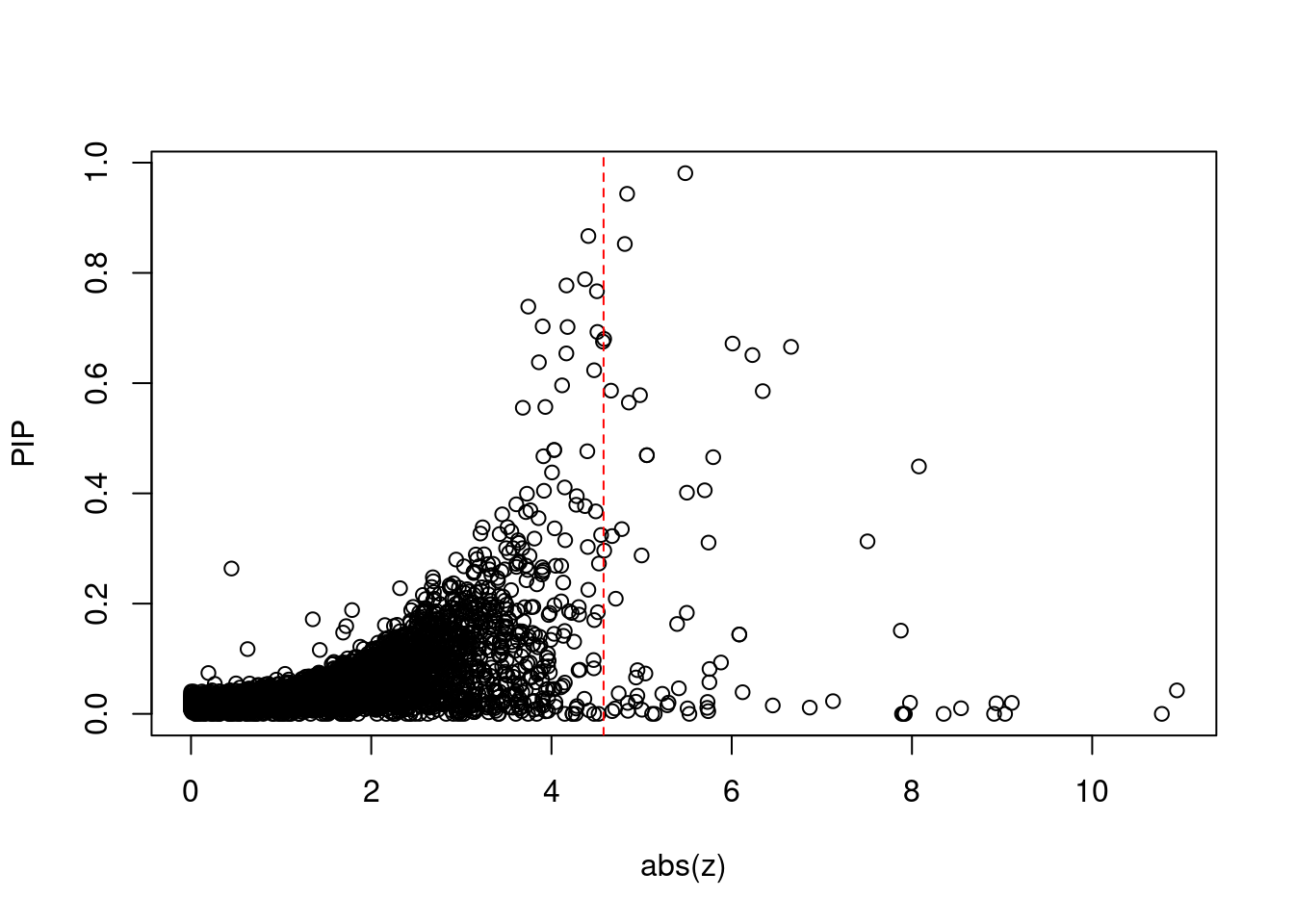
[1] 0.00657GO enrichment analysis for genes with PIP>0.5
#number of genes for gene set enrichment
length(genes)[1] 26Uploading data to Enrichr... Done.
Querying GO_Biological_Process_2021... Done.
Querying GO_Cellular_Component_2021... Done.
Querying GO_Molecular_Function_2021... Done.
Parsing results... Done.
[1] "GO_Biological_Process_2021"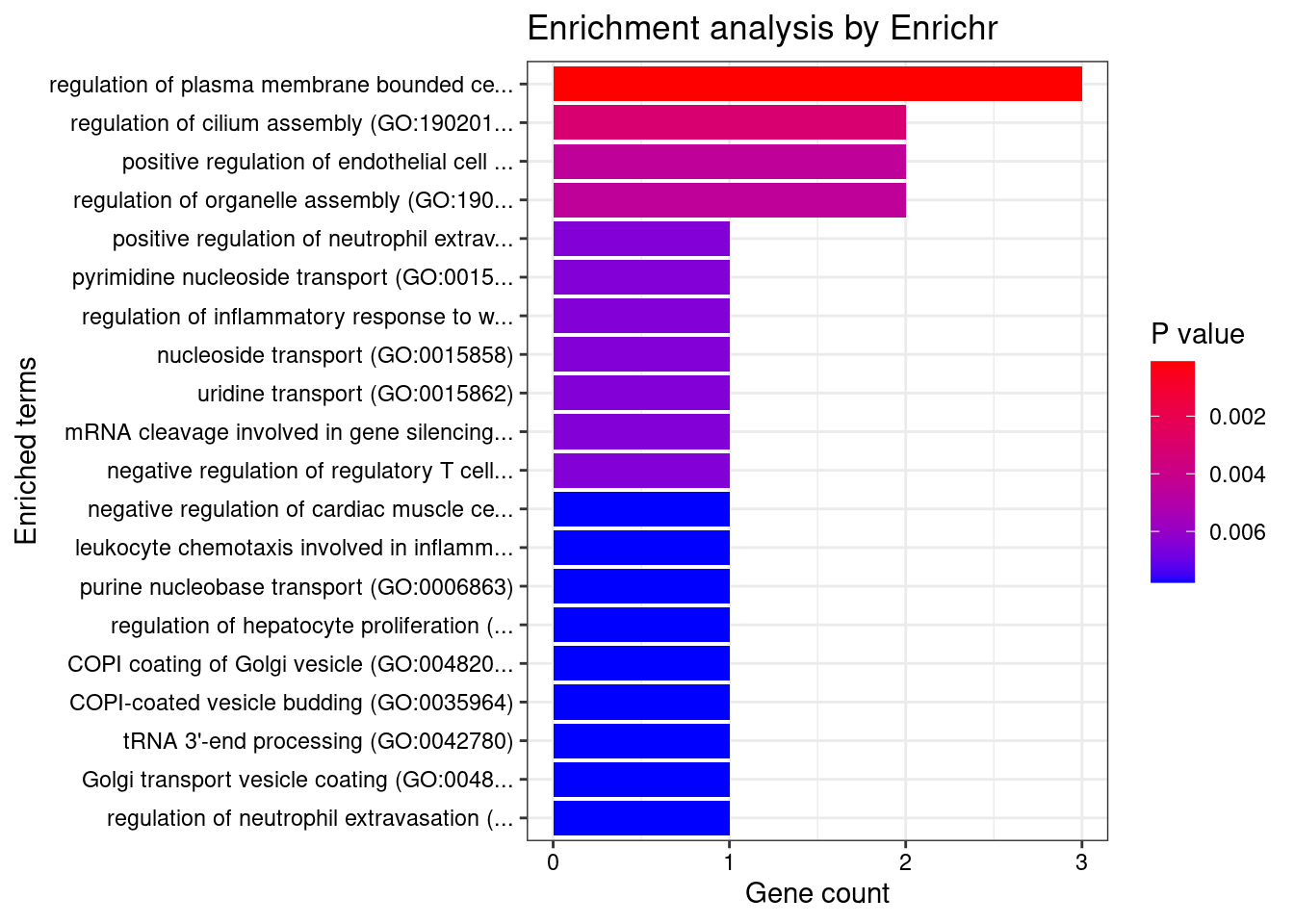
Term
1 regulation of plasma membrane bounded cell projection assembly (GO:0120032)
Overlap Adjusted.P.value Genes
1 3/70 0.02821 PPP1R16B;TBC1D19;TBC1D15
[1] "GO_Cellular_Component_2021"
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)
[1] "GO_Molecular_Function_2021"
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)DisGeNET enrichment analysis for genes with PIP>0.5
Description FDR Ratio
45 Snowflake vitreoretinal degeneration 0.007421 1/8
47 LEBER CONGENITAL AMAUROSIS 16 0.007421 1/8
48 PROSTATE CANCER, HEREDITARY, 2 0.007421 1/8
50 COMBINED OXIDATIVE PHOSPHORYLATION DEFICIENCY 17 0.007421 1/8
52 MENTAL RETARDATION, AUTOSOMAL RECESSIVE 52 0.007421 1/8
53 Bile acid CoA ligase deficiency and defective amidation 0.007421 1/8
33 Long Sleeper Syndrome 0.025918 1/8
34 Short Sleeper Syndrome 0.025918 1/8
35 Sleep-Related Neurogenic Tachypnea 0.025918 1/8
36 Subwakefullness Syndrome 0.025918 1/8
BgRatio
45 1/9703
47 1/9703
48 1/9703
50 1/9703
52 1/9703
53 1/9703
33 7/9703
34 7/9703
35 7/9703
36 7/9703WebGestalt enrichment analysis for genes with PIP>0.5
Loading the functional categories...
Loading the ID list...
Loading the reference list...
Performing the enrichment analysis...Warning in oraEnrichment(interestGeneList, referenceGeneList, geneSet, minNum =
minNum, : No significant gene set is identified based on FDR 0.05!NULLPIP Manhattan Plot
Warning: 'timedatectl' indicates the non-existent timezone name 'n/a'Warning: Your system is mis-configured: '/etc/localtime' is not a symlinkWarning: It is strongly recommended to set envionment variable TZ to 'America/
Chicago' (or equivalent)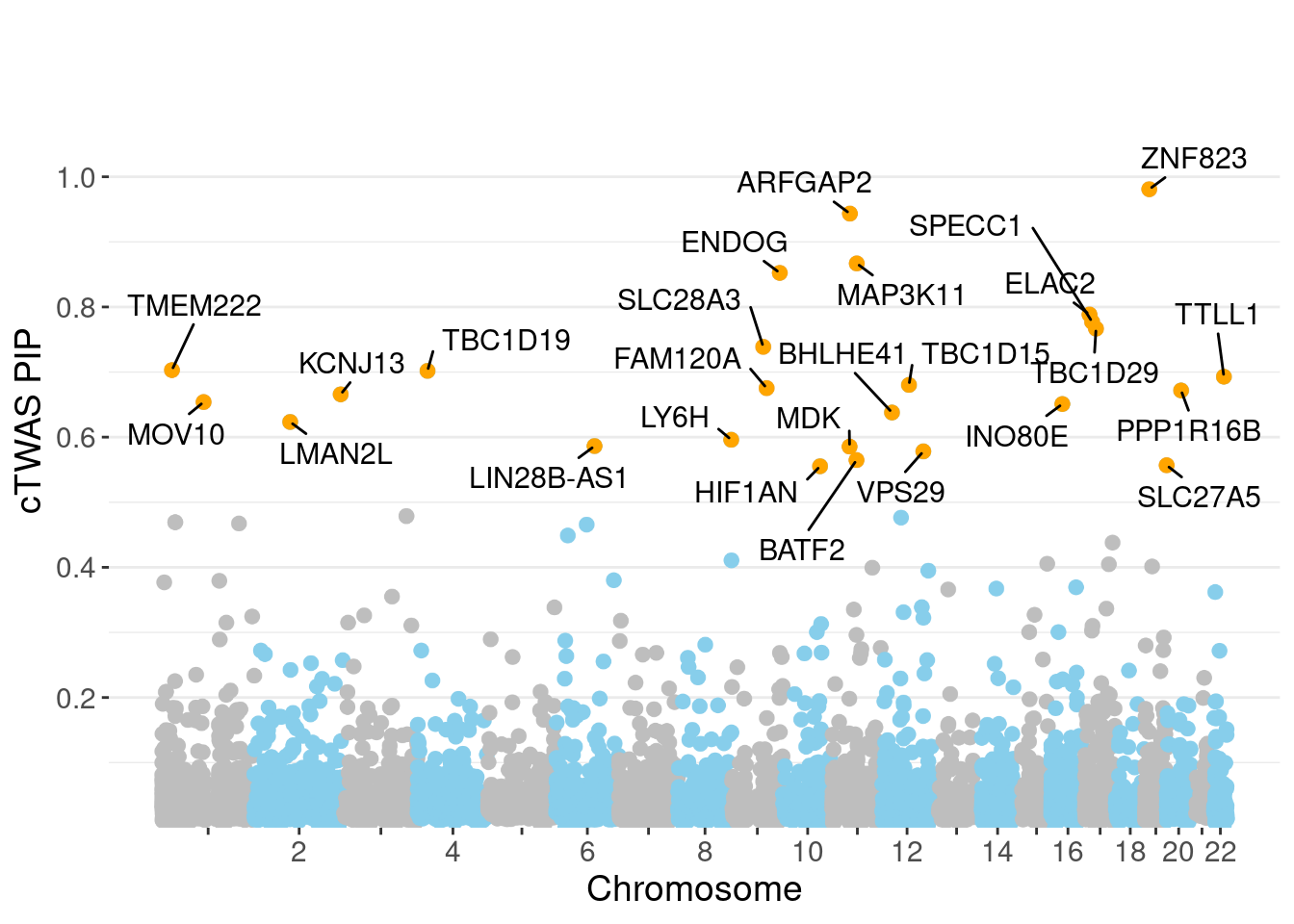
Sensitivity, specificity and precision for silver standard genes
#number of genes in known annotations
print(length(known_annotations))[1] 130#number of genes in known annotations with imputed expression
print(sum(known_annotations %in% ctwas_gene_res$genename))[1] 67#significance threshold for TWAS
print(sig_thresh)[1] 4.578#number of ctwas genes
length(ctwas_genes)[1] 4#number of TWAS genes
length(twas_genes)[1] 70#show novel genes (ctwas genes with not in TWAS genes)
ctwas_gene_res[ctwas_gene_res$genename %in% novel_genes,report_cols] genename region_tag susie_pip mu2 PVE z num_eqtl
8759 MAP3K11 11_36 0.8669 22.56 0.0002537 -4.409 1#sensitivity / recall
print(sensitivity) ctwas TWAS
0.007692 0.076923 #specificity
print(specificity) ctwas TWAS
0.9997 0.9943 #precision / PPV
print(precision) ctwas TWAS
0.2500 0.1429 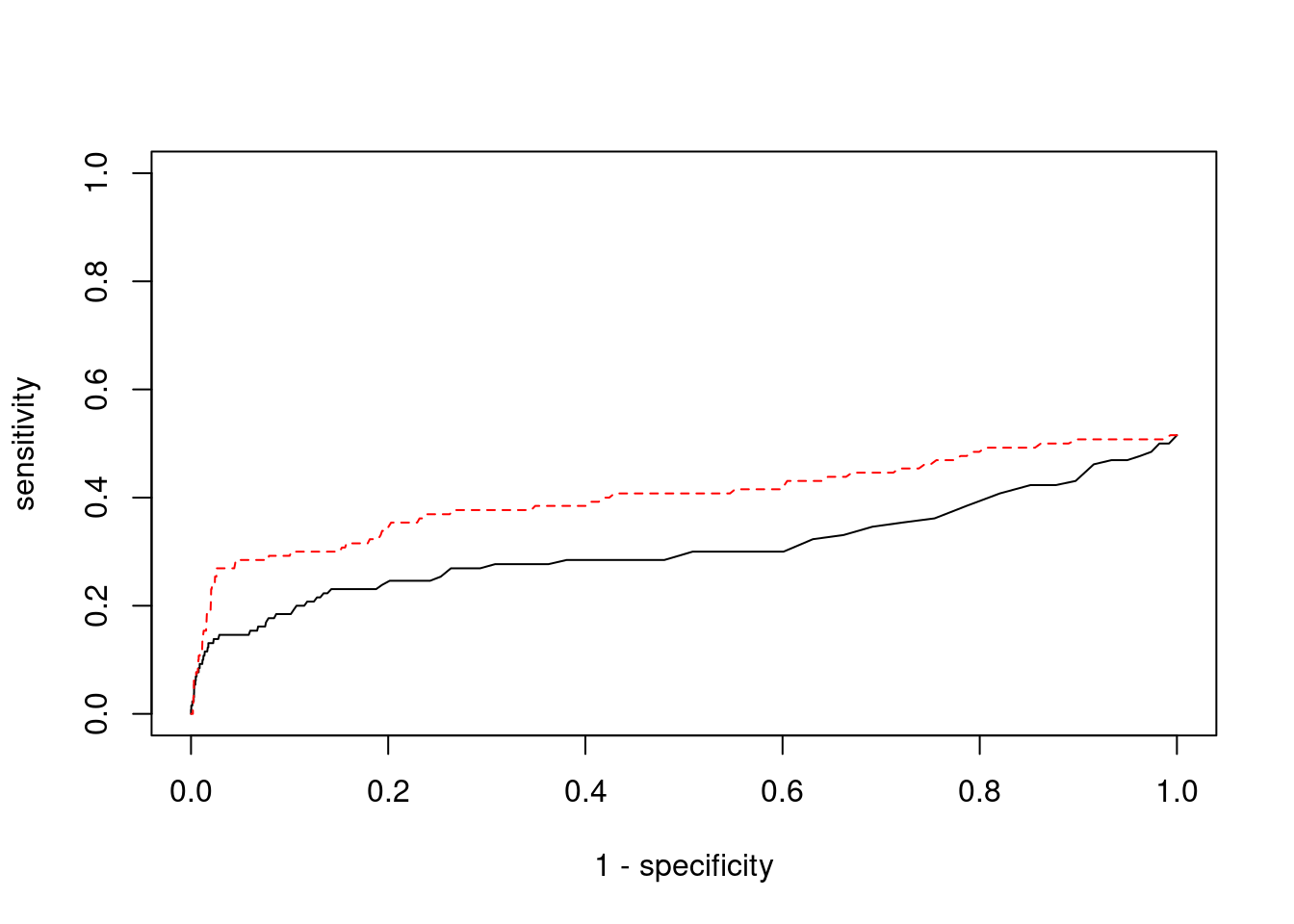
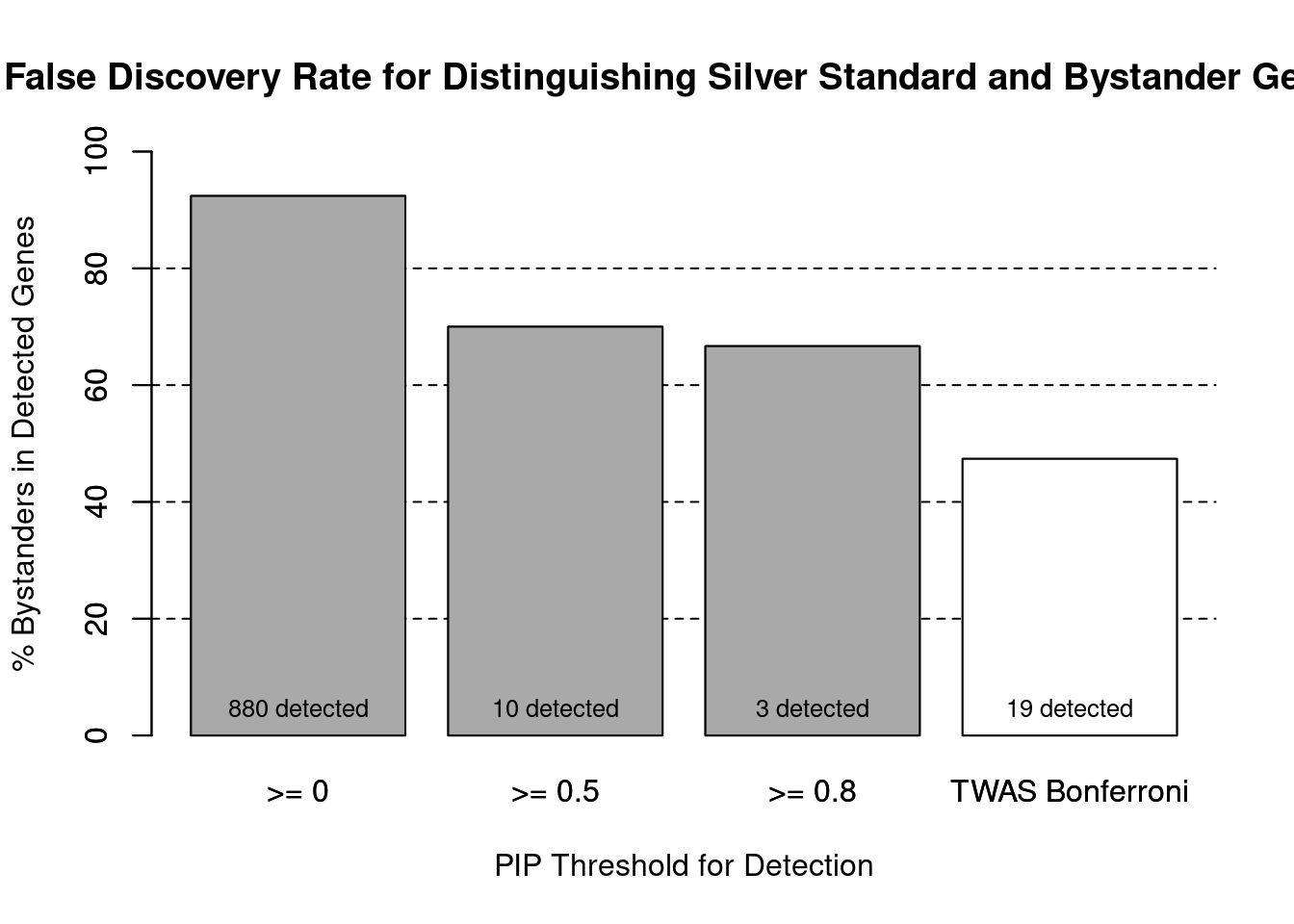
cTWAS is more precise than TWAS in distinguishing silver standard and bystander genes
#number of genes in known annotations (with imputed expression)
print(length(known_annotations))[1] 67#number of bystander genes (with imputed expression)
print(length(unrelated_genes))[1] 813#subset results to genes in known annotations or bystanders
ctwas_gene_res_subset <- ctwas_gene_res[ctwas_gene_res$genename %in% c(known_annotations, unrelated_genes),]
#assign ctwas and TWAS genes
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>0.8]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>sig_thresh]
#significance threshold for TWAS
print(sig_thresh)[1] 4.578#number of ctwas genes (in known annotations or bystanders)
length(ctwas_genes)[1] 3#number of TWAS genes (in known annotations or bystanders)
length(twas_genes)[1] 19#sensitivity / recall
sensitivity ctwas TWAS
0.01493 0.14925 #specificity / (1 - False Positive Rate)
specificity ctwas TWAS
0.9975 0.9889 #precision / PPV / (1 - False Discovery Rate)
precision ctwas TWAS
0.3333 0.5263 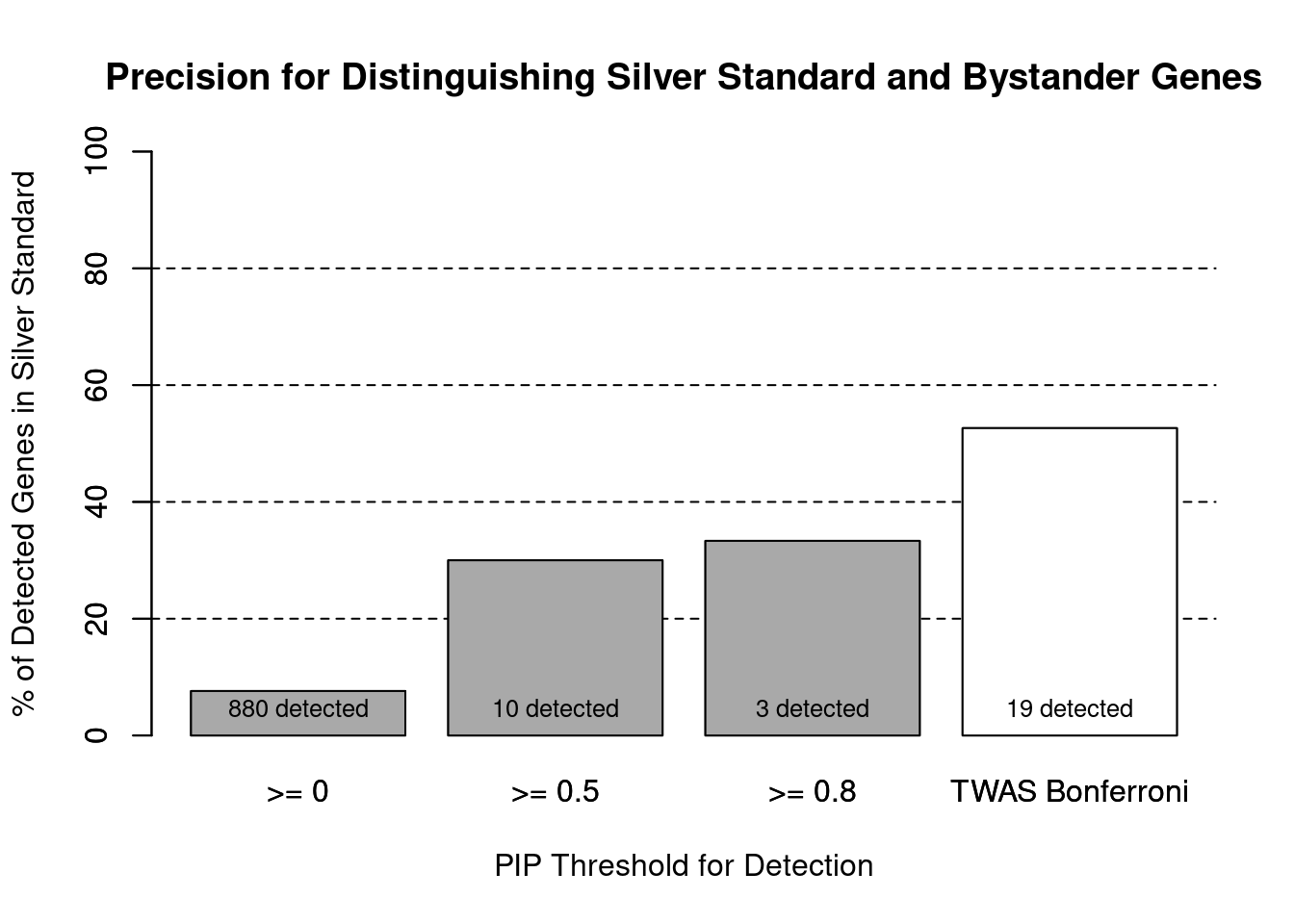
pip_range <- (0:1000)/1000
sensitivity <- rep(NA, length(pip_range))
specificity <- rep(NA, length(pip_range))
for (index in 1:length(pip_range)){
pip <- pip_range[index]
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>=pip]
sensitivity[index] <- sum(ctwas_genes %in% known_annotations)/length(known_annotations)
specificity[index] <- sum(!(unrelated_genes %in% ctwas_genes))/length(unrelated_genes)
}
plot(1-specificity, sensitivity, type="l", xlim=c(0,1), ylim=c(0,1), main="", xlab="1 - Specificity", ylab="Sensitivity")
title(expression("ROC Curve for cTWAS (black) and TWAS (" * phantom("red") * ")"))
title(expression(phantom("ROC Curve for cTWAS (black) and TWAS (") * "red" * phantom(")")), col.main="red")
sig_thresh_range <- seq(from=0, to=max(abs(ctwas_gene_res_subset$z)), length.out=length(pip_range))
for (index in 1:length(sig_thresh_range)){
sig_thresh_plot <- sig_thresh_range[index]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>=sig_thresh_plot]
sensitivity[index] <- sum(twas_genes %in% known_annotations)/length(known_annotations)
specificity[index] <- sum(!(unrelated_genes %in% twas_genes))/length(unrelated_genes)
}
lines(1-specificity, sensitivity, xlim=c(0,1), ylim=c(0,1), col="red", lty=1)
abline(a=0,b=1,lty=3)
#add previously computed points from the analysis
ctwas_genes <- ctwas_gene_res_subset$genename[ctwas_gene_res_subset$susie_pip>0.8]
twas_genes <- ctwas_gene_res_subset$genename[abs(ctwas_gene_res_subset$z)>sig_thresh]
points(1-specificity_plot["ctwas"], sensitivity_plot["ctwas"], pch=21, bg="black")
points(1-specificity_plot["TWAS"], sensitivity_plot["TWAS"], pch=21, bg="red")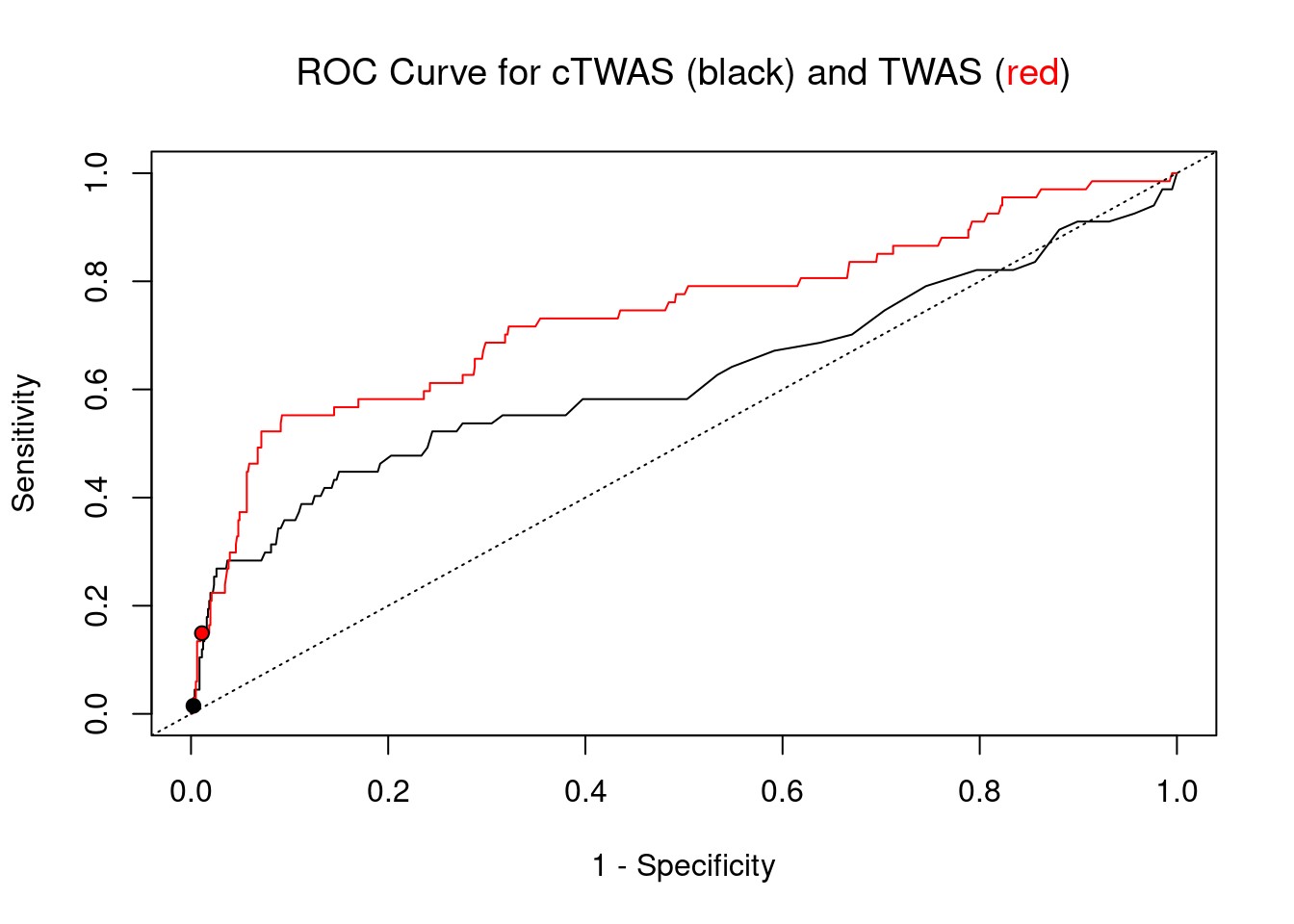
Undetected silver standard genes have low TWAS z-scores or stronger signal from nearby variants
#table of outcomes for silver standard genes
-sort(-table(silver_standard_case))silver_standard_case
Not Imputed Insignificant z-score Nearby SNP(s)
63 57 9
Detected (PIP > 0.8)
1 #show inconclusive genes
silver_standard_case[silver_standard_case=="Inconclusive"]named character(0)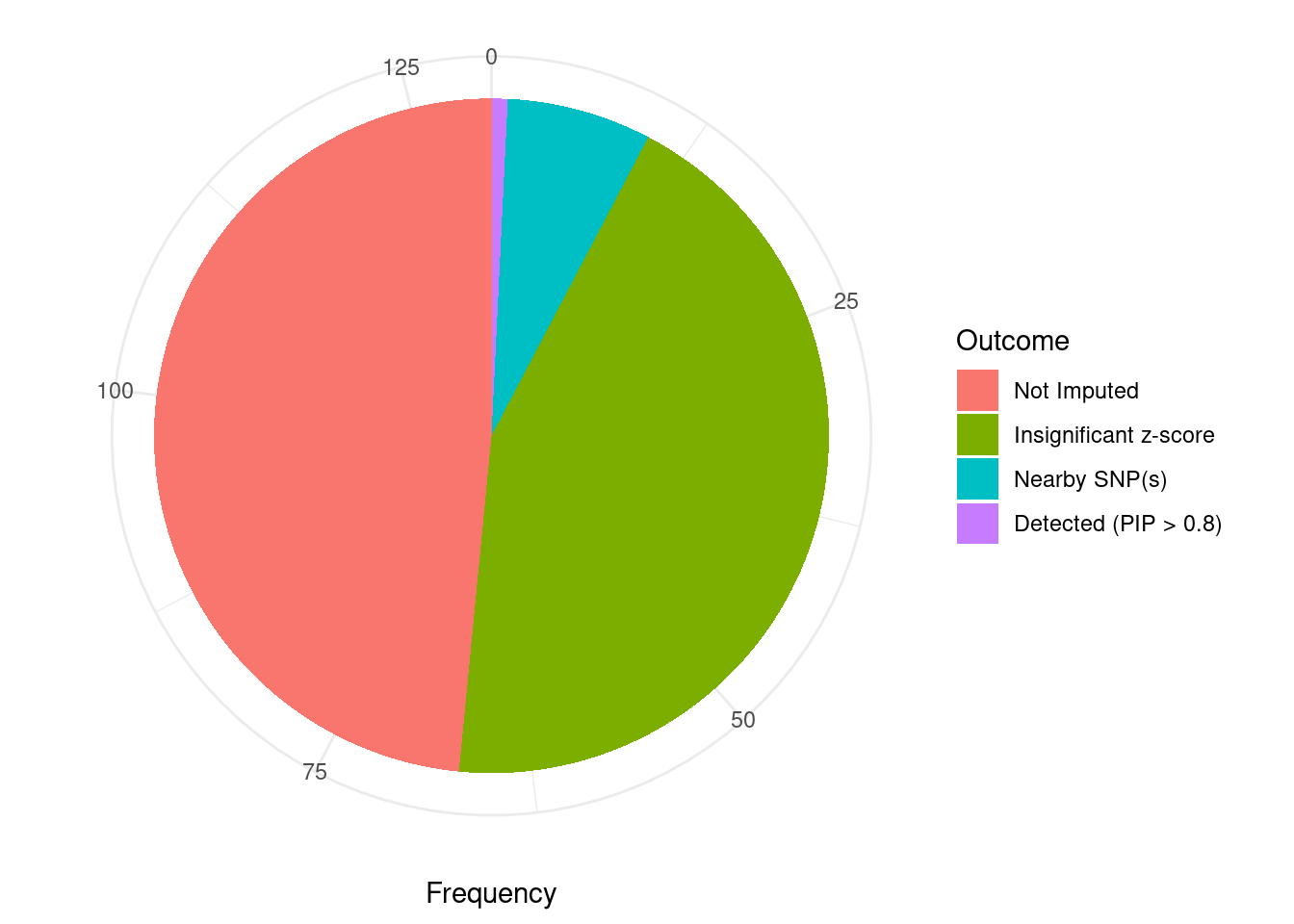
sessionInfo()R version 3.6.1 (2019-07-05)
Platform: x86_64-pc-linux-gnu (64-bit)
Running under: Scientific Linux 7.4 (Nitrogen)
Matrix products: default
BLAS/LAPACK: /software/openblas-0.2.19-el7-x86_64/lib/libopenblas_haswellp-r0.2.19.so
locale:
[1] LC_CTYPE=en_US.UTF-8 LC_NUMERIC=C
[3] LC_TIME=en_US.UTF-8 LC_COLLATE=en_US.UTF-8
[5] LC_MONETARY=en_US.UTF-8 LC_MESSAGES=en_US.UTF-8
[7] LC_PAPER=en_US.UTF-8 LC_NAME=C
[9] LC_ADDRESS=C LC_TELEPHONE=C
[11] LC_MEASUREMENT=en_US.UTF-8 LC_IDENTIFICATION=C
attached base packages:
[1] parallel stats4 stats graphics grDevices utils datasets
[8] methods base
other attached packages:
[1] GenomicRanges_1.36.1 GenomeInfoDb_1.20.0 IRanges_2.18.1
[4] S4Vectors_0.22.1 BiocGenerics_0.30.0 biomaRt_2.40.1
[7] readxl_1.3.1 forcats_0.5.1 stringr_1.4.0
[10] dplyr_1.0.7 purrr_0.3.4 readr_2.1.1
[13] tidyr_1.1.4 tidyverse_1.3.1 tibble_3.1.6
[16] WebGestaltR_0.4.4 disgenet2r_0.99.2 enrichR_3.0
[19] cowplot_1.0.0 ggplot2_3.3.5 workflowr_1.7.0
loaded via a namespace (and not attached):
[1] ggbeeswarm_0.6.0 colorspace_2.0-2 rjson_0.2.20
[4] ellipsis_0.3.2 rprojroot_2.0.2 XVector_0.24.0
[7] fs_1.5.2 rstudioapi_0.13 farver_2.1.0
[10] ggrepel_0.9.1 bit64_4.0.5 AnnotationDbi_1.46.0
[13] fansi_1.0.2 lubridate_1.8.0 xml2_1.3.3
[16] codetools_0.2-16 doParallel_1.0.17 cachem_1.0.6
[19] knitr_1.36 jsonlite_1.7.2 apcluster_1.4.8
[22] Cairo_1.5-12.2 broom_0.7.10 dbplyr_2.1.1
[25] compiler_3.6.1 httr_1.4.2 backports_1.4.1
[28] assertthat_0.2.1 Matrix_1.2-18 fastmap_1.1.0
[31] cli_3.1.0 later_0.8.0 prettyunits_1.1.1
[34] htmltools_0.5.2 tools_3.6.1 igraph_1.2.10
[37] GenomeInfoDbData_1.2.1 gtable_0.3.0 glue_1.6.2
[40] reshape2_1.4.4 doRNG_1.8.2 Rcpp_1.0.8
[43] Biobase_2.44.0 cellranger_1.1.0 jquerylib_0.1.4
[46] vctrs_0.3.8 svglite_1.2.2 iterators_1.0.14
[49] xfun_0.29 ps_1.6.0 rvest_1.0.2
[52] lifecycle_1.0.1 rngtools_1.5.2 XML_3.99-0.3
[55] zlibbioc_1.30.0 getPass_0.2-2 scales_1.1.1
[58] vroom_1.5.7 hms_1.1.1 promises_1.0.1
[61] yaml_2.2.1 curl_4.3.2 memoise_2.0.1
[64] ggrastr_1.0.1 gdtools_0.1.9 stringi_1.7.6
[67] RSQLite_2.2.8 highr_0.9 foreach_1.5.2
[70] rlang_1.0.1 pkgconfig_2.0.3 bitops_1.0-7
[73] evaluate_0.14 lattice_0.20-38 labeling_0.4.2
[76] bit_4.0.4 processx_3.5.2 tidyselect_1.1.1
[79] plyr_1.8.6 magrittr_2.0.2 R6_2.5.1
[82] generics_0.1.1 DBI_1.1.2 pillar_1.6.4
[85] haven_2.4.3 whisker_0.3-2 withr_2.4.3
[88] RCurl_1.98-1.5 modelr_0.1.8 crayon_1.5.0
[91] utf8_1.2.2 tzdb_0.2.0 rmarkdown_2.11
[94] progress_1.2.2 grid_3.6.1 data.table_1.14.2
[97] blob_1.2.2 callr_3.7.0 git2r_0.26.1
[100] reprex_2.0.1 digest_0.6.29 httpuv_1.5.1
[103] munsell_0.5.0 beeswarm_0.2.3 vipor_0.4.5