SCZ 2018 - Brain_Cerebellar_Hemisphere
sheng Qian
2021-2-6
Last updated: 2022-05-19
Checks: 5 2
Knit directory: cTWAS_analysis/
This reproducible R Markdown analysis was created with workflowr (version 1.7.0). The Checks tab describes the reproducibility checks that were applied when the results were created. The Past versions tab lists the development history.
The R Markdown file has unstaged changes. To know which version of the R Markdown file created these results, you’ll want to first commit it to the Git repo. If you’re still working on the analysis, you can ignore this warning. When you’re finished, you can run wflow_publish to commit the R Markdown file and build the HTML.
Great job! The global environment was empty. Objects defined in the global environment can affect the analysis in your R Markdown file in unknown ways. For reproduciblity it’s best to always run the code in an empty environment.
The command set.seed(20211220) was run prior to running the code in the R Markdown file. Setting a seed ensures that any results that rely on randomness, e.g. subsampling or permutations, are reproducible.
Great job! Recording the operating system, R version, and package versions is critical for reproducibility.
Nice! There were no cached chunks for this analysis, so you can be confident that you successfully produced the results during this run.
Using absolute paths to the files within your workflowr project makes it difficult for you and others to run your code on a different machine. Change the absolute path(s) below to the suggested relative path(s) to make your code more reproducible.
| absolute | relative |
|---|---|
| /project2/xinhe/shengqian/cTWAS/cTWAS_analysis/data/ | data |
| /project2/xinhe/shengqian/cTWAS/cTWAS_analysis/code/ctwas_config.R | code/ctwas_config.R |
Great! You are using Git for version control. Tracking code development and connecting the code version to the results is critical for reproducibility.
The results in this page were generated with repository version be614ed. See the Past versions tab to see a history of the changes made to the R Markdown and HTML files.
Note that you need to be careful to ensure that all relevant files for the analysis have been committed to Git prior to generating the results (you can use wflow_publish or wflow_git_commit). workflowr only checks the R Markdown file, but you know if there are other scripts or data files that it depends on. Below is the status of the Git repository when the results were generated:
Ignored files:
Ignored: .Rhistory
Ignored: .ipynb_checkpoints/
Untracked files:
Untracked: G_list.RData
Untracked: Rplot.png
Untracked: SCZ_annotation.xlsx
Untracked: analysis/.ipynb_checkpoints/
Untracked: code/.ipynb_checkpoints/
Untracked: code/AF_out/
Untracked: code/Autism_out/
Untracked: code/BMI_S_out/
Untracked: code/BMI_out/
Untracked: code/Glucose_out/
Untracked: code/LDL_S_out/
Untracked: code/SCZ_2014_EUR_out/
Untracked: code/SCZ_2018_S_out/
Untracked: code/SCZ_2018_out/
Untracked: code/SCZ_2020_Single_out/
Untracked: code/SCZ_2020_out/
Untracked: code/SCZ_S_out/
Untracked: code/SCZ_out/
Untracked: code/T2D_out/
Untracked: code/ctwas_config.R
Untracked: code/mapping.R
Untracked: code/out/
Untracked: code/process_scz_2018_snps.R
Untracked: code/run_AF_analysis.sbatch
Untracked: code/run_AF_analysis.sh
Untracked: code/run_AF_ctwas_rss_LDR.R
Untracked: code/run_Autism_analysis.sbatch
Untracked: code/run_Autism_analysis.sh
Untracked: code/run_Autism_ctwas_rss_LDR.R
Untracked: code/run_BMI_analysis.sbatch
Untracked: code/run_BMI_analysis.sh
Untracked: code/run_BMI_analysis_S.sbatch
Untracked: code/run_BMI_analysis_S.sh
Untracked: code/run_BMI_ctwas_rss_LDR.R
Untracked: code/run_BMI_ctwas_rss_LDR_S.R
Untracked: code/run_Glucose_analysis.sbatch
Untracked: code/run_Glucose_analysis.sh
Untracked: code/run_Glucose_ctwas_rss_LDR.R
Untracked: code/run_LDL_analysis_S.sbatch
Untracked: code/run_LDL_analysis_S.sh
Untracked: code/run_LDL_ctwas_rss_LDR_S.R
Untracked: code/run_SCZ_2014_EUR_analysis.sbatch
Untracked: code/run_SCZ_2014_EUR_analysis.sh
Untracked: code/run_SCZ_2014_EUR_ctwas_rss_LDR.R
Untracked: code/run_SCZ_2018_analysis.sbatch
Untracked: code/run_SCZ_2018_analysis.sh
Untracked: code/run_SCZ_2018_analysis_S.sbatch
Untracked: code/run_SCZ_2018_analysis_S.sh
Untracked: code/run_SCZ_2018_ctwas_rss_LDR.R
Untracked: code/run_SCZ_2018_ctwas_rss_LDR_S.R
Untracked: code/run_SCZ_2020_Single_analysis.sbatch
Untracked: code/run_SCZ_2020_Single_analysis.sh
Untracked: code/run_SCZ_2020_Single_ctwas_rss_LDR.R
Untracked: code/run_SCZ_2020_analysis.sbatch
Untracked: code/run_SCZ_2020_analysis.sh
Untracked: code/run_SCZ_2020_ctwas_rss_LDR.R
Untracked: code/run_SCZ_analysis.sbatch
Untracked: code/run_SCZ_analysis.sh
Untracked: code/run_SCZ_analysis_S.sbatch
Untracked: code/run_SCZ_analysis_S.sh
Untracked: code/run_SCZ_ctwas_rss_LDR.R
Untracked: code/run_SCZ_ctwas_rss_LDR_S.R
Untracked: code/run_T2D_analysis.sbatch
Untracked: code/run_T2D_analysis.sh
Untracked: code/run_T2D_ctwas_rss_LDR.R
Untracked: code/wflow_build.R
Untracked: code/wflow_build.sbatch
Untracked: data/.ipynb_checkpoints/
Untracked: data/GO_Terms/
Untracked: data/PGC3_SCZ_wave3_public.v2.tsv
Untracked: data/SCZ/
Untracked: data/SCZ_2014_EUR/
Untracked: data/SCZ_2018/
Untracked: data/SCZ_2018_S/
Untracked: data/SCZ_2020/
Untracked: data/SCZ_S/
Untracked: data/Supplementary Table 15 - MAGMA.xlsx
Untracked: data/Supplementary Table 20 - Prioritised Genes.xlsx
Untracked: data/T2D/
Untracked: data/UKBB/
Untracked: data/UKBB_SNPs_Info.text
Untracked: data/gene_OMIM.txt
Untracked: data/gene_pip_0.8.txt
Untracked: data/mashr_Heart_Atrial_Appendage.db
Untracked: data/mashr_sqtl/
Untracked: data/scz_2018.RDS
Untracked: data/summary_known_genes_annotations.xlsx
Untracked: data/untitled.txt
Untracked: top_genes_32.txt
Untracked: top_genes_37.txt
Untracked: top_genes_43.txt
Untracked: top_genes_54.txt
Untracked: top_genes_81.txt
Untracked: z_snp_pos_SCZ.RData
Untracked: z_snp_pos_SCZ_2014_EUR.RData
Untracked: z_snp_pos_SCZ_2018.RData
Untracked: z_snp_pos_SCZ_2020.RData
Unstaged changes:
Deleted: analysis/BMI_S_results.Rmd
Modified: analysis/SCZ_2018_Brain_Amygdala_S.Rmd
Modified: analysis/SCZ_2018_Brain_Anterior_cingulate_cortex_BA24_S.Rmd
Modified: analysis/SCZ_2018_Brain_Caudate_basal_ganglia_S.Rmd
Modified: analysis/SCZ_2018_Brain_Cerebellar_Hemisphere_S.Rmd
Modified: analysis/SCZ_2018_Brain_Cerebellum_S.Rmd
Modified: analysis/SCZ_2018_Brain_Cortex_S.Rmd
Modified: analysis/SCZ_2018_Brain_Frontal_Cortex_BA9_S.Rmd
Modified: analysis/SCZ_2018_Brain_Hippocampus_S.Rmd
Modified: analysis/SCZ_2018_Brain_Hypothalamus_S.Rmd
Modified: analysis/SCZ_2018_Brain_Nucleus_accumbens_basal_ganglia_S.Rmd
Modified: analysis/SCZ_2018_Brain_Putamen_basal_ganglia_S.Rmd
Modified: analysis/SCZ_2018_Brain_Spinal_cord_cervical_c-1_S.Rmd
Modified: analysis/SCZ_2018_Brain_Substantia_nigra_S.Rmd
Modified: analysis/ttt.Rmd
Note that any generated files, e.g. HTML, png, CSS, etc., are not included in this status report because it is ok for generated content to have uncommitted changes.
These are the previous versions of the repository in which changes were made to the R Markdown (analysis/SCZ_2018_Brain_Cerebellar_Hemisphere_S.Rmd) and HTML (docs/SCZ_2018_Brain_Cerebellar_Hemisphere_S.html) files. If you’ve configured a remote Git repository (see ?wflow_git_remote), click on the hyperlinks in the table below to view the files as they were in that past version.
| File | Version | Author | Date | Message |
|---|---|---|---|---|
| Rmd | be614ed | sq-96 | 2022-05-19 | update |
| html | be614ed | sq-96 | 2022-05-19 | update |
| Rmd | 7d08c9b | sq-96 | 2022-05-18 | update |
| html | 7d08c9b | sq-96 | 2022-05-18 | update |
| Rmd | 2749be9 | sq-96 | 2022-05-12 | update |
| html | 2749be9 | sq-96 | 2022-05-12 | update |
| html | 011327d | sq-96 | 2022-05-12 | update |
| Rmd | 6c6abbd | sq-96 | 2022-05-12 | update |
library(reticulate)
use_python("/scratch/midway2/shengqian/miniconda3/envs/PythonForR/bin/python",required=T)Weight QC
#number of imputed weights
nrow(qclist_all)[1] 26564#number of imputed weights by chromosome
table(qclist_all$chr)
1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16
2520 1814 1594 973 1137 1377 1526 911 1106 1166 1579 1419 520 921 928 1179
17 18 19 20 21 22
1880 325 1895 891 51 852 #number of imputed weights without missing variants
sum(qclist_all$nmiss==0)[1] 23201#proportion of imputed weights without missing variants
mean(qclist_all$nmiss==0)[1] 0.8734INFO:numexpr.utils:Note: NumExpr detected 56 cores but "NUMEXPR_MAX_THREADS" not set, so enforcing safe limit of 8.finish
Attaching package: 'dplyr'The following objects are masked from 'package:stats':
filter, lagThe following objects are masked from 'package:base':
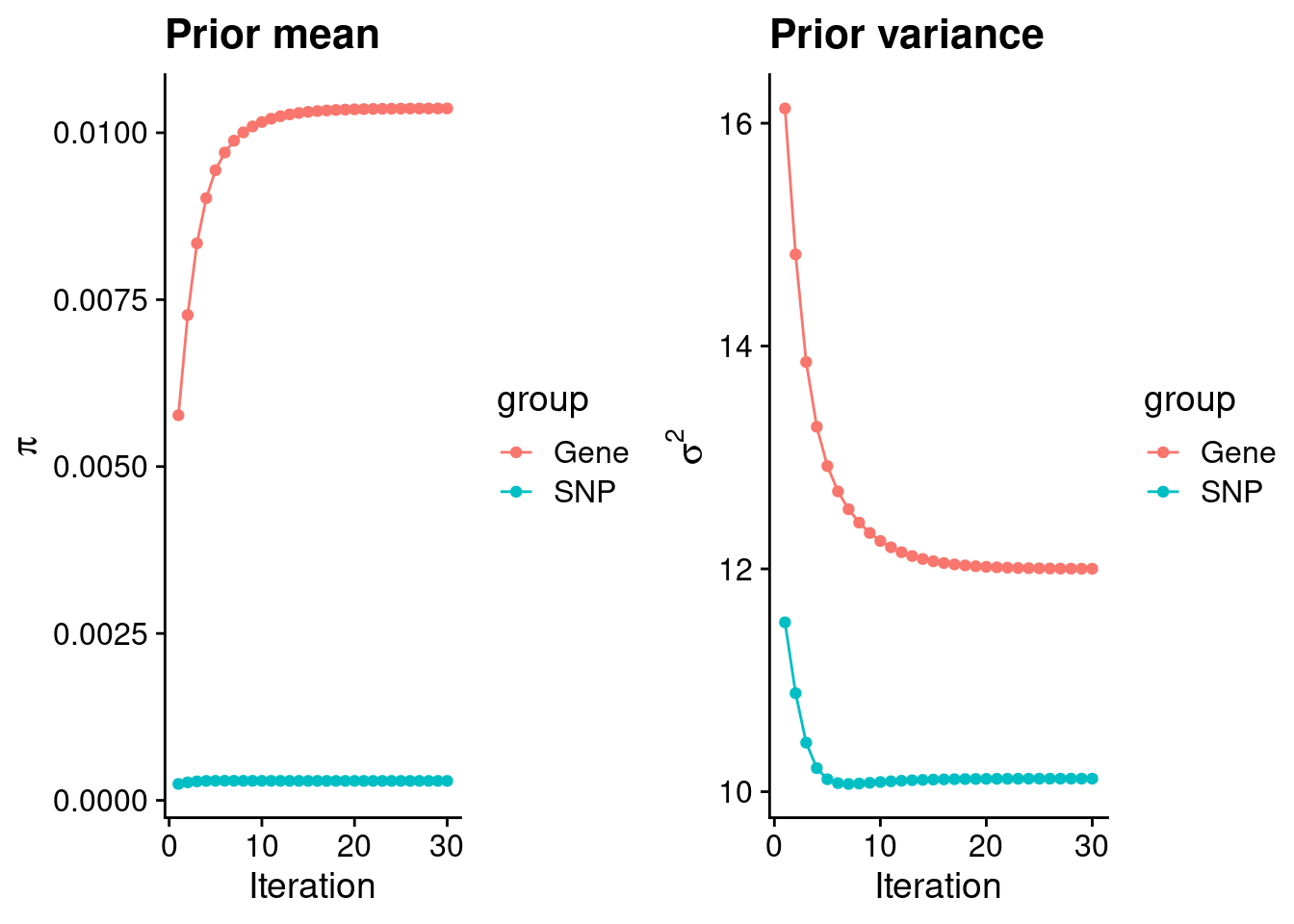
intersect, setdiff, setequal, unionCheck convergence of parameters
| Version | Author | Date |
|---|---|---|
| 2749be9 | sq-96 | 2022-05-12 |
gene snp
0.0103642 0.0002912 gene snp
12.00 10.12 [1] 105318[1] 7860 6309950 gene snp
0.009283 0.176540 [1] 0.01795 1.05335Genes with highest PIPs
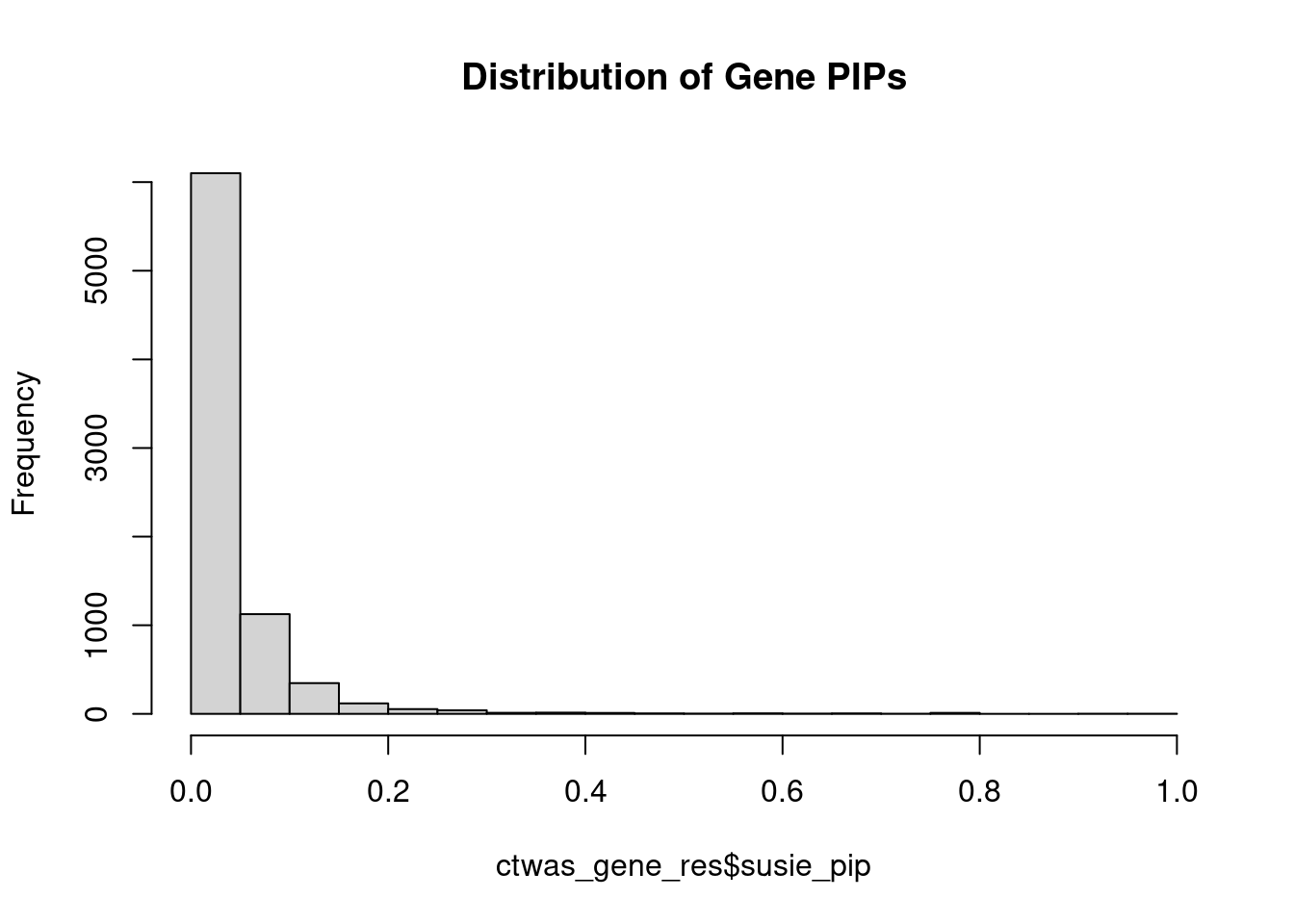
genename region_tag susie_pip mu2 PVE z num_intron
5293 R3HDM2 12_36 0.9766 43.83 4.119e-04 6.634 9
6205 SLC8B1 12_68 0.9706 28.59 3.598e-04 -4.047 11
3651 LPCAT4 15_10 0.9325 25.36 2.153e-04 4.892 3
842 BUB1B-PAK6 15_14 0.9242 29.86 2.437e-04 -5.588 2
2038 DPYSL3 5_86 0.7910 21.54 1.280e-04 -4.157 1
2719 GIGYF2 2_137 0.7882 56.96 4.059e-04 -8.128 6
4513 NTRK3 15_41 0.7875 24.09 1.426e-04 4.457 3
1631 CRTAP 3_24 0.7796 20.88 1.215e-04 3.929 2
7078 TSNARE1 8_93 0.7781 34.12 1.701e-04 6.364 10
6263 SMYD2 1_108 0.7713 21.62 1.225e-04 -3.952 2
2396 FAM177A1 14_9 0.7657 24.30 1.707e-04 -4.872 12
1039 CAMKK2 12_74 0.7621 35.78 1.702e-04 4.159 6
751 BDNF 11_19 0.7588 23.84 1.316e-04 4.348 3
7567 ZDHHC20 13_2 0.7584 25.00 1.400e-04 -4.832 3
6353 SPECC1 17_16 0.7548 25.87 1.409e-04 -4.822 2
3097 HSPA9 5_82 0.7412 25.57 1.334e-04 5.633 1
294 AKT3 1_128 0.7158 35.12 1.906e-04 6.266 6
2902 GTF2A1 14_39 0.6708 24.76 1.117e-04 4.550 2
1662 CTB-31O20.2 19_3 0.6660 23.34 9.828e-05 4.456 1
4355 NFATC3 16_36 0.6648 28.83 1.219e-04 -5.480 3
num_sqtl
5293 11
6205 12
3651 4
842 2
2038 1
2719 6
4513 3
1631 2
7078 10
6263 2
2396 13
1039 8
751 3
7567 4
6353 2
3097 1
294 6
2902 3
1662 1
4355 3Genes with highest PVE
genename region_tag susie_pip mu2 PVE z num_intron
7339 VARS 6_26 0.3832 628.56 0.0008763 -11.620 2
456 APOM 6_26 0.2790 626.01 0.0007298 11.590 3
837 BTN3A1 6_20 0.6226 145.15 0.0005473 13.091 7
5293 R3HDM2 12_36 0.9766 43.83 0.0004119 6.634 9
2719 GIGYF2 2_137 0.7882 56.96 0.0004059 -8.128 6
6205 SLC8B1 12_68 0.9706 28.59 0.0003598 -4.047 11
842 BUB1B-PAK6 15_14 0.9242 29.86 0.0002437 -5.588 2
3651 LPCAT4 15_10 0.9325 25.36 0.0002153 4.892 3
294 AKT3 1_128 0.7158 35.12 0.0001906 6.266 6
5994 SF3B1 2_117 0.4462 45.85 0.0001751 -7.053 3
2396 FAM177A1 14_9 0.7657 24.30 0.0001707 -4.872 12
7005 TRANK1 3_27 0.6544 38.76 0.0001707 -6.365 6
1039 CAMKK2 12_74 0.7621 35.78 0.0001702 4.159 6
7078 TSNARE1 8_93 0.7781 34.12 0.0001701 6.364 10
4513 NTRK3 15_41 0.7875 24.09 0.0001426 4.457 3
6353 SPECC1 17_16 0.7548 25.87 0.0001409 -4.822 2
7567 ZDHHC20 13_2 0.7584 25.00 0.0001400 -4.832 3
3097 HSPA9 5_82 0.7412 25.57 0.0001334 5.633 1
751 BDNF 11_19 0.7588 23.84 0.0001316 4.348 3
2038 DPYSL3 5_86 0.7910 21.54 0.0001280 -4.157 1
num_sqtl
7339 2
456 4
837 8
5293 11
2719 6
6205 12
842 2
3651 4
294 6
5994 3
2396 13
7005 6
1039 8
7078 10
4513 3
6353 2
7567 4
3097 1
751 3
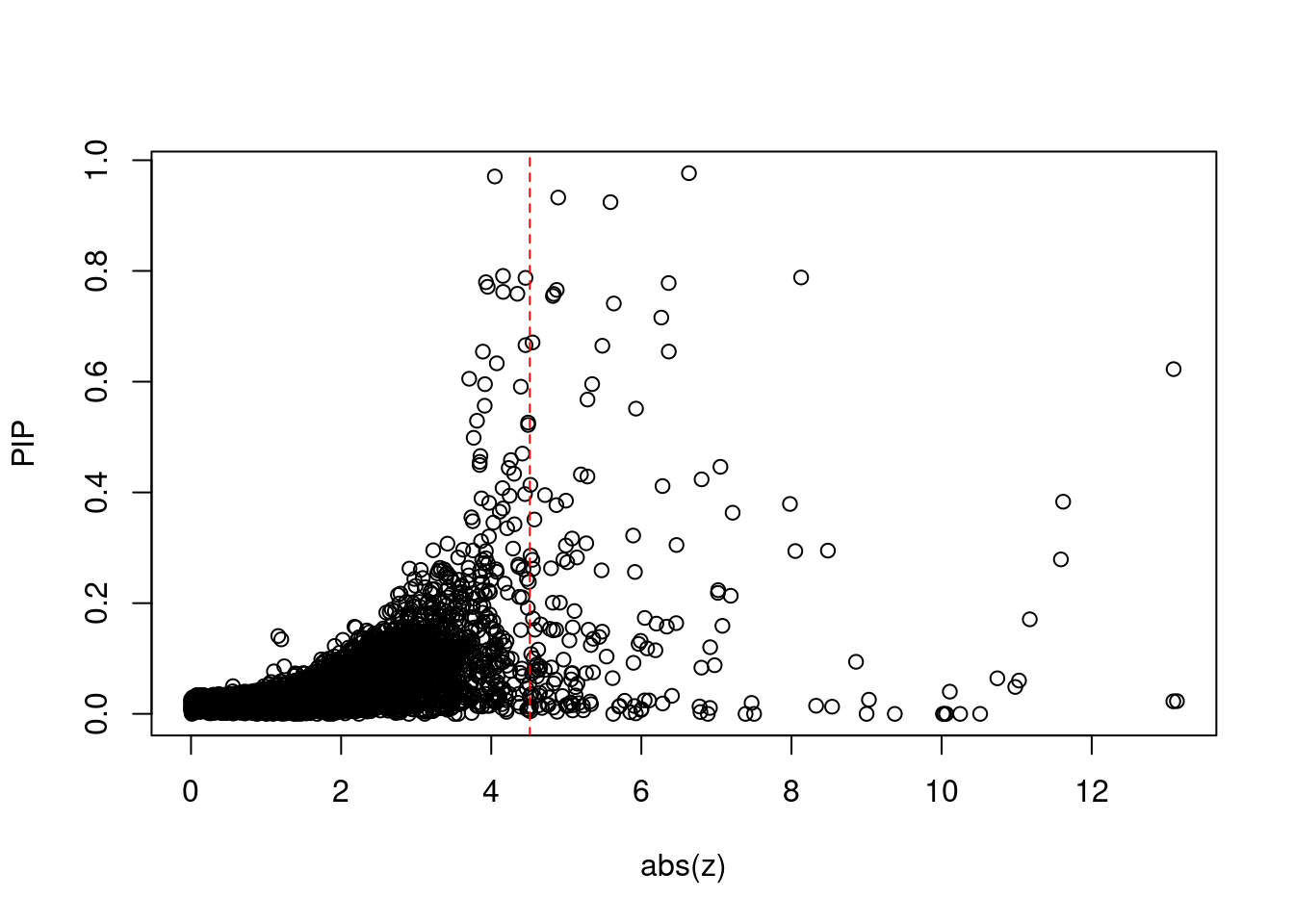
2038 1Comparing z scores and PIPs
[1] 0.02176 genename region_tag susie_pip mu2 PVE z num_intron num_sqtl
7606 ZKSCAN3 6_22 2.302e-02 160.21 1.217e-06 -13.135 4 4
837 BTN3A1 6_20 6.226e-01 145.15 5.473e-04 13.091 7 8
4797 PGBD1 6_22 2.270e-02 159.13 1.708e-06 -13.087 5 7
7339 VARS 6_26 3.832e-01 628.56 8.763e-04 -11.620 2 2
456 APOM 6_26 2.790e-01 626.01 7.298e-04 11.590 3 4
1851 DDR1 6_25 1.708e-01 101.78 5.658e-05 -11.175 4 4
964 C6orf136 6_24 6.024e-02 80.18 5.525e-06 -11.031 2 2
2559 FLOT1 6_24 4.864e-02 78.83 1.273e-05 10.981 8 8
838 BTN3A2 6_20 6.427e-02 94.90 4.458e-06 -10.743 5 7
1781 CYP21A2 6_26 5.976e-06 607.99 2.062e-13 -10.513 1 2
699 BAG6 6_26 1.969e-09 500.57 5.529e-20 10.247 9 9
835 BTN2A1 6_20 4.016e-02 84.19 1.707e-06 10.110 7 7
5104 PPT2 6_26 5.412e-12 466.36 1.297e-25 10.061 7 9
2138 EGFL8 6_26 4.301e-12 465.72 8.201e-26 10.036 6 7
5165 PRRT1 6_26 3.762e-12 464.63 6.243e-26 -10.018 1 1
2850 GPSM3 6_26 1.178e-13 416.63 1.098e-28 -9.377 2 2
1176 CCHCR1 6_25 2.559e-02 59.77 5.477e-07 -9.032 11 18
6952 TNXB 6_26 2.108e-13 454.39 1.918e-28 9.001 4 5
3026 HLA-DMA 6_27 9.405e-02 70.57 5.975e-06 8.860 5 6
7849 ZSCAN23 6_22 1.294e-02 46.07 7.324e-08 -8.541 1 1GO enrichment analysis for genes with PIP>0.5
#number of genes for gene set enrichment
length(genes)[1] 34Uploading data to Enrichr... Done.
Querying GO_Biological_Process_2021... Done.
Querying GO_Cellular_Component_2021... Done.
Querying GO_Molecular_Function_2021... Done.
Parsing results... Done.
[1] "GO_Biological_Process_2021"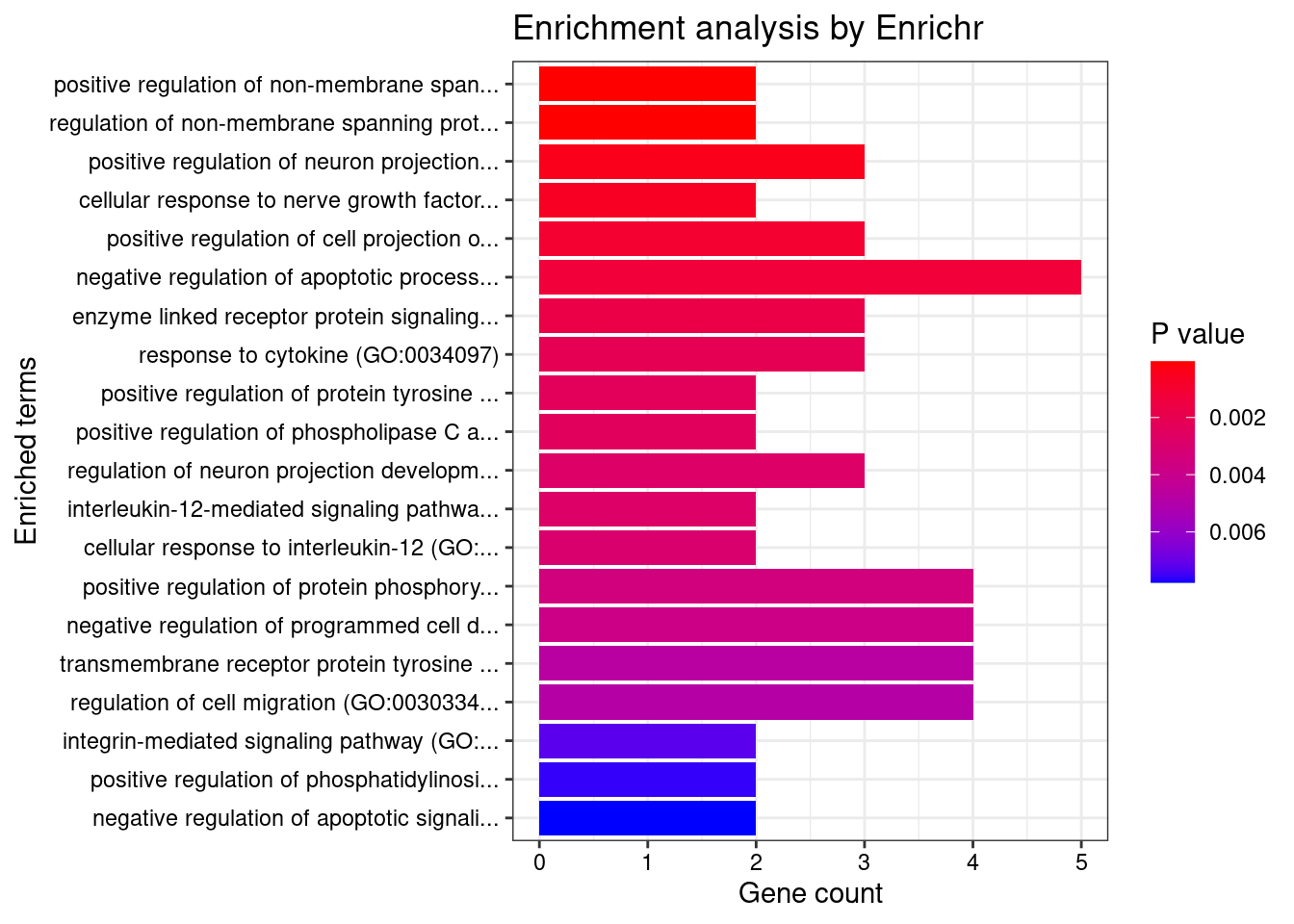
Term
1 positive regulation of non-membrane spanning protein tyrosine kinase activity (GO:1903997)
2 regulation of non-membrane spanning protein tyrosine kinase activity (GO:1903995)
Overlap Adjusted.P.value Genes
1 2/6 0.01482 BDNF;SRC
2 2/7 0.01482 BDNF;SRC
[1] "GO_Cellular_Component_2021"
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)
[1] "GO_Molecular_Function_2021"
[1] Term Overlap Adjusted.P.value Genes
<0 rows> (or 0-length row.names)DisGeNET enrichment analysis for genes with PIP>0.5
Description FDR Ratio BgRatio
73 Status Epilepticus 0.01241 3/21 68/9703
110 Petit mal status 0.01241 3/21 67/9703
118 Grand Mal Status Epilepticus 0.01241 3/21 67/9703
126 Complex Partial Status Epilepticus 0.01241 3/21 67/9703
166 Status Epilepticus, Subclinical 0.01241 3/21 67/9703
167 Non-Convulsive Status Epilepticus 0.01241 3/21 67/9703
168 Simple Partial Status Epilepticus 0.01241 3/21 67/9703
199 TOBACCO ADDICTION, SUSCEPTIBILITY TO (finding) 0.01241 2/21 12/9703
70 Schizophrenia 0.02137 7/21 883/9703
197 AICARDI-GOUTIERES SYNDROME 3 0.02137 1/21 1/9703WebGestalt enrichment analysis for genes with PIP>0.5
Warning: replacing previous import 'lifecycle::last_warnings' by
'rlang::last_warnings' when loading 'hms'Loading the functional categories...
Loading the ID list...
Loading the reference list...
Performing the enrichment analysis...
description size overlap FDR database
1 Bipolar Disorder 136 7 0.003887 disease_GLAD4U
userId
1 BDNF;CAMKK2;GABBR2;NTRK3;SDCCAG8;TRANK1;TSNARE1PIP Manhattan Plot
Warning: ggrepel: 1 unlabeled data points (too many overlaps). Consider
increasing max.overlaps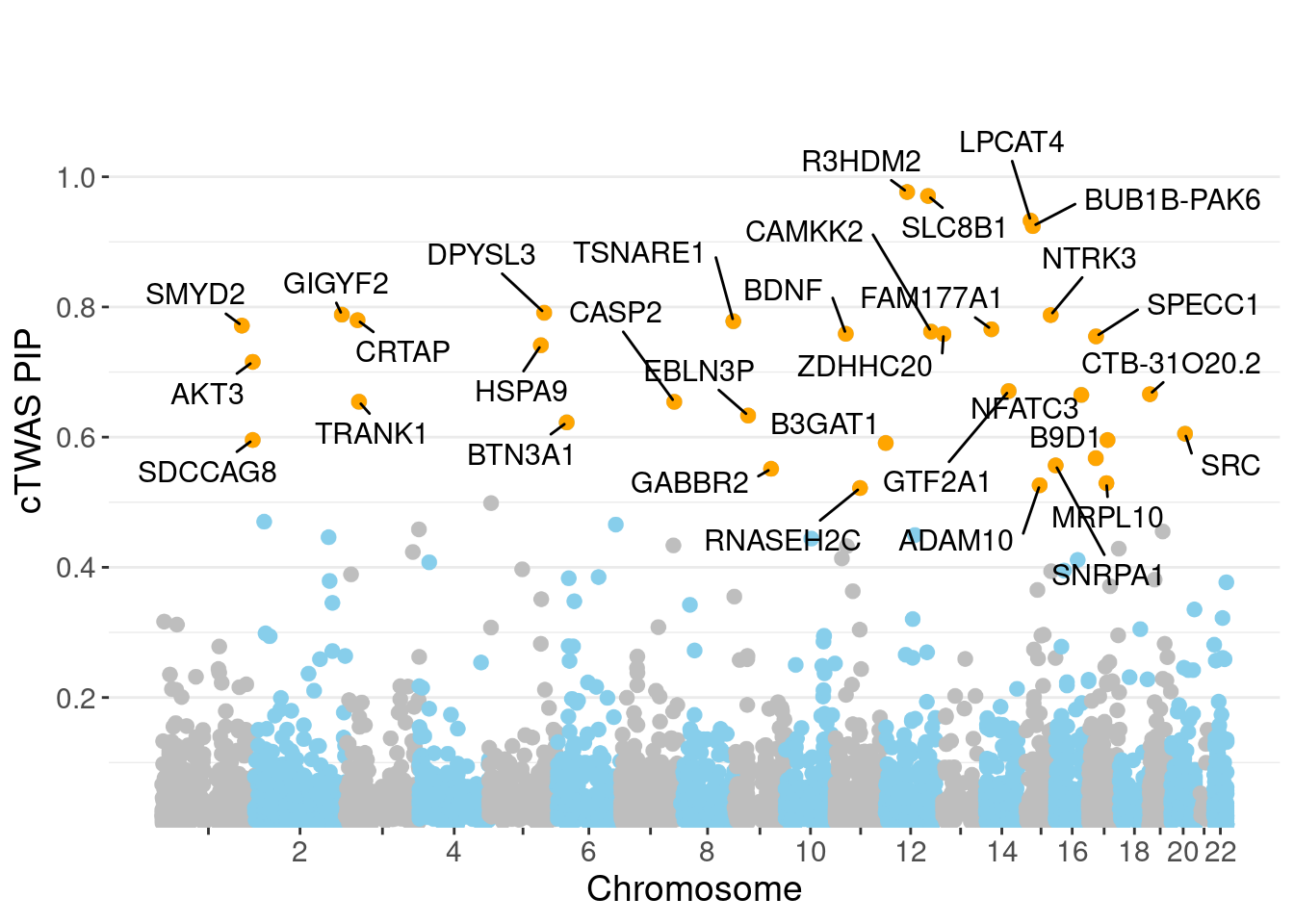
Sensitivity, specificity and precision for silver standard genes
#number of genes in known annotations
print(length(known_annotations))[1] 130#number of genes in known annotations with imputed expression
print(sum(known_annotations %in% ctwas_gene_res$genename))[1] 59#significance threshold for TWAS
print(sig_thresh)[1] 4.514#number of ctwas genes
length(ctwas_genes)[1] 4#number of TWAS genes
length(twas_genes)[1] 171#show novel genes (ctwas genes with not in TWAS genes)
ctwas_gene_res[ctwas_gene_res$genename %in% novel_genes,report_cols] genename region_tag susie_pip mu2 PVE z num_intron num_sqtl
6205 SLC8B1 12_68 0.9706 28.59 0.0003598 -4.047 11 12#sensitivity / recall
print(sensitivity) ctwas TWAS
0.007692 0.130769 #specificity
print(specificity) ctwas TWAS
0.9996 0.9803 #precision / PPV
print(precision) ctwas TWAS
0.25000 0.09942
sessionInfo()R version 4.1.0 (2021-05-18)
Platform: x86_64-pc-linux-gnu (64-bit)
Running under: Scientific Linux 7.4 (Nitrogen)
Matrix products: default
BLAS/LAPACK: /software/openblas-0.3.13-el7-x86_64/lib/libopenblas_haswellp-r0.3.13.so
locale:
[1] LC_CTYPE=en_US.UTF-8 LC_NUMERIC=C
[3] LC_TIME=en_US.UTF-8 LC_COLLATE=en_US.UTF-8
[5] LC_MONETARY=en_US.UTF-8 LC_MESSAGES=en_US.UTF-8
[7] LC_PAPER=en_US.UTF-8 LC_NAME=C
[9] LC_ADDRESS=C LC_TELEPHONE=C
[11] LC_MEASUREMENT=en_US.UTF-8 LC_IDENTIFICATION=C
attached base packages:
[1] stats graphics grDevices utils datasets methods base
other attached packages:
[1] readxl_1.4.0 forcats_0.5.1 stringr_1.4.0 purrr_0.3.4
[5] readr_1.4.0 tidyr_1.1.3 tidyverse_1.3.1 tibble_3.1.7
[9] WebGestaltR_0.4.4 disgenet2r_0.99.2 enrichR_3.0 cowplot_1.1.1
[13] ggplot2_3.3.5 dplyr_1.0.7 reticulate_1.25 workflowr_1.7.0
loaded via a namespace (and not attached):
[1] fs_1.5.0 lubridate_1.7.10 doParallel_1.0.16 httr_1.4.2
[5] rprojroot_2.0.2 tools_4.1.0 backports_1.2.1 doRNG_1.8.2
[9] bslib_0.2.5.1 utf8_1.2.1 R6_2.5.0 vipor_0.4.5
[13] DBI_1.1.1 colorspace_2.0-2 withr_2.4.2 ggrastr_1.0.1
[17] tidyselect_1.1.1 processx_3.5.2 curl_4.3.2 compiler_4.1.0
[21] git2r_0.28.0 rvest_1.0.0 cli_3.0.0 Cairo_1.5-15
[25] xml2_1.3.2 labeling_0.4.2 sass_0.4.0 scales_1.1.1
[29] callr_3.7.0 systemfonts_1.0.4 apcluster_1.4.9 digest_0.6.27
[33] rmarkdown_2.9 svglite_2.0.0 pkgconfig_2.0.3 htmltools_0.5.1.1
[37] dbplyr_2.1.1 highr_0.9 rlang_1.0.2 rstudioapi_0.13
[41] jquerylib_0.1.4 farver_2.1.0 generics_0.1.0 jsonlite_1.7.2
[45] magrittr_2.0.1 Matrix_1.3-3 ggbeeswarm_0.6.0 Rcpp_1.0.7
[49] munsell_0.5.0 fansi_0.5.0 lifecycle_1.0.0 stringi_1.6.2
[53] whisker_0.4 yaml_2.2.1 plyr_1.8.6 grid_4.1.0
[57] ggrepel_0.9.1 parallel_4.1.0 promises_1.2.0.1 crayon_1.4.1
[61] lattice_0.20-44 haven_2.4.1 hms_1.1.0 knitr_1.33
[65] ps_1.6.0 pillar_1.7.0 igraph_1.2.6 rjson_0.2.20
[69] rngtools_1.5 reshape2_1.4.4 codetools_0.2-18 reprex_2.0.0
[73] glue_1.4.2 evaluate_0.14 getPass_0.2-2 modelr_0.1.8
[77] data.table_1.14.0 png_0.1-7 vctrs_0.3.8 httpuv_1.6.1
[81] foreach_1.5.1 cellranger_1.1.0 gtable_0.3.0 assertthat_0.2.1
[85] xfun_0.24 broom_0.7.8 later_1.2.0 iterators_1.0.13
[89] beeswarm_0.4.0 ellipsis_0.3.2 here_1.0.1