resistance
lukeholman
2020-11-30
Last updated: 2020-12-18
Checks: 7 0
Knit directory: exp_evol_respiration/
This reproducible R Markdown analysis was created with workflowr (version 1.6.2). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
Great! Since the R Markdown file has been committed to the Git repository, you know the exact version of the code that produced these results.
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
The command set.seed(20190703) was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
Nice! There were no cached chunks for this analysis, so you can be confident that you successfully produced the results during this run.
Great job! Using relative paths to the files within your workflowr project makes it easier to run your code on other machines.
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility.
The results in this page were generated with repository version 5a81a83. See the Past versions tab to see a history of the changes made to the R Markdown and HTML files.
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can use wflow_publish or wflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Ignored files:
Ignored: .DS_Store
Ignored: .Rapp.history
Ignored: .Rproj.user/
Ignored: analysis/.DS_Store
Ignored: output/.DS_Store
Untracked files:
Untracked: output/brms_SEM_respiration.rds
Untracked: output/metabolite_interaction_plot.pdf
Untracked: output/metabolite_pairs_plot.pdf
Untracked: output/metabolite_plot.pdf
Untracked: output/respiration_figure.pdf
Untracked: output/respiration_pairs_plot.pdf
Untracked: post_preds_respiration.rds
Unstaged changes:
Deleted: analysis/about.Rmd
Deleted: output/Figure 2.pdf
Deleted: output/bayesian_R2_values.rds
Deleted: output/brms_SEM.rds
Deleted: output/pairs_plot.pdf
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
These are the previous versions of the repository in which changes were made to the R Markdown (analysis/resistance.Rmd) and HTML (docs/resistance.html) files. If you’ve configured a remote Git repository (see ?wflow_git_remote), click on the hyperlinks in the table below to view the files as they were in that past version.
| File | Version | Author | Date | Message |
|---|---|---|---|---|
| Rmd | 96d1188 | Martin Garlovsky | 2020-12-13 | MDG commit |
| html | 96d1188 | Martin Garlovsky | 2020-12-13 | MDG commit |
| Rmd | 7d4b609 | Martin Garlovsky | 2020-12-05 | MDG commit |
| html | 7d4b609 | Martin Garlovsky | 2020-12-05 | MDG commit |
| Rmd | 8e1e5c1 | Martin Garlovsky | 2020-12-04 | MDG commit again |
| html | 8e1e5c1 | Martin Garlovsky | 2020-12-04 | MDG commit again |
| html | 287d75b | Martin Garlovsky | 2020-12-04 | MDG commit |
| html | 45751bf | Martin Garlovsky | 2020-12-04 | MDG commit |
| Rmd | c175be4 | Martin Garlovsky | 2020-12-04 | MDG commit |
| html | c175be4 | Martin Garlovsky | 2020-12-04 | MDG commit |
| html | df61dde | Martin Garlovsky | 2020-12-04 | MDG commit |
| Rmd | 0714753 | Martin Garlovsky | 2020-12-04 | workflowr::wflow_git_commit(all = T) |
| Rmd | 3fdbcb2 | lukeholman | 2020-11-30 | Tweaks Nov 2020 |
Load packages
library(tidyverse)
library(coxme)
library(lme4)
library(brms)
library(tidybayes)
library(ggridges)
library(kableExtra)
library(knitrhooks) # install with devtools::install_github("nathaneastwood/knitrhooks")
output_max_height() # a knitrhook option
options(stringsAsFactors = FALSE)Load data
# load desiccation resistance data
DesRes <- read.csv("data/3.DesRes.csv") %>%
# add event (all flies died)
mutate(EVENT = 1,
LINE = paste0(Treatment, substr(Replicate, 2, 2)))
# calculate survival times
# paste time and date
DesRes$d <- paste(DesRes$Death_date, DesRes$Death_time, sep = ' ')
# experiment start time
start_timeDes <- "04/02/2017 12:00"
DesRes$survival.time <- as.numeric(strptime(DesRes$d, format = "%d/%m/%Y %H") - strptime(start_timeDes, format = "%d/%m/%Y %H"))
des.surv <- Surv(DesRes$survival.time, DesRes$EVENT)
# load starvation resistance data
StaRes <- read.csv("data/3.StarvRes.csv") %>%
# add event (all flies died)
mutate(EVENT = 1,
LINE = paste0(Treatment, substr(Replicate, 2, 2)))
# calculate survival times
# paste time and date
StaRes$d <- paste(StaRes$Death_date, StaRes$Death_time, sep = ' ')
# experiment start time
start_timeSta <- "04/02/2017 12:00"
StaRes$survival.time <- as.numeric(strptime(StaRes$d, format = "%d/%m/%Y %H") - strptime(start_timeSta, format = "%d/%m/%Y %H"))
summary(StaRes)
# 5 individuals have missing survival times which we will right censor at max. survival time
StaRes[which(is.na(StaRes$survival.time)), 'EVENT'] <- 0
StaRes[which(is.na(StaRes$survival.time)), 'survival.time'] <- max(na.omit(StaRes$survival.time))Inspecting the raw data
bind_rows(
DesRes %>%
select(Treatment, Sex, survival.time) %>% mutate(var = 'Desiccation'),
StaRes %>% filter(EVENT == 1) %>%
select(Treatment, Sex, survival.time) %>% mutate(var = 'Starvation')
) %>%
mutate(var2 = paste(Treatment, Sex)) %>%
ggplot(aes(x = survival.time, y = Sex, fill = var2)) +
geom_boxplot() +
scale_fill_manual(values = c("pink", "skyblue", "red", "blue"), name = "") +
labs(x = 'Survival time (hours)') +
facet_wrap(~var, ncol = 2) +
theme_bw() +
NULL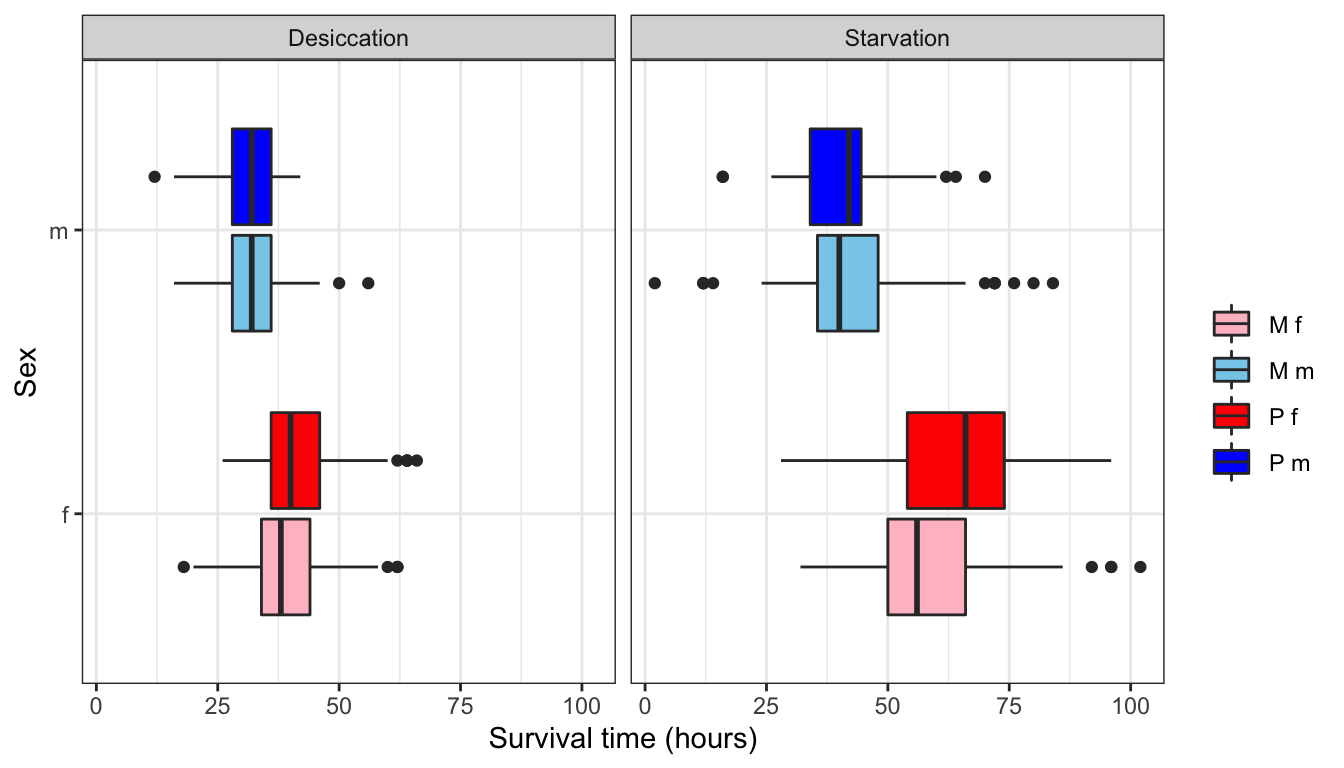
Figure 1: Survival time in hours for flies in each treatment split by sex.
Fit the model for desiccation/starvation resistance
Plot the survival curves and median survival times 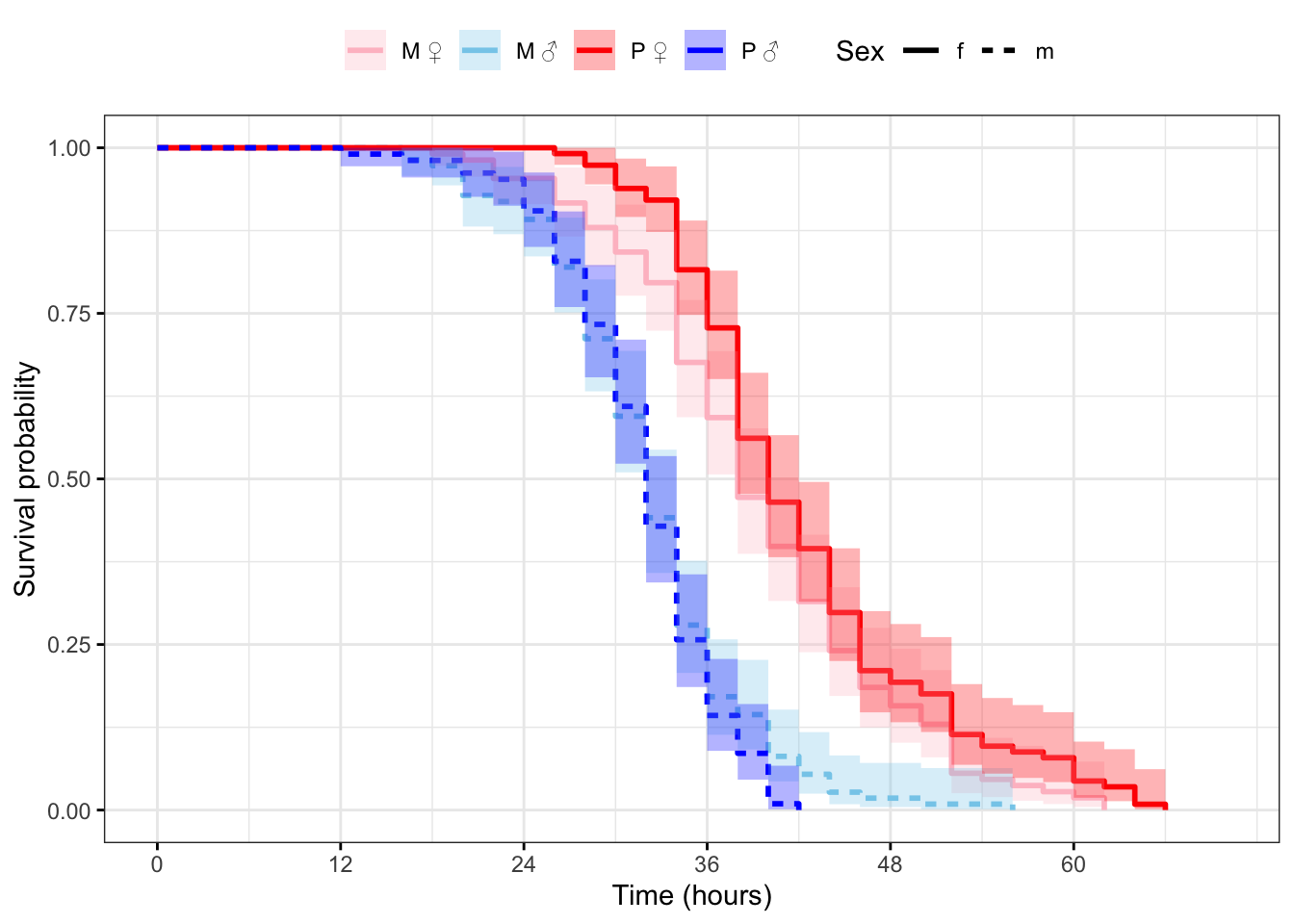
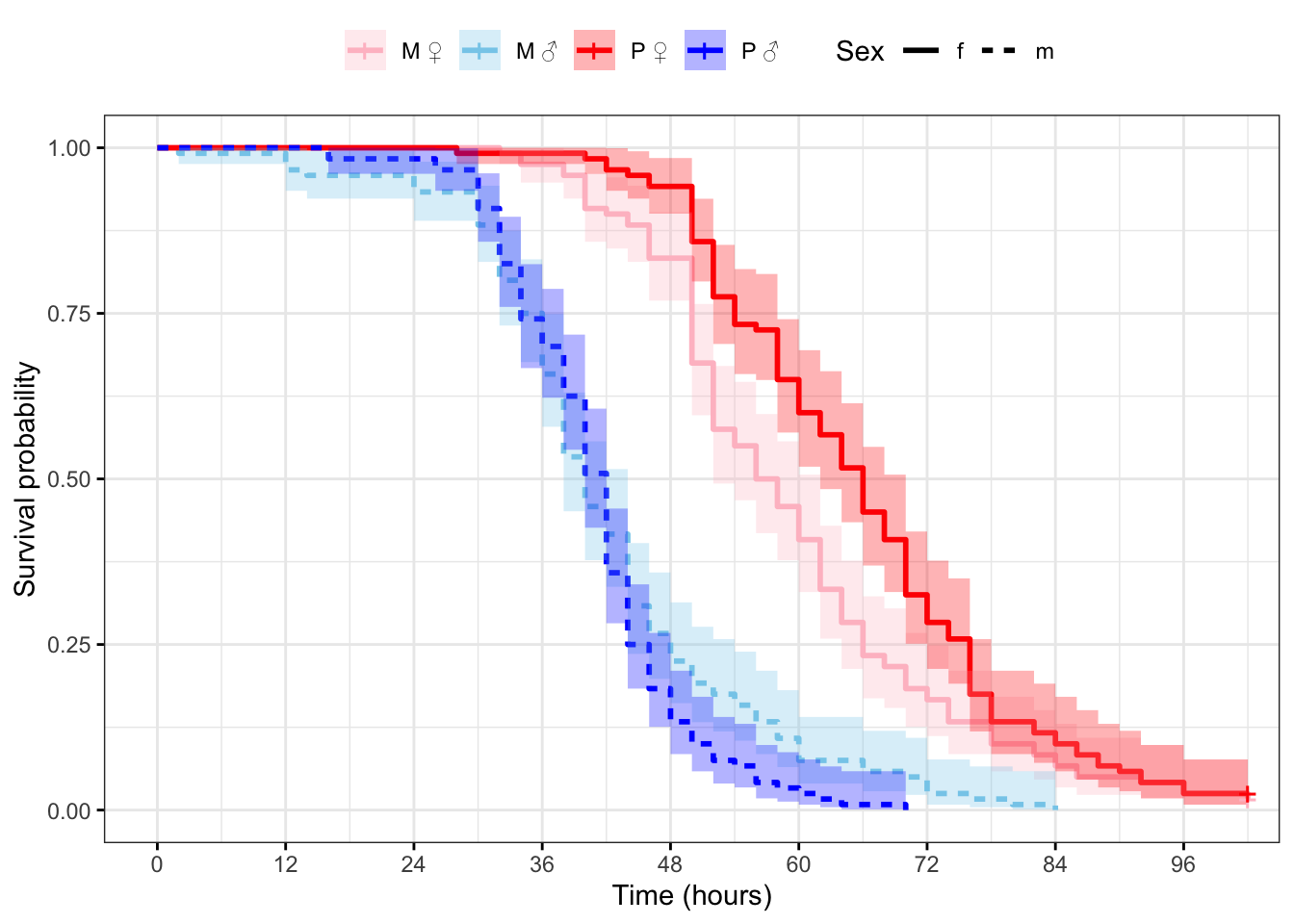
Figure 2: Kaplan-Meier survival curves for flies in each treatment split by sex. + indicates censored individuals (n = 5).
# median eclosion times
survfit(Surv(survival.time, EVENT) ~ Treatment + Sex, data = DesRes)Call: survfit(formula = Surv(survival.time, EVENT) ~ Treatment + Sex,
data = DesRes)
n events median 0.95LCL 0.95UCL
Treatment=M, Sex=f 108 108 38 38 42
Treatment=M, Sex=m 111 111 32 32 34
Treatment=P, Sex=f 114 114 40 38 42
Treatment=P, Sex=m 105 105 32 32 34
survfit(Surv(survival.time, EVENT) ~ Treatment + Sex, data = StaRes)Call: survfit(formula = Surv(survival.time, EVENT) ~ Treatment + Sex,
data = StaRes)
n events median 0.95LCL 0.95UCL
Treatment=M, Sex=f 120 118 57 52 62
Treatment=M, Sex=m 120 120 40 38 44
Treatment=P, Sex=f 120 117 66 62 70
Treatment=P, Sex=m 120 120 42 40 42
Next we check that the proportional hazards assumption is met.
# assess proportional hazards assumption
par(mar = c(2, 2, 2, 2), mfrow = c(1, 2))
plot(survfit(Surv(survival.time, EVENT) ~ Treatment + Sex, data = DesRes),
lty = 1:2, lwd = 2,
col = c("pink", "skyblue", "red", "blue"),
main = 'Desiccation',
fun = "cloglog")
legend("topleft", c("M \u2640","M \u2642",'P \u2640','P \u2642'),
col = c("pink", "skyblue", "red", "blue"),
lty = 1:2,
lwd = 2,
bty = 'n'
)
plot(survfit(Surv(survival.time, EVENT) ~ Treatment + Sex, data = StaRes),
lty = 1:2, lwd = 2,
col = c("pink", "skyblue", "red", "blue"),
main = 'Starvation',
fun = "cloglog")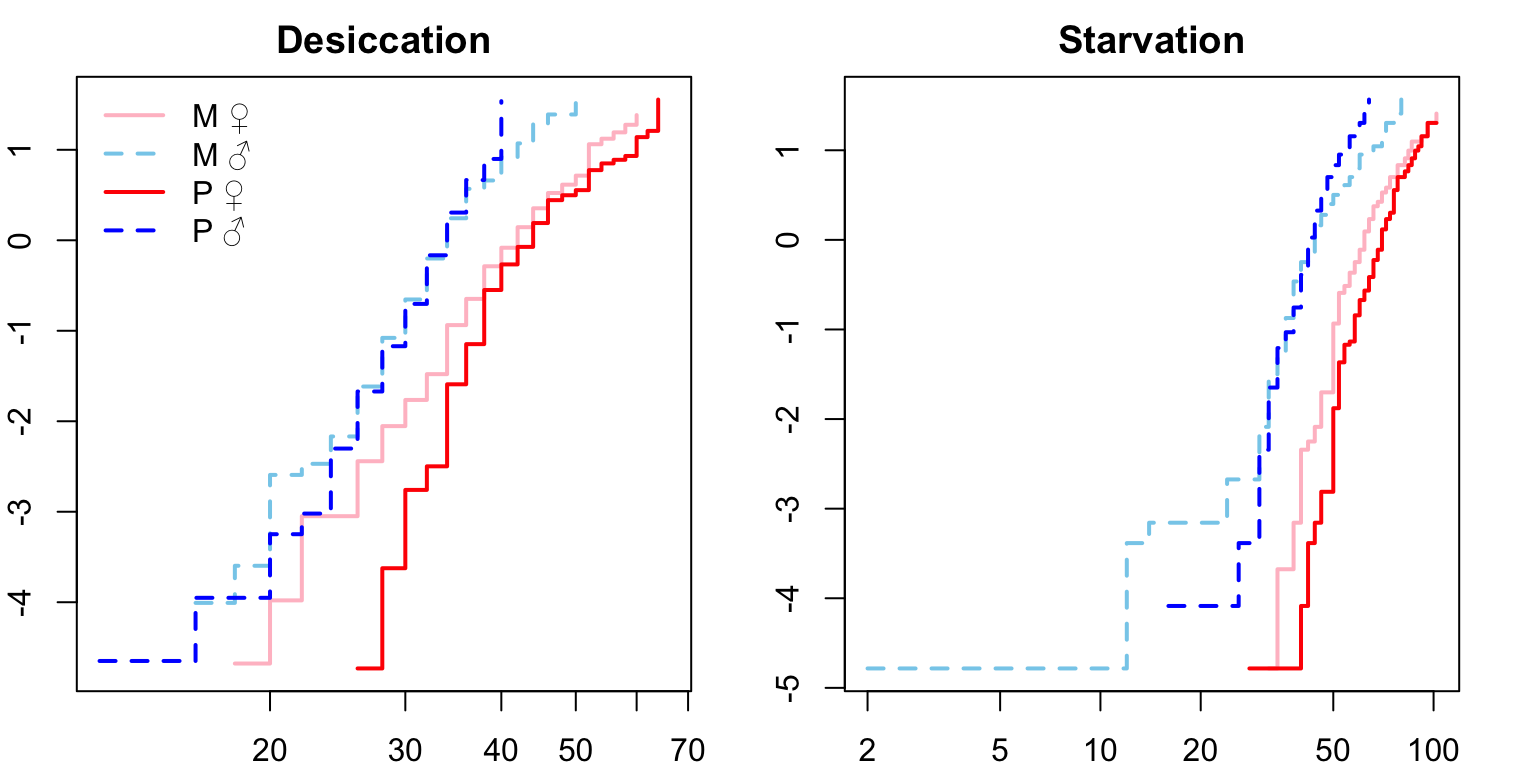
For both desiccation and starvation we see crossing hazards for the male survival curves. We will therefore fit accelerated failure time (AFT) models with a Weibull distribution (see this paper) and a frailty term to account for replicates within each treatment. We can define the degrees of freedom explicitly (df = 6).
Fit the Accelerated failure time models
weibull.des <- survreg(Surv(survival.time, EVENT) ~ Treatment * Sex + frailty(LINE, df = 6),
data = DesRes, dist = "weibull")
weibull.sta <- survreg(Surv(survival.time, EVENT) ~ Treatment * Sex + frailty(LINE, df = 6),
data = StaRes, dist = "weibull")
bind_rows(anova(weibull.des), anova(weibull.sta)) %>%
cbind(Parameter = c('Null', 'Treatment', 'Sex', '`frailty(LINE)`', 'Treatment x Sex')) %>%
mutate(across(1:5, round, 3)) %>%
mutate(star = ifelse(`Pr(>Chi)` < 0.05, "\\*", "")) %>%
select(Parameter, Df, `Resid. Df`, Deviance, `Pr(>Chi)`, star) %>%
filter(Parameter!='`frailty(LINE)`') %>%
rename(` ` = star) %>%
mutate(`Pr(>Chi)` = ifelse(`Pr(>Chi)` > 0.001, round(`Pr(>Chi)`, 3), '< 0.001')) %>%
kable() %>%
kable_styling() %>%
kable_styling(full_width = FALSE) %>%
group_rows("Desiccation", 1, 4) %>%
group_rows("Starvation", 5, 8)| Parameter | Df | Resid. Df | Deviance | Pr(>Chi) | |
|---|---|---|---|---|---|
| Desiccation | |||||
| Null | NA | 436.000 | NA | NA | NA |
| Treatment | 1.000 | 435.000 | 3.849 | 0.05 | |
| Sex | 1.000 | 434.000 | 154.904 | < 0.001 | * |
| Treatment x Sex | 1.010 | 429.020 | 8.682 | 0.003 | * |
| Starvation | |||||
| Null | NA | 478.000 | NA | NA | NA |
| Treatment | 1.000 | 477.000 | 2.681 | 0.102 | |
| Sex | 1.000 | 476.000 | 219.907 | < 0.001 | * |
| Treatment x Sex | 1.019 | 471.028 | 16.045 | < 0.001 | * |
We see equivocal support for a treatment effect for desiccation resistance and no effect for starvation resistance. For both assays there is support for a sex effect and a treatment x sex interaction.
Calculate hazard ratios
We can use the following equation to translate the AFT coefficients, \(\beta\), to a hazard ratio, \(\alpha\): \[ \beta = -\alpha * p \] where \(p\) is the shape parameter (\(1/\)scale parameter) (see here). The shape parameter describes the change in hazard over time where:
- \(p\) = 1: constant hazard
- \(p\) > 1: increasing hazard over time
- \(p\) < 1: decreasing hazard over time
# function to get hazard ratios and standard errors
hazR <- function(mod) {
b_coef = c(coefficients(summary(mod)))
coef = (b_coef * -1 * 1/mod$scale)
HazardRatio = exp(coef)
b_se = summary(mod)$table[, 2]
se = (b_se * -1 * 1/mod$scale)
HR.se = exp(se)
return(data.frame(round(cbind(HazardRatio, HR.se), 3)[-c(1,5), ]))
}For both desiccation and starvation resistance Polyandrous females live longer than Monogamy females (although not significantly so). Males die sooner than females, and Polyandry males die sooner than Monogamy males.
bind_rows(hazR(weibull.des), hazR(weibull.sta)) %>% as_tibble() %>%
cbind(Parameter = c('Treatment', 'Sex', 'Treatment x Sex')) %>%
select(Parameter, `Hazard ratio` = HazardRatio, `Std. Err.` = HR.se) %>%
kable() %>%
kable_styling() %>%
kable_styling(full_width = FALSE) %>%
group_rows("Desiccation", 1, 3) %>%
group_rows("Starvation", 4, 6)| Parameter | Hazard ratio | Std. Err. |
|---|---|---|
| Desiccation | ||
| Treatment | 0.736 | 0.354 |
| Sex | 2.872 | 0.866 |
| Treatment x Sex | 1.834 | 0.815 |
| Starvation | ||
| Treatment | 0.674 | 0.458 |
| Sex | 4.231 | 0.876 |
| Treatment x Sex | 2.134 | 0.830 |
Plot the predicted curves
par(mar = c(5,5,2,1), mfrow = c(1,2))
# Desiccation plot
# M female
curve(pweibull(x, scale = exp(coef(weibull.des)[1]), shape = 1/weibull.des$scale,
lower.tail = FALSE),
from = 0, to = max(na.omit(DesRes$survival.time)),
col = 'pink', ylab = expression(hat(S)(t)), xlab='t', lwd = 2,
main = "Desiccation resistance")
# P female
curve(pweibull(x, scale = exp(coef(weibull.des)[1] + coef(weibull.des)[2]),
shape = 1/weibull.des$scale,
lower.tail = FALSE),
from = 0, to = max(na.omit(DesRes$survival.time)),
add = T, lwd = 2, col = 'red')
# M male
curve(pweibull(x, scale = exp(coef(weibull.des)[1] + coef(weibull.des)[3]),
shape = 1/weibull.des$scale,
lower.tail = FALSE),
from = 0, to = max(na.omit(DesRes$survival.time)),
add = T, col = 'skyblue', lty = 2, lwd = 2)
# P male
curve(pweibull(x,
scale = exp(coef(weibull.des)[1] + coef(weibull.des)[2] + coef(weibull.des)[3] + coef(weibull.des)[4]),
shape = 1/weibull.des$scale,
lower.tail = FALSE), from = 0, to = max(na.omit(DesRes$survival.time)),
add=T, col = 'blue', lty = 2, lwd = 2)
legend("topright", c("M \u2640","M \u2642",'P \u2640','P \u2642'),
col = c("pink", "skyblue", "red", "blue"),
lty = 1:2,
lwd = 2,
bty = 'n'
)
# Starvation plot
# M female
curve(pweibull(x, scale = exp(coef(weibull.sta)[1]), shape = 1/weibull.sta$scale,
lower.tail = FALSE),
from = 0, to = max(na.omit(StaRes$survival.time)),
col = 'pink', ylab = expression(hat(S)(t)), xlab='t', lwd = 2,
main = "Starvation resistance")
# P female
curve(pweibull(x, scale = exp(coef(weibull.sta)[1] + coef(weibull.sta)[2]),
shape = 1/weibull.sta$scale,
lower.tail = FALSE),
from = 0, to = max(na.omit(StaRes$survival.time)),
add = T, lwd = 2, col = 'red')
# M male
curve(pweibull(x, scale = exp(coef(weibull.sta)[1] + coef(weibull.sta)[3]),
shape = 1/weibull.sta$scale,
lower.tail = FALSE),
from = 0, to = max(na.omit(StaRes$survival.time)),
add = T, col = 'skyblue', lty = 2, lwd = 2)
# P male
curve(pweibull(x,
scale = exp(coef(weibull.sta)[1] + coef(weibull.sta)[2] + coef(weibull.sta)[3] + coef(weibull.sta)[4]),
shape = 1/weibull.sta$scale,
lower.tail = FALSE), from = 0, to = max(na.omit(StaRes$survival.time)),
add=T, col = 'blue', lty = 2, lwd = 2)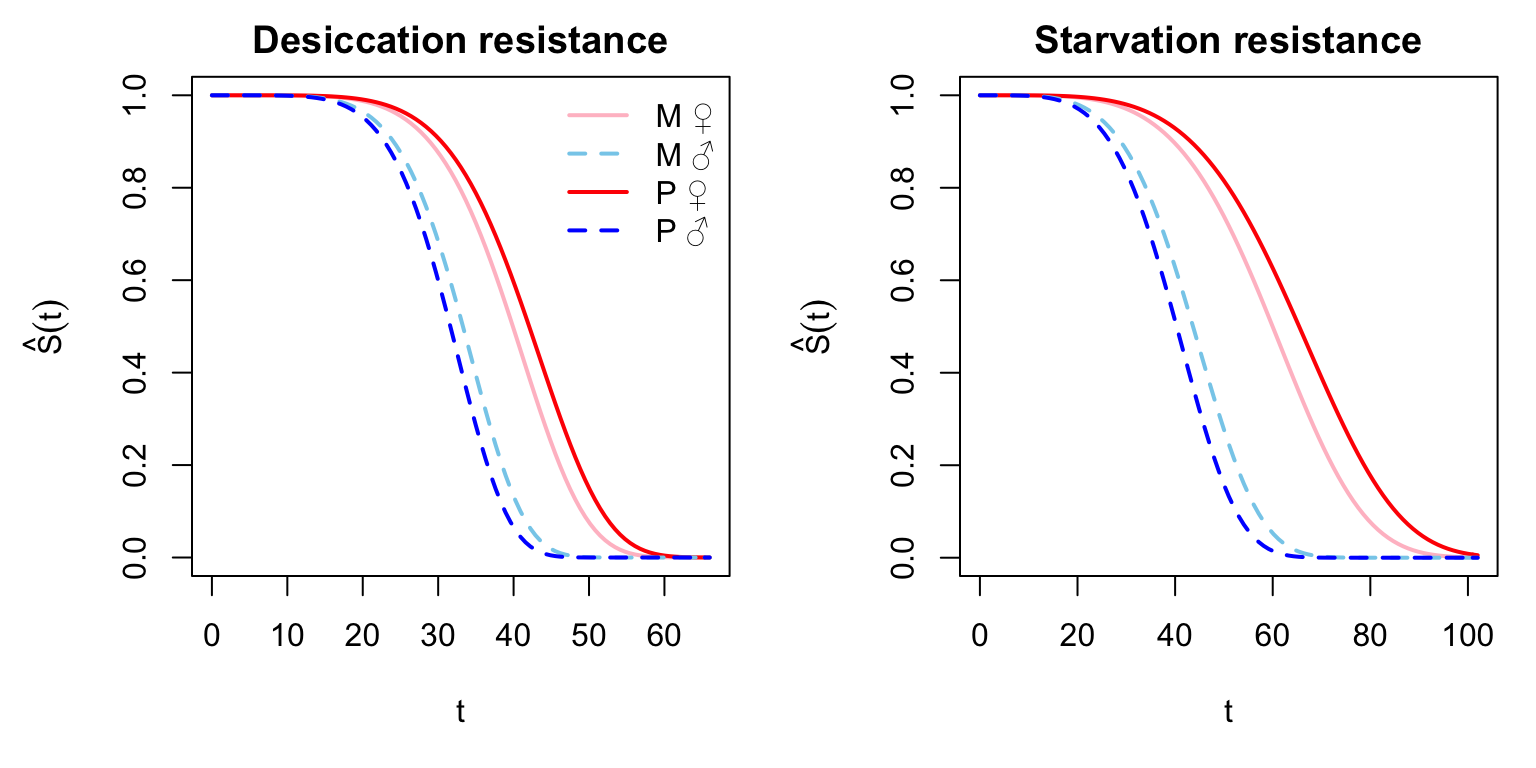
Figure X: Here we plot the model predicted survival functions (\(\hat{S}_{(t)}\)) for each sex and treatment for the two assays.
Fit the brms survival models for desiccation and starvation resistance
Here I have attempted to fit the analyses using brms. While the results of the models are qualitatively similar (sex and treatment x sex effects), I am not sure my calculation of hazard ratios is correct. Documentation for fitting survival analysis in brms still fairly sparse. Altogether I think using the simpler AFT models is sufficient.
if(!file.exists("output/des_brm.rds")){ # if the model doesn't exist fit it, else load it
des_brm <- brm(survival.time | cens(1 - EVENT) ~ Treatment * Sex + (1|LINE),
iter = 5000, chains = 4, cores = 4,
control = list(max_treedepth = 20,
adapt_delta = 0.999),
data = DesRes, family = weibull())
saveRDS(des_brm, "output/des_brm.rds")
} else {
des_brm <- readRDS('output/des_brm.rds')
}
if(!file.exists("output/sta_brm.rds")){ # if the model doesn't exist fit it, else load it
sta_brm <- brm(survival.time | cens(EVENT) ~ Treatment * Sex + (1|LINE),
iter = 5000, chains = 4, cores = 4,
control = list(max_treedepth = 20,
adapt_delta = 0.999),
# brm uses 0 = event, 1 = censor so need to recode
data = StaRes %>% mutate(EVENT = if_else(EVENT == 1, 0, 1)),
family = weibull())
saveRDS(sta_brm, "output/sta_brm.rds")
} else {
sta_brm <- readRDS('output/sta_brm.rds')
}
# function to get hazard ratios and standard errors - needs adapting
hazR <- function(mod) {
a = c(fixef(mod)[, 1])
coef = (a * -1 * 1/summary(mod)$spec_pars[1])
HazardRatio = exp(coef)
b = c(fixef(mod)[, 2])
se = (b * -1 * 1/summary(mod)$spec_pars[1])
HR.se = exp(se)
return(data.frame(round(cbind(HazardRatio, HR.se), 3)[-c(1,5), ]))
}Hypothesis testing
des_test <- bind_rows(
hypothesis(des_brm, 'TreatmentP = 0')$hypothesis,
hypothesis(des_brm, 'Sexm = 0')$hypothesis,
hypothesis(des_brm, 'TreatmentP:Sexm = 0')$hypothesis
) %>%
mutate(Parameter = c('Polandry', 'Male', 'Polyandry x Male'),
across(2:5, round, 3)) %>%
#select(-Hypothesis) %>%
relocate(Parameter, Estimate, Est.Error, CI.Lower, CI.Upper, Star)
sta_test <- bind_rows(
hypothesis(sta_brm, 'TreatmentP = 0')$hypothesis,
hypothesis(sta_brm, 'Sexm = 0')$hypothesis,
hypothesis(sta_brm, 'TreatmentP:Sexm = 0')$hypothesis
) %>%
mutate(Parameter = c('Polandry', 'Male', 'Polyandry x Male'),
across(2:5, round, 3)) %>%
#select(-Hypothesis) %>%
relocate(Parameter, Estimate, Est.Error, CI.Lower, CI.Upper, Star)
des_pvals <- bayestestR::p_direction(des_brm) %>%
as.data.frame() %>%
mutate(vars = map_chr(str_split(Parameter, "_"), ~ .x[2]),
p_val = 1 - pd,
star = ifelse(p_val < 0.05, "\\*", "")) %>%
select(vars, p_val, star)
sta_pvals <- bayestestR::p_direction(sta_brm) %>%
as.data.frame() %>%
mutate(vars = map_chr(str_split(Parameter, "_"), ~ .x[2]),
p_val = 1 - pd,
star = ifelse(p_val < 0.05, "\\*", "")) %>%
select(vars, p_val, star)
bind_rows(
des_test %>%
mutate(vars = c('TreatmentP', 'Sexm', 'TreatmentP.Sexm')) %>%
left_join(des_pvals %>% filter(vars != 'Intercept'),
by = c("vars")) %>%
select(Parameter, Estimate, Est.Error, CI.Lower, CI.Upper, `p` = p_val, star),
sta_test %>%
mutate(vars = c('TreatmentP', 'Sexm', 'TreatmentP.Sexm')) %>%
left_join(sta_pvals %>% filter(vars != 'Intercept'),
by = c("vars")) %>%
select(Parameter, Estimate, Est.Error, CI.Lower, CI.Upper, `p` = p_val, star)
) %>%
mutate(p = ifelse(p > 0.001, round(p, 3), '< 0.001')) %>%
rename(` ` = star) %>%
kable() %>%
kable_styling(full_width = FALSE) %>%
group_rows("Desiccation", 1, 3) %>%
group_rows("Starvation", 4, 6)| Parameter | Estimate | Est.Error | CI.Lower | CI.Upper | p | |
|---|---|---|---|---|---|---|
| Desiccation | ||||||
| Polandry | 0.054 | 0.107 | -0.151 | 0.278 | 0.272 | |
| Male | -0.183 | 0.025 | -0.232 | -0.134 | < 0.001 | * |
| Polyandry x Male | -0.104 | 0.036 | -0.173 | -0.034 | 0.002 | * |
| Starvation | ||||||
| Polandry | 0.101 | 0.147 | -0.147 | 0.352 | 0.182 | |
| Male | -0.318 | 0.030 | -0.377 | -0.259 | < 0.001 | * |
| Polyandry x Male | -0.167 | 0.042 | -0.251 | -0.085 | < 0.001 | * |
Plot posteriors.
# get posterior predictions
post_des <- posterior_samples(des_brm) %>%
as_tibble() %>%
select(contains("b_"), -contains("Intercept")) %>%
mutate(draw = 1:n()) %>%
pivot_longer(-draw) %>%
mutate(key = str_remove_all(name, "b_"))
post_sta <- posterior_samples(sta_brm) %>%
as_tibble() %>%
select(contains("b_"), -contains("Intercept")) %>%
mutate(draw = 1:n()) %>%
pivot_longer(-draw) %>%
mutate(key = str_remove_all(name, "b_"))Plotting the hazard ratio gives an estimate of the effect size, so we can plot the effect sizes from both experiments together.
bind_rows(post_des %>% mutate(var = 'Desiccation'),
post_sta %>% mutate(var = 'Starvation')) %>%
mutate(HR = exp(value)) %>%
ggplot(aes(x = HR, y = key, fill = var)) +
geom_vline(xintercept = 1, linetype = 2) +
stat_halfeye(alpha = .8, position = position_dodge(width = .1)) +
scale_fill_brewer(palette = "Dark2") +
coord_cartesian(xlim = c(0.6, 1.5)) +
labs(x = "Hazard ratio", y = "Model parameter") +
theme_ridges() +
theme(#legend.position = "none",
legend.title = element_blank()) +
NULL
| Version | Author | Date |
|---|---|---|
| 96d1188 | Martin Garlovsky | 2020-12-13 |
Summary of the results of the full model
bind_rows(
fixef(des_brm) %>% data.frame() %>% rownames_to_column(),
fixef(sta_brm) %>% data.frame() %>% rownames_to_column()) %>%
as_tibble() %>%
select(Parameter = rowname, Estimate, Est.Error, Q2.5, Q97.5) %>%
mutate(Parameter = rep(c('Intercept', 'Polandry', 'Male', 'Polandry x Male'), 2),
#across(2:5, exp), # this will convert to hazard ratio?
across(2:5, round, 3)) %>%
kable() %>%
kable_styling() %>%
kable_styling(full_width = FALSE) %>%
group_rows("Desiccation", 1, 4) %>%
group_rows("Starvation", 5, 8)| Parameter | Estimate | Est.Error | Q2.5 | Q97.5 |
|---|---|---|---|---|
| Desiccation | ||||
| Intercept | 3.670 | 0.076 | 3.515 | 3.819 |
| Polandry | 0.054 | 0.107 | -0.151 | 0.278 |
| Male | -0.183 | 0.025 | -0.232 | -0.134 |
| Polandry x Male | -0.104 | 0.036 | -0.173 | -0.034 |
| Starvation | ||||
| Intercept | 4.071 | 0.101 | 3.889 | 4.249 |
| Polandry | 0.101 | 0.147 | -0.147 | 0.352 |
| Male | -0.318 | 0.030 | -0.377 | -0.259 |
| Polandry x Male | -0.167 | 0.042 | -0.251 | -0.085 |
Extract posterior estimates and plot
# wrangle
des_fit <-
fixef(des_brm) %>%
data.frame() %>%
rownames_to_column() %>%
mutate(param = str_remove(rowname, "m|P")) %>%
tidyr::expand(nesting(Estimate, Q2.5, Q97.5, param),
survival.time = 0:100) %>%
mutate(m = 1 - pexp(survival.time, rate = 1 / exp(Estimate)),
ll = 1 - pexp(survival.time, rate = 1 / exp(Q2.5)),
ul = 1 - pexp(survival.time, rate = 1 / exp(Q97.5)))
# plot!
des_fit %>%
ggplot(aes(x = survival.time)) +
# geom_hline(yintercept = .5, linetype = 3, aes(color = param)) +
geom_ribbon(aes(ymin = ll, ymax = ul, fill = param),
alpha = 1/2) +
# geom_line(aes(y = m, aes(color = cols))) +
# scale_fill_manual(values = wes_palette("Moonrise2")[c(4, 1)], breaks = NULL) +
# scale_color_manual(values = wes_palette("Moonrise2")[c(4, 1)], breaks = NULL) +
# scale_y_continuous("proportion remaining", , breaks = c(0, .5, 1), limits = c(0, 1)) +
labs(x = "Survival time (hours)") +
NULL
| Version | Author | Date |
|---|---|---|
| 96d1188 | Martin Garlovsky | 2020-12-13 |
des_fit %>%
ggplot(aes(x = survival.time, y = m, colour = param)) +
geom_line() +
geom_ribbon(aes(ymin = ll, ymax = ul, fill = param), alpha = 1/2) +
scale_colour_manual(values = c("pink", "red", "skyblue", "blue")) +
scale_fill_manual(values = c("pink", "red", "skyblue", "blue")) +
theme_bw() +
theme() +
NULL
| Version | Author | Date |
|---|---|---|
| 96d1188 | Martin Garlovsky | 2020-12-13 |
sessionInfo()R version 4.0.3 (2020-10-10) Platform: x86_64-apple-darwin17.0 (64-bit) Running under: macOS Catalina 10.15.4 Matrix products: default BLAS: /Library/Frameworks/R.framework/Versions/4.0/Resources/lib/libRblas.dylib LAPACK: /Library/Frameworks/R.framework/Versions/4.0/Resources/lib/libRlapack.dylib locale: [1] en_AU.UTF-8/en_AU.UTF-8/en_AU.UTF-8/C/en_AU.UTF-8/en_AU.UTF-8 attached base packages: [1] stats graphics grDevices utils datasets methods base other attached packages: [1] knitrhooks_0.0.4 knitr_1.30 kableExtra_1.1.0 ggridges_0.5.2 [5] tidybayes_2.0.3 brms_2.14.4 Rcpp_1.0.4.6 lme4_1.1-23 [9] Matrix_1.2-18 coxme_2.2-16 bdsmatrix_1.3-4 survival_3.2-7 [13] forcats_0.5.0 stringr_1.4.0 dplyr_1.0.0 purrr_0.3.4 [17] readr_1.3.1 tidyr_1.1.0 tibble_3.0.1 ggplot2_3.3.2 [21] tidyverse_1.3.0 workflowr_1.6.2 loaded via a namespace (and not attached): [1] readxl_1.3.1 backports_1.1.7 plyr_1.8.6 [4] igraph_1.2.5 svUnit_1.0.3 splines_4.0.3 [7] crosstalk_1.1.0.1 TH.data_1.0-10 rstantools_2.1.1 [10] inline_0.3.15 digest_0.6.25 htmltools_0.5.0 [13] rsconnect_0.8.16 fansi_0.4.1 magrittr_2.0.1 [16] openxlsx_4.1.5 modelr_0.1.8 RcppParallel_5.0.1 [19] matrixStats_0.56.0 xts_0.12-0 sandwich_2.5-1 [22] prettyunits_1.1.1 colorspace_1.4-1 blob_1.2.1 [25] rvest_0.3.5 haven_2.3.1 xfun_0.19 [28] callr_3.4.3 crayon_1.3.4 jsonlite_1.7.0 [31] zoo_1.8-8 glue_1.4.2 survminer_0.4.8 [34] gtable_0.3.0 emmeans_1.4.7 webshot_0.5.2 [37] V8_3.4.0 car_3.0-8 pkgbuild_1.0.8 [40] rstan_2.21.2 abind_1.4-5 scales_1.1.1 [43] mvtnorm_1.1-0 DBI_1.1.0 rstatix_0.5.0 [46] miniUI_0.1.1.1 viridisLite_0.3.0 xtable_1.8-4 [49] foreign_0.8-80 km.ci_0.5-2 stats4_4.0.3 [52] StanHeaders_2.21.0-3 DT_0.13 htmlwidgets_1.5.1 [55] httr_1.4.1 threejs_0.3.3 arrayhelpers_1.1-0 [58] ellipsis_0.3.1 farver_2.0.3 pkgconfig_2.0.3 [61] loo_2.3.1 dbplyr_1.4.4 labeling_0.3 [64] tidyselect_1.1.0 rlang_0.4.6 reshape2_1.4.4 [67] later_1.0.0 munsell_0.5.0 cellranger_1.1.0 [70] tools_4.0.3 cli_2.0.2 generics_0.0.2 [73] broom_0.5.6 evaluate_0.14 fastmap_1.0.1 [76] yaml_2.2.1 processx_3.4.2 fs_1.4.1 [79] zip_2.0.4 survMisc_0.5.5 nlme_3.1-149 [82] whisker_0.4 mime_0.9 projpred_2.0.2 [85] xml2_1.3.2 compiler_4.0.3 bayesplot_1.7.2 [88] shinythemes_1.1.2 rstudioapi_0.11 gamm4_0.2-6 [91] curl_4.3 ggsignif_0.6.0 reprex_0.3.0 [94] statmod_1.4.34 stringi_1.5.3 highr_0.8 [97] ps_1.3.3 Brobdingnag_1.2-6 lattice_0.20-41 [100] nloptr_1.2.2.1 markdown_1.1 KMsurv_0.1-5 [103] shinyjs_1.1 vctrs_0.3.0 pillar_1.4.4 [106] lifecycle_0.2.0 bridgesampling_1.0-0 estimability_1.3 [109] insight_0.8.4 data.table_1.12.8 httpuv_1.5.3.1 [112] R6_2.4.1 promises_1.1.0 rio_0.5.16 [115] gridExtra_2.3 codetools_0.2-16 boot_1.3-25 [118] colourpicker_1.0 MASS_7.3-53 gtools_3.8.2 [121] assertthat_0.2.1 rprojroot_1.3-2 withr_2.2.0 [124] shinystan_2.5.0 multcomp_1.4-13 bayestestR_0.6.0 [127] mgcv_1.8-33 parallel_4.0.3 hms_0.5.3 [130] grid_4.0.3 coda_0.19-3 minqa_1.2.4 [133] rmarkdown_2.5 carData_3.0-4 ggpubr_0.3.0 [136] git2r_0.27.1 shiny_1.4.0.2 lubridate_1.7.8 [139] base64enc_0.1-3 dygraphs_1.1.1.6